Ishanu Chattopadhyay
Assistant Professor of Medicine
UChicago
Dmytro Onishchenko
UChicago
Rapid Universal Point-of-care Screening for ILD/IPF Using Comorbidity Signatures in Electronic Health Records
University of Chicago Medicine


NHLBI IPF Stakeholder Summit
Nov 2022
Fernando Martinez, Weill Cornell
Gary Hunninghake
Harvard Med School
Andrew Limper Mayo Clinic




shortness of breath
dry cough
doctor can hear velcro crackles
Common Symptoms
>50 years old
more men than women
IPF
Rare disease
~5 in 10,000
Post-Dx
Survival
~4 years
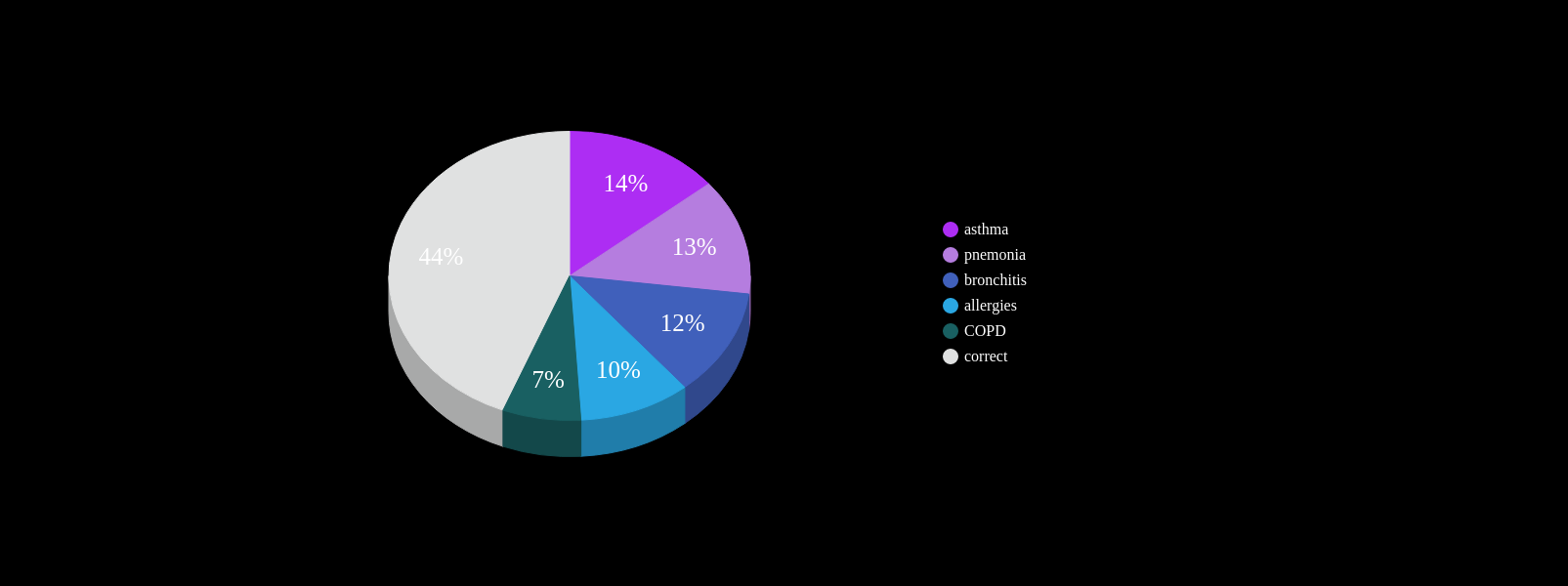
At least one misdiagnosis
~55%
Two or more misdiagnosis
38%
Initially attributed to age related symptoms:
72%

Cannot always be seen on CXR
Non-specific symptoms
PCP workflow demands

~ 4yrs
current survival ~4yrs
~ 4yrs
current clinical DX
ZCoR screening



Onishchenko, D., Marlowe, R.J., Ngufor, C.G. et al. Screening for idiopathic pulmonary fibrosis using comorbidity signatures in electronic health records. Nat Med 28, 2107–2116 (2022). https://doi.org/10.1038/s41591-022-02010-y
n=~3M
AUC~90%
Likelihood ratio ~30



Conventional AI/ML attempts to model the physician
AI in IPF Research
- Co-morbidity Patterns
- No data demands
- Use whatever data is already on patient file
- Co-morbidity Patterns
- No data demands
- Use whatever data is already on patient file
Primary Care
Pulmonologist
ZCoR Flag
- No blood tests
- No imaging
- No pulmonary function tests
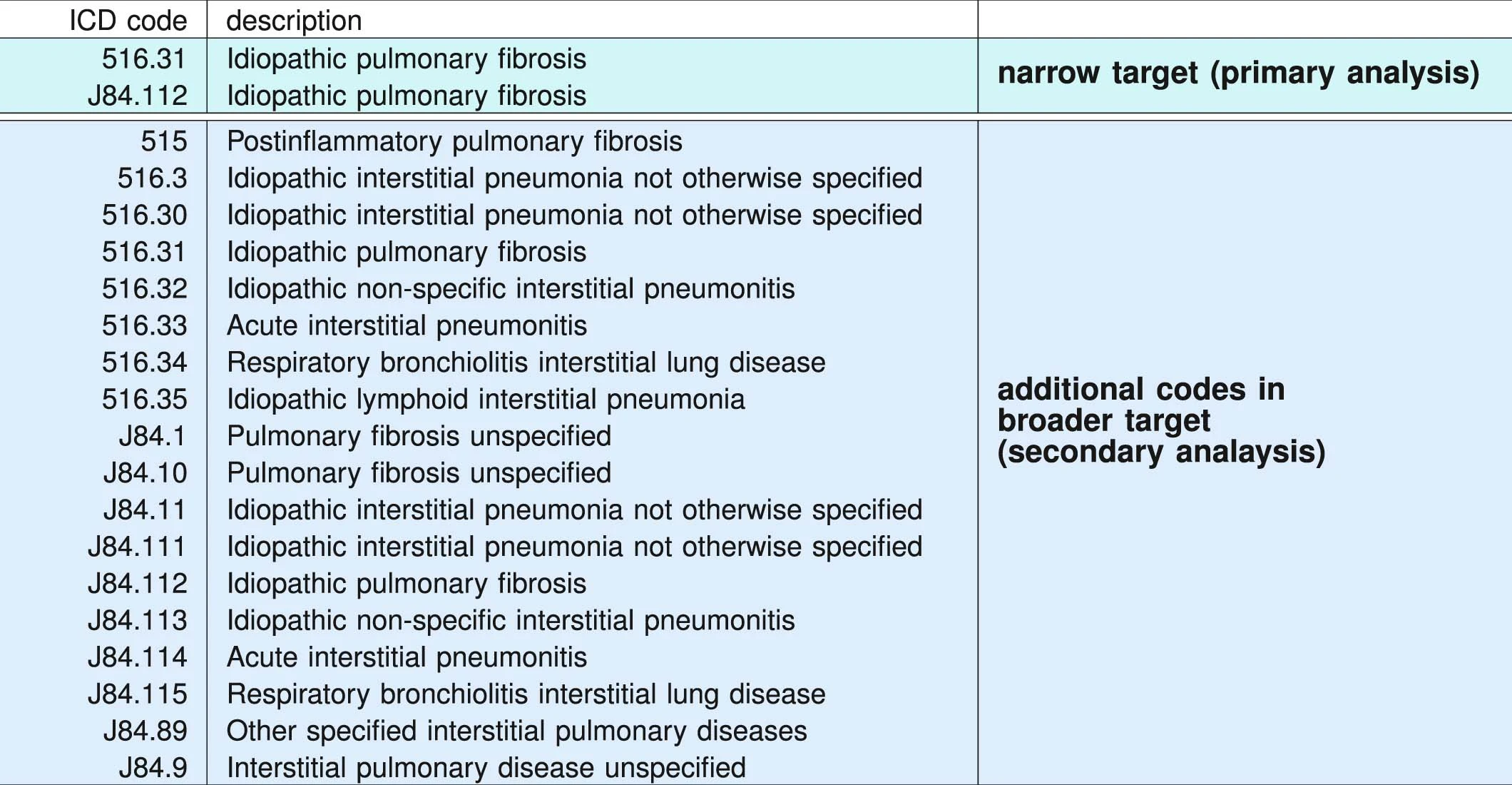
ICD administrative codes

IPF
ILD
target codes appear
Past medical history
No target codes appear
case
control
2yrs
2yrs
target codes appear
Past medical history
No target codes appear
case
control
2yrs
2yrs
IPF drugs prescribed
Signature of IPF diagnostic sequence
pirfenidone or nintedanib
- age > 50 years
- at least two IPF target codes identified at least 1 month apart
- chest CT procedure (ICD-9-CM 87.41 and Current Procedural Terminology, 4th Edition, codes 71250, 71260 and 71270) before the first diagnostic claim for IPF
- no claims for alternative ILD codes occurring on or after the first IPF claim
Truven MarketScan (IBM) Commerical Claims & Encounters Database 2003-2018
>100M patients visible
>7B individual claims
>87K unique diagnostic codes
>7% Medicare data present
2,053,277 patients included in study

Univesity of Chicago Medicam Center 2012-2021
68,658 patients
Random sample from Optumlabs Data Warehouse courtsey Mayo Clinic
861,280 patients
2,983,215 patients
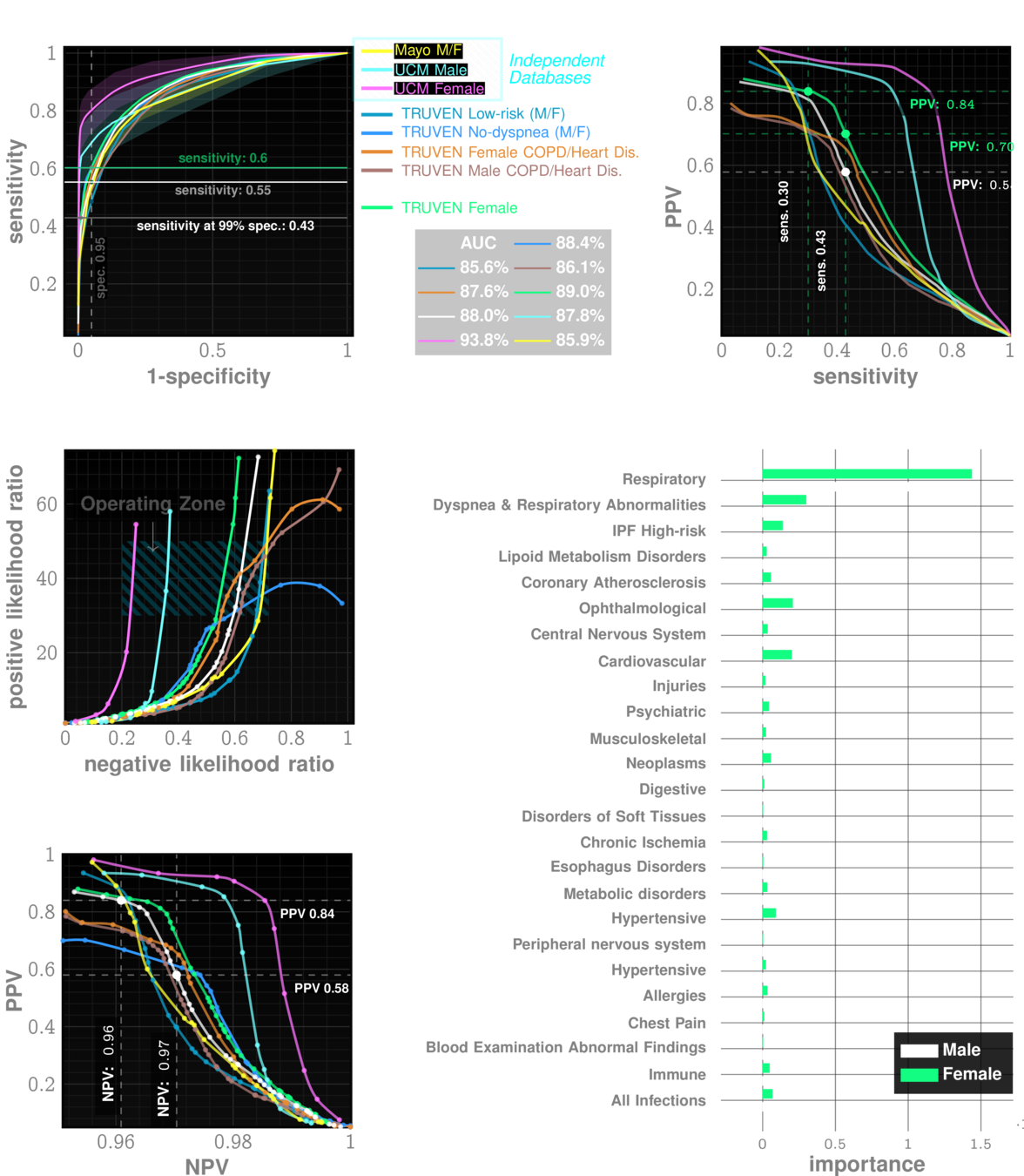

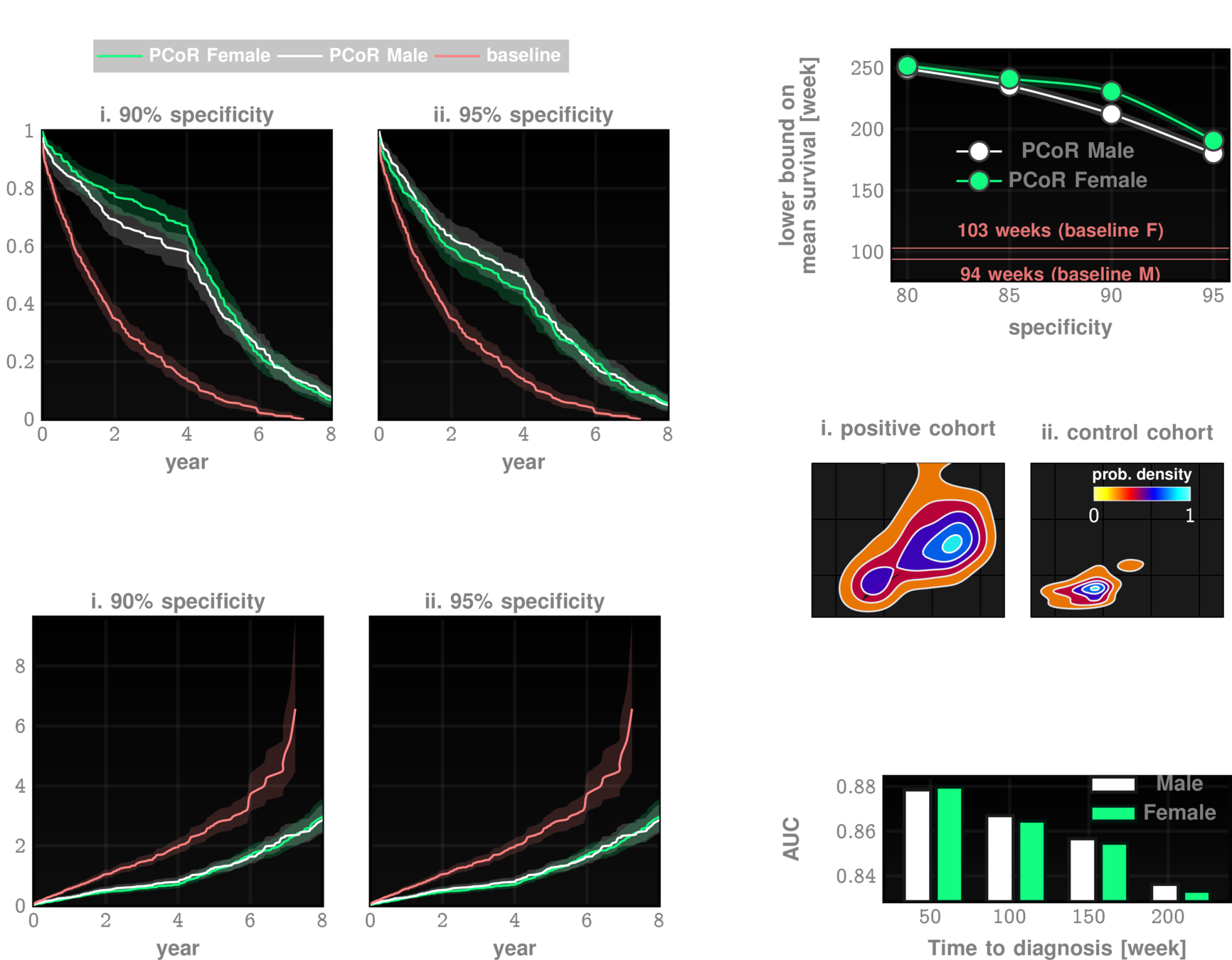
performance tables
Marketscan Out-of-sample Results
specificty~99%
NPV>99.9%
IPF
ILD
performance tables
UCM Out-of-sample Results
specificty~99%
NPV>99.9%
IPF
ILD




False Positives:
- Heathcare Capacity
Ethics:
- Risk from Imaging Tests
For every 20-30 flags,
1 is positive
- General likelihood ratio 60-80
- PPV 3.5-5%
- Notifying patients 4 years early?
- No cure, why screen
minimal
acceptable?
Better outcomes
- early anti-fibrotic therapy seems increasingly promising
- better shot at lung transplant
- early dx reduces hospital-izations by a factor of 1-3
Collard, Harold R., Alex J. Ward, Stephan Lanes, D. Cortney Hayflinger, Daniel M. Rosenberg, and Elke Hunsche. "Burden of illness in idiopathic pulmonary fibrosis." Journal of medical economics 15, no. 5 (2012): 829-835.
Future
- Seamless Embedding in EHR system (EPIC App)
- Pragmatic Clinical Trial in Primary Care Setting
- Track impact from initial flag to diagnosis with regards to patient outcome and healthcare utilization
- Develop extended ZCoR version which uses data available after initial flag, such as CXR outcomes, and clinical notes
The Team
Gary Hunninghake, Pulmonary C, Harvard


Fernando Martinez, Pulmonary Critical Care, Weill Cornell
Director, Thoracic Research Unit, Mayo Clinic





Backup Slides
Method Details
Longitudinal history is important, cannot simply process snapshots
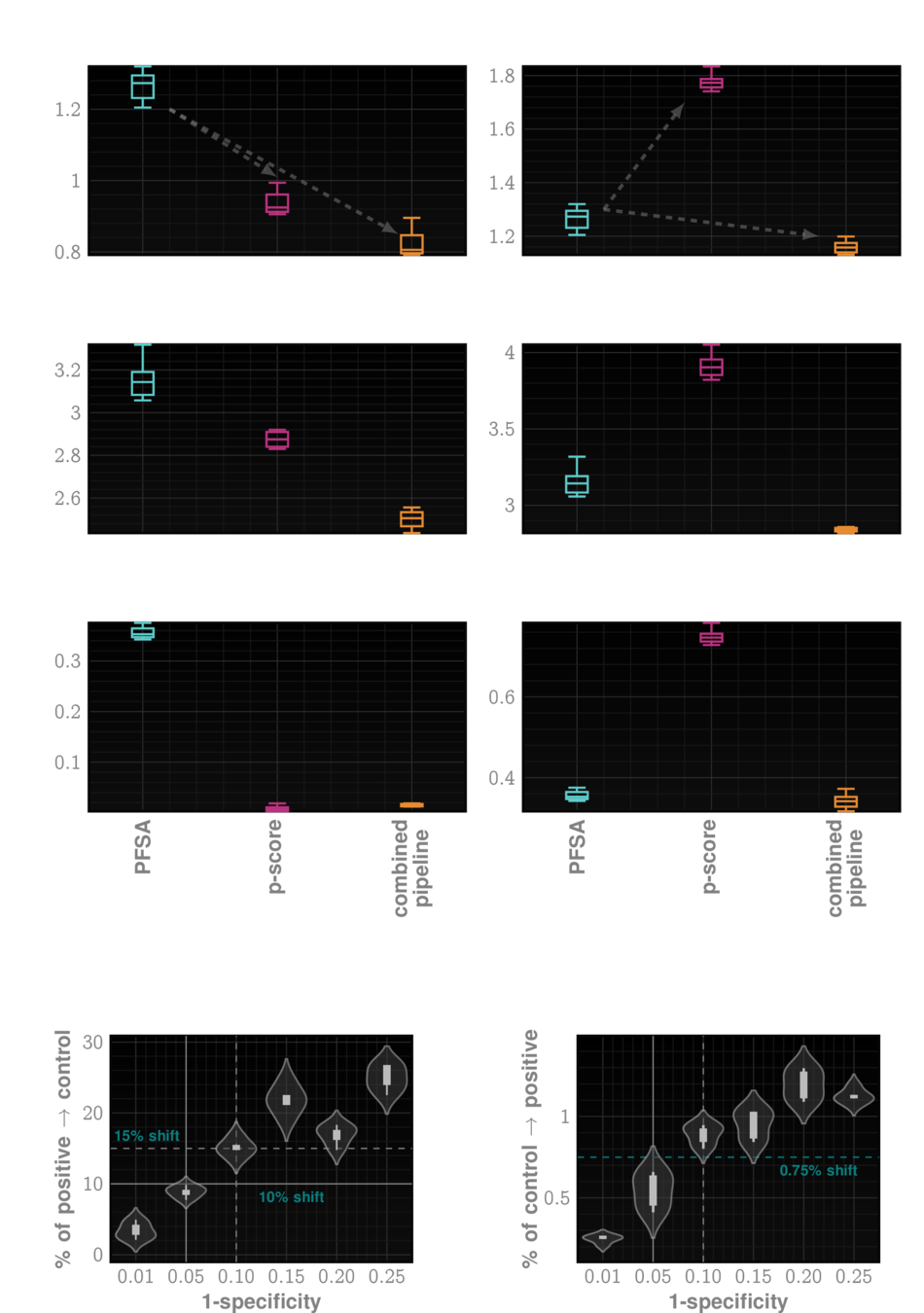
* For IPF screening
*



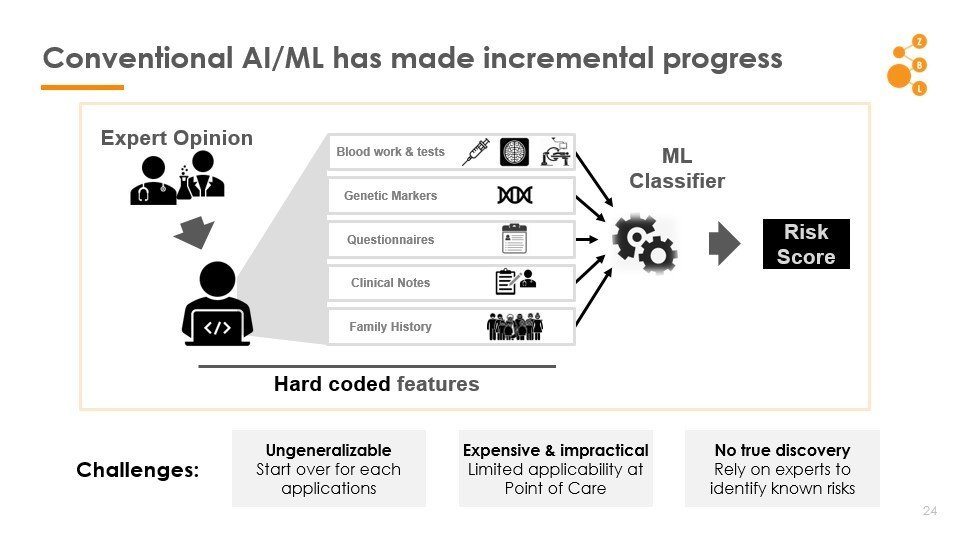
Conventional AI/ML has made incremental progress
Challenges
Non-generalizable
Start-over for each application
Expensive
Limited applicability at point of care
No true discovery
Rely on experts to leverage known risks
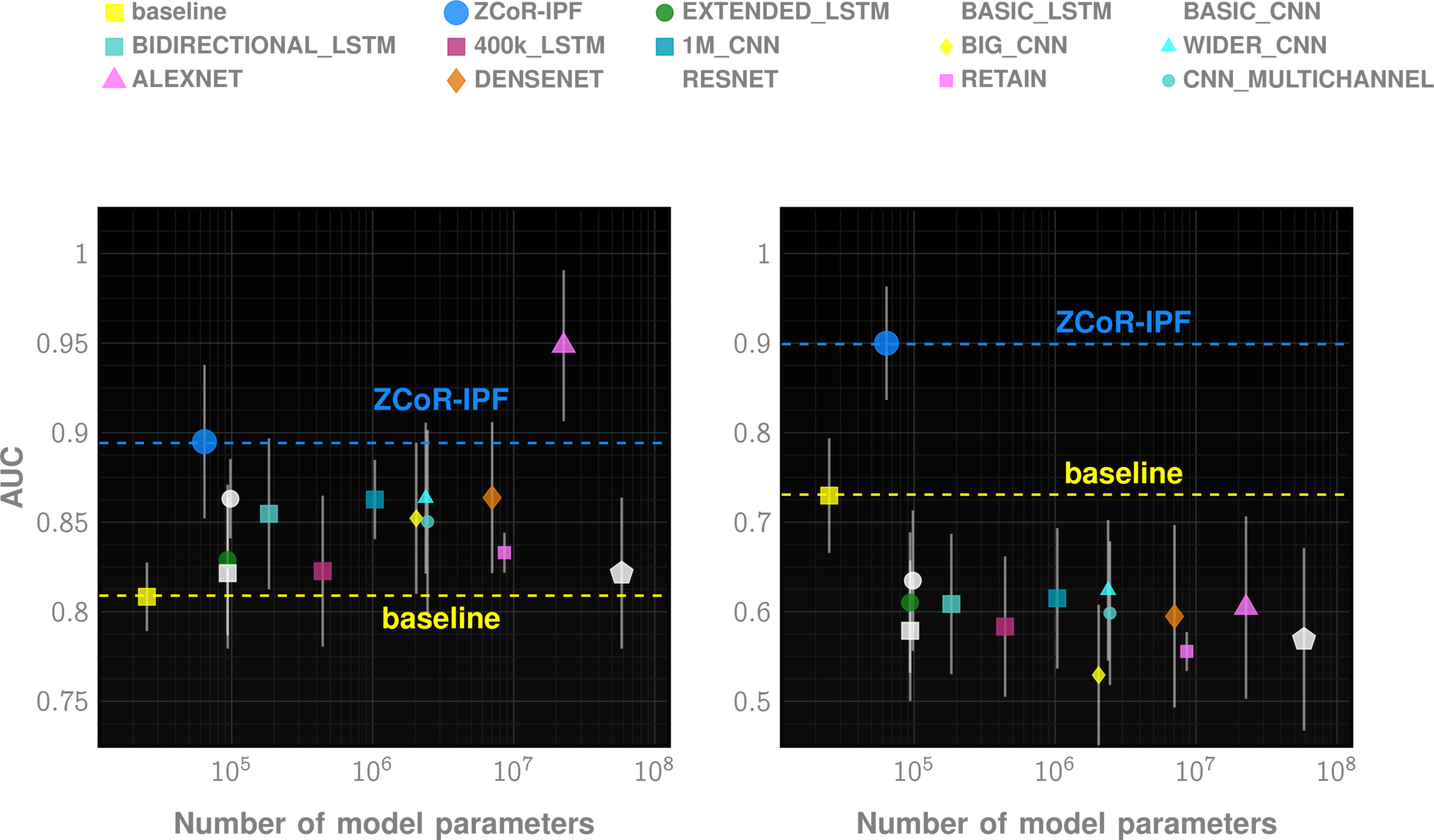
Leveraging Longitudinal Patterns
Specialized HMM models from code sequences
Model control and case cohorts seprately
given a new test case, compute likelihood of sample arising from case models vs control models
sequence likelihood defect
ZeD Lab: Predictive Screening from Comorbidity Footprints



Nature Medicine
JAHA
CELL Reports
Science Adv.
ZeD Lab: Predictive Screening from Comorbidity Footprints
| ZED performance | Competition | |
|---|---|---|
| Autism* | >80% AUC at 2 yrs | Double false positives |
| Alzheimer's Disease | ~90% AUC | 60-70% AUC |
| Idiopathic Pulmonary Fibrosis | ~90% AUC | NA |
| MACE | ~80% AUC | ~70% AUC |
| Bipolar Disorder | ~85% AUC | NA |
| CKD | ~85% AUC | NA |
| Cancers | ~75% AUC | NA |
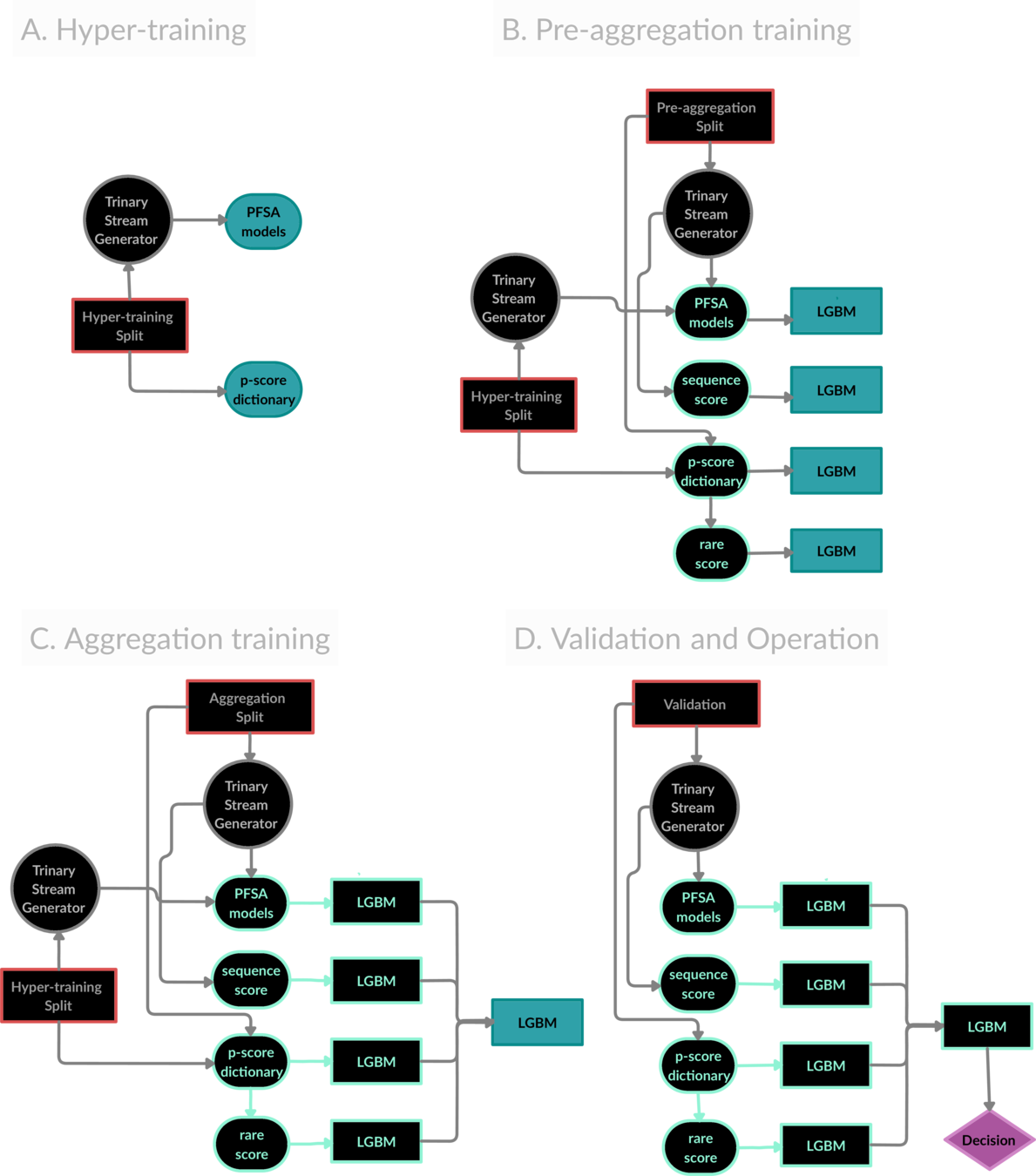
The ZeD Pipeline prototype for risk estimation from co-morbidity signatures
Primary Care
Risk
No additional tests


Bio-aware Generalizable Feature Engineering from co-morbidities
Clinically Useful
Advance Science
Bio-AI
Machine Learning
Information Theory
Economics
Healthcare Policy
Ethics
Comorbidities
Unknown Risk factors
Known risks
Knowledge of underlying genetic and epigenetic pathways