Interpreting the evolution of
SARS-CoV-2 in context of Omicron
Jesse Bloom
Fred Hutch Cancer Center / HHMI
Slides at https://slides.com/jbloom/sars-cov-2-evolution-long
Disclosures
- I am on the scientific advisory boards of Apriori Bio and Oncorus
- I have had consulting arrangements with Moderna and Merck
- I am an inventor on Fred Hutch licensed patents related to deep mutational scanning of viral proteins
- My lab has unfunded research collaborations with Vir Biotechnology
Outline
- Principles of viral antigenic evolution and emergence of Omicron
Outline
- Principles of viral antigenic evolution and emergence of Omicron
- Importance of the RBD
Outline
- Principles of viral antigenic evolution and emergence of Omicron
- Importance of the RBD
- Antibody escape in Omicron
Outline
- Principles of viral antigenic evolution and emergence of Omicron
- Importance of the RBD
- Antibody escape in Omicron
Only sometimes does virus evolution lead to changes in antigenic phenotype
- Measles virus: Does not evolve to escape immunity. People are infected at most once in their lives. A vaccine developed in the 1960s still works today.
- Influenza virus: Evolves to escape immunity. People are infected every ~5 years. The vaccine needs to be updated annually.

We decided to look at another human coronavirus: CoV-229E causes common colds and has been circulating in humans since at least 1960s.
Reconstructing evolution of CoV-229E spike

We experimentally generated CoV-229E spikes at ~8 year intervals so we could study them in the lab:
- 1984
- 1992
- 2001
- 2008
- 2016
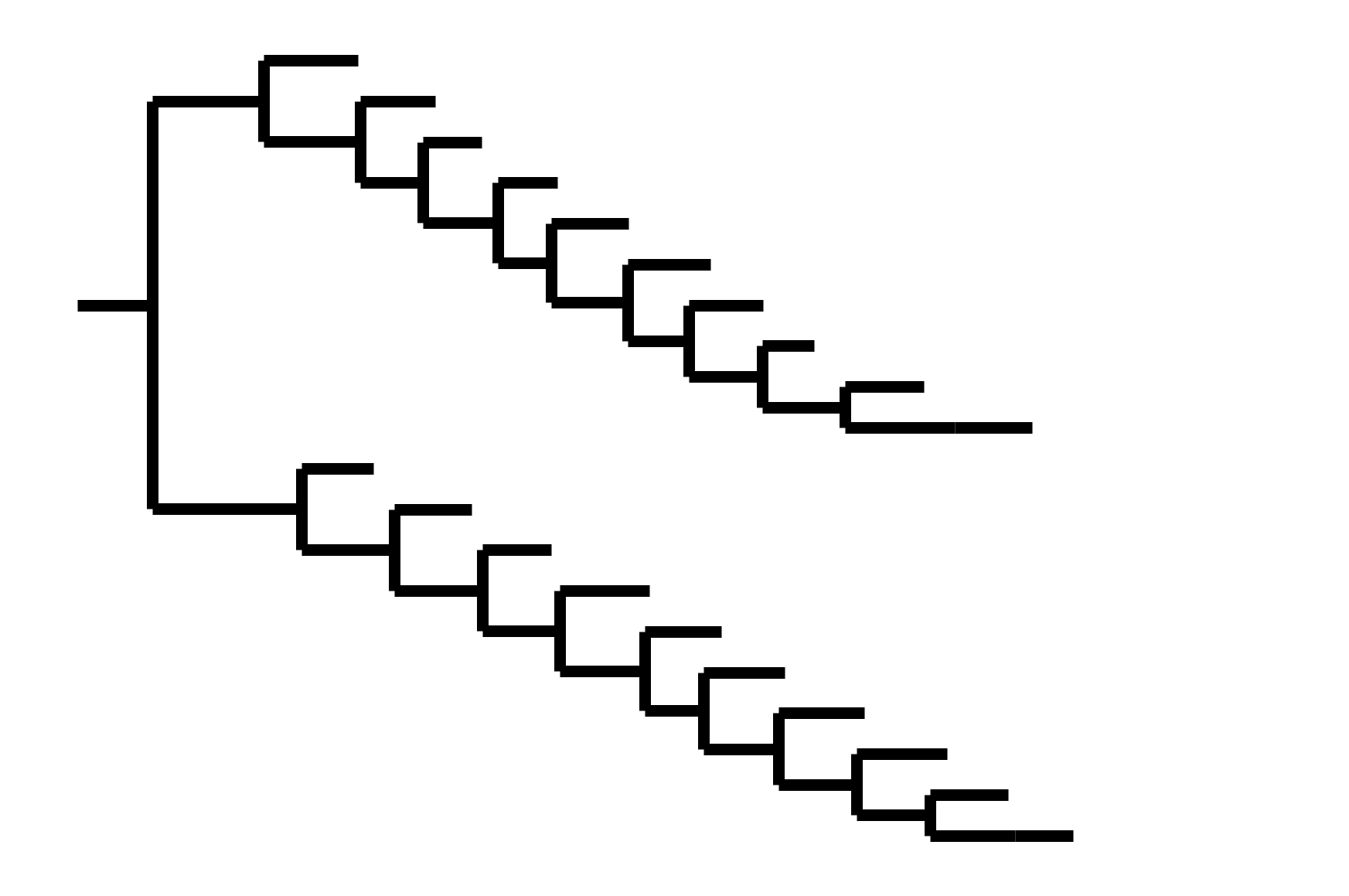
Note "ladder-like" shape of tree
Evolution of CoV-229E spike erodes neutralization by human antibody immunity
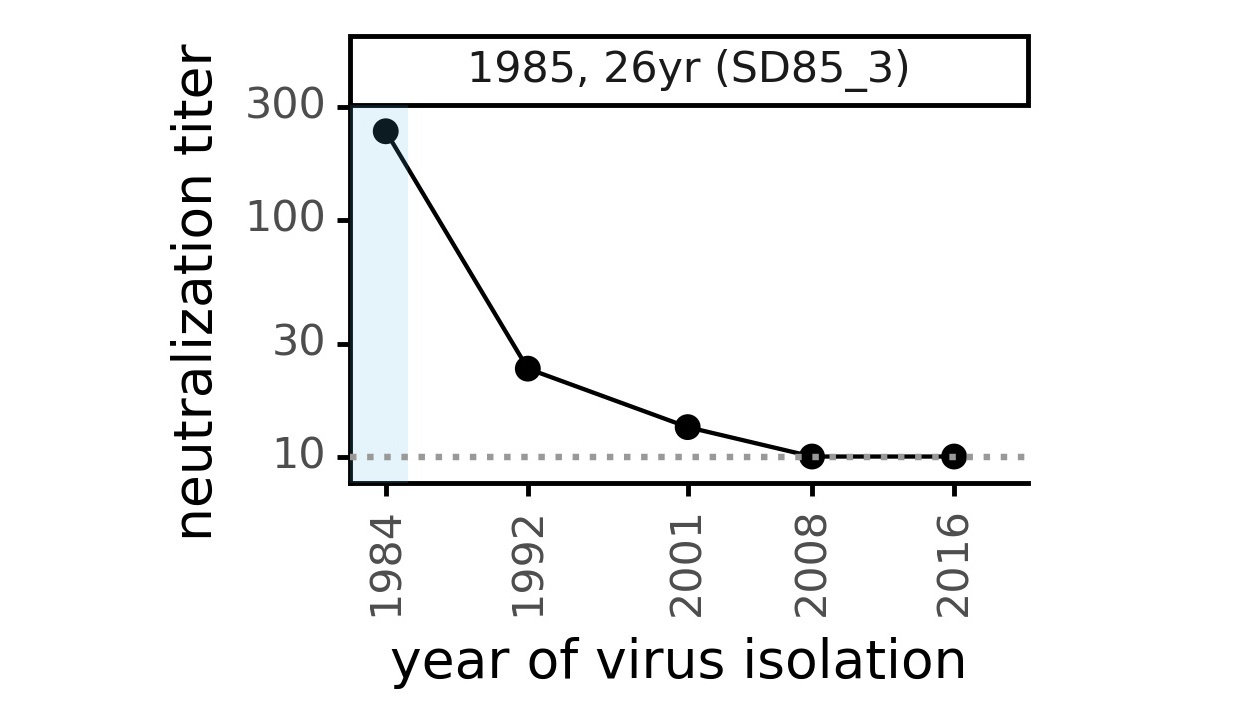
Serum collected in 1985 neutralizes virus with spike from 1984, but less effective against more recent viruses.
Viral evolution erodes antibody immunity of different people at different rates

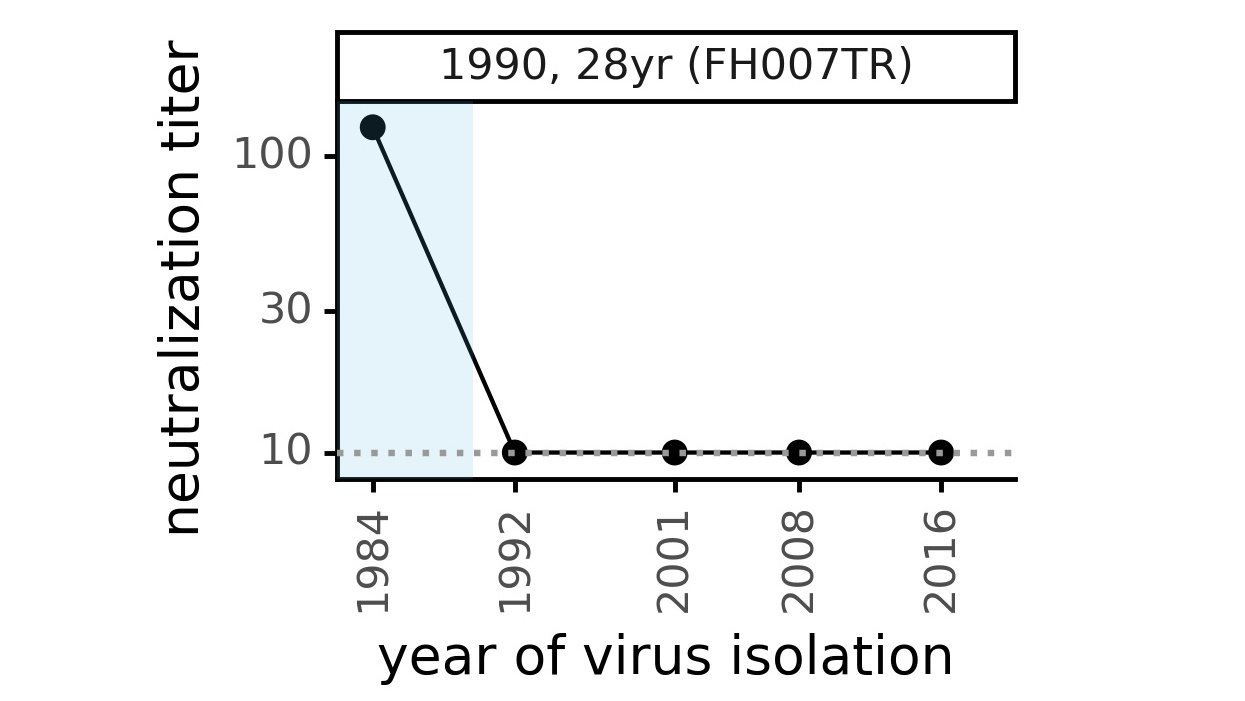
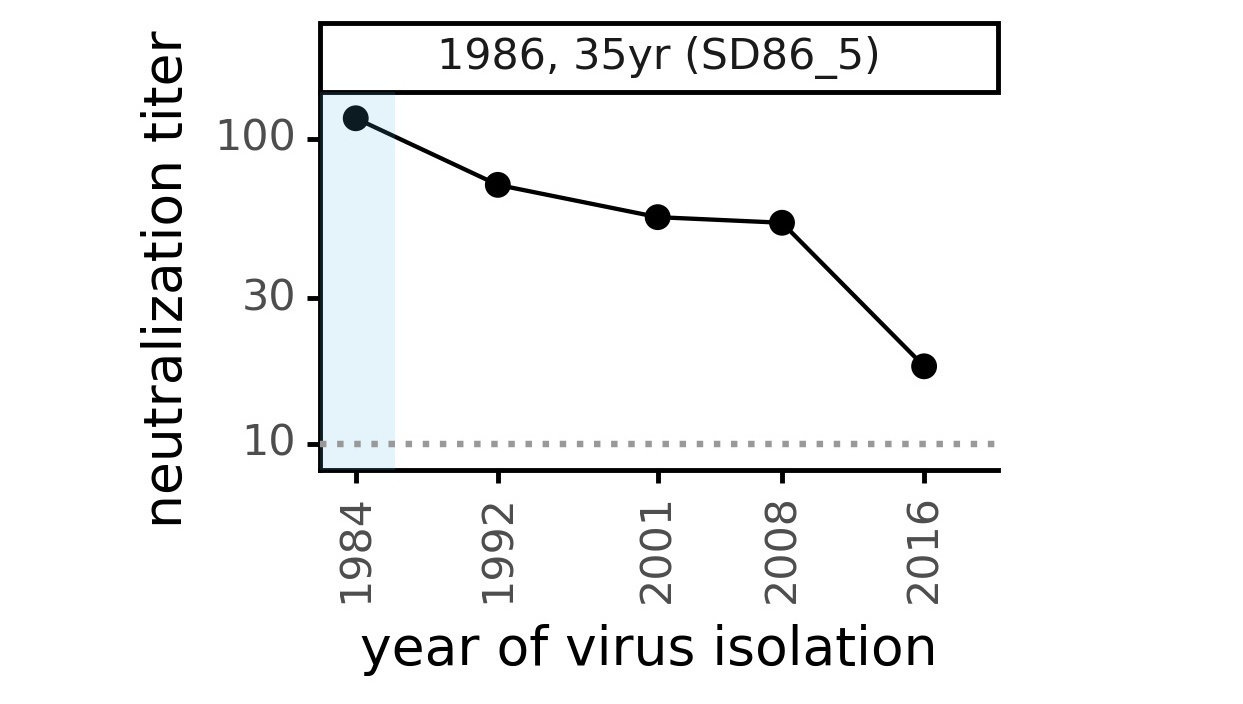
We are studying basis of these differences, as ideally vaccines would elicit more evolution-resistant sera as on the right.
Phylogenetic tree shape and vaccine strategy
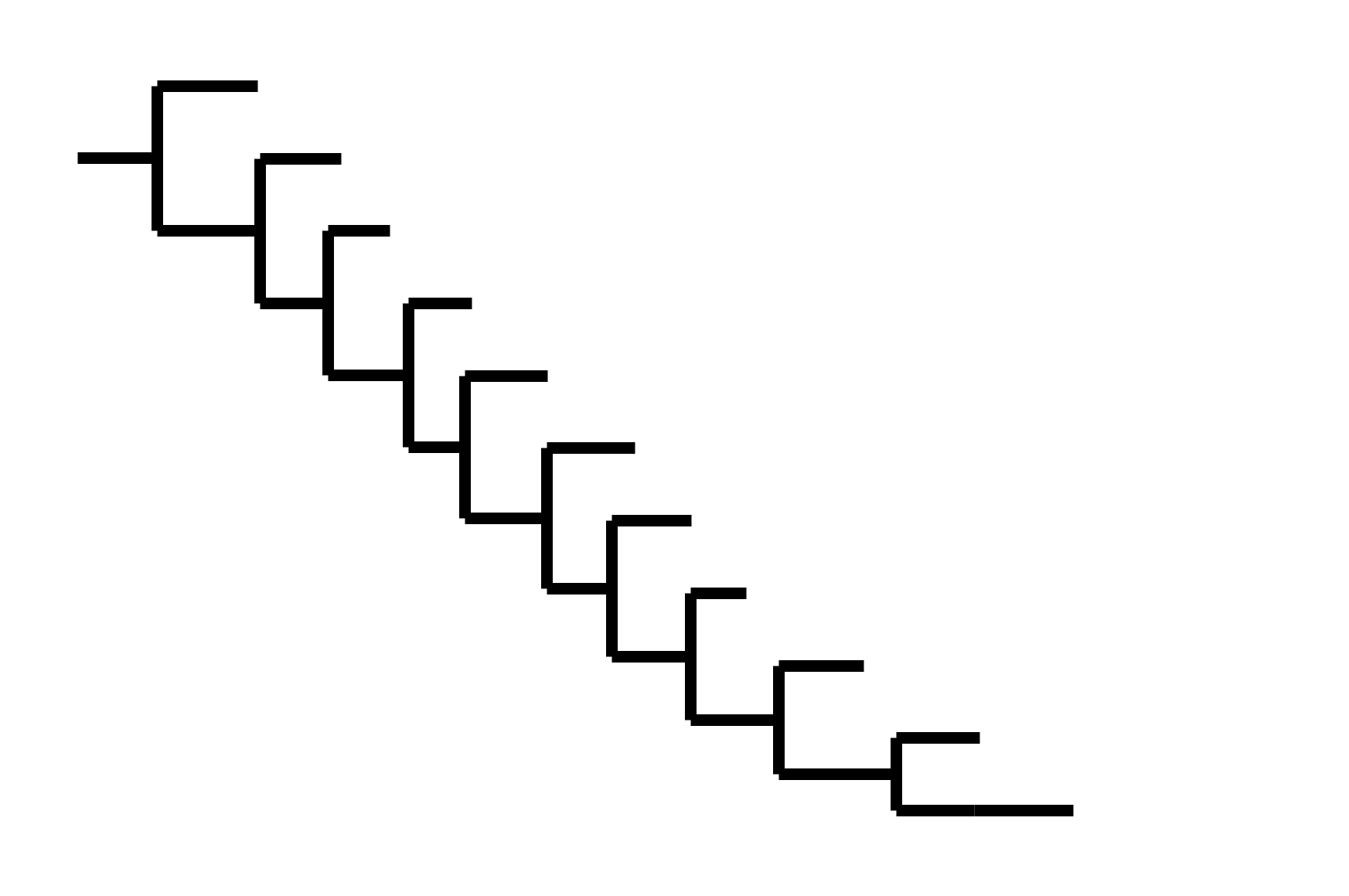
CoV-229E has ladder-like tree:
- new variants displace old ones
- new variants descend from recent successful ones
Human influenza A evolves this way too. It's theoretically possible to pick single well-matched vaccine strain.
Phylogenetic tree shape and vaccine strategy

CoV-229E has ladder-like tree:
- new variants displace old ones
- new variants descend from recent successful ones
Human influenza A evolves this way too. It's theoretically possible to pick single well-matched vaccine strain.
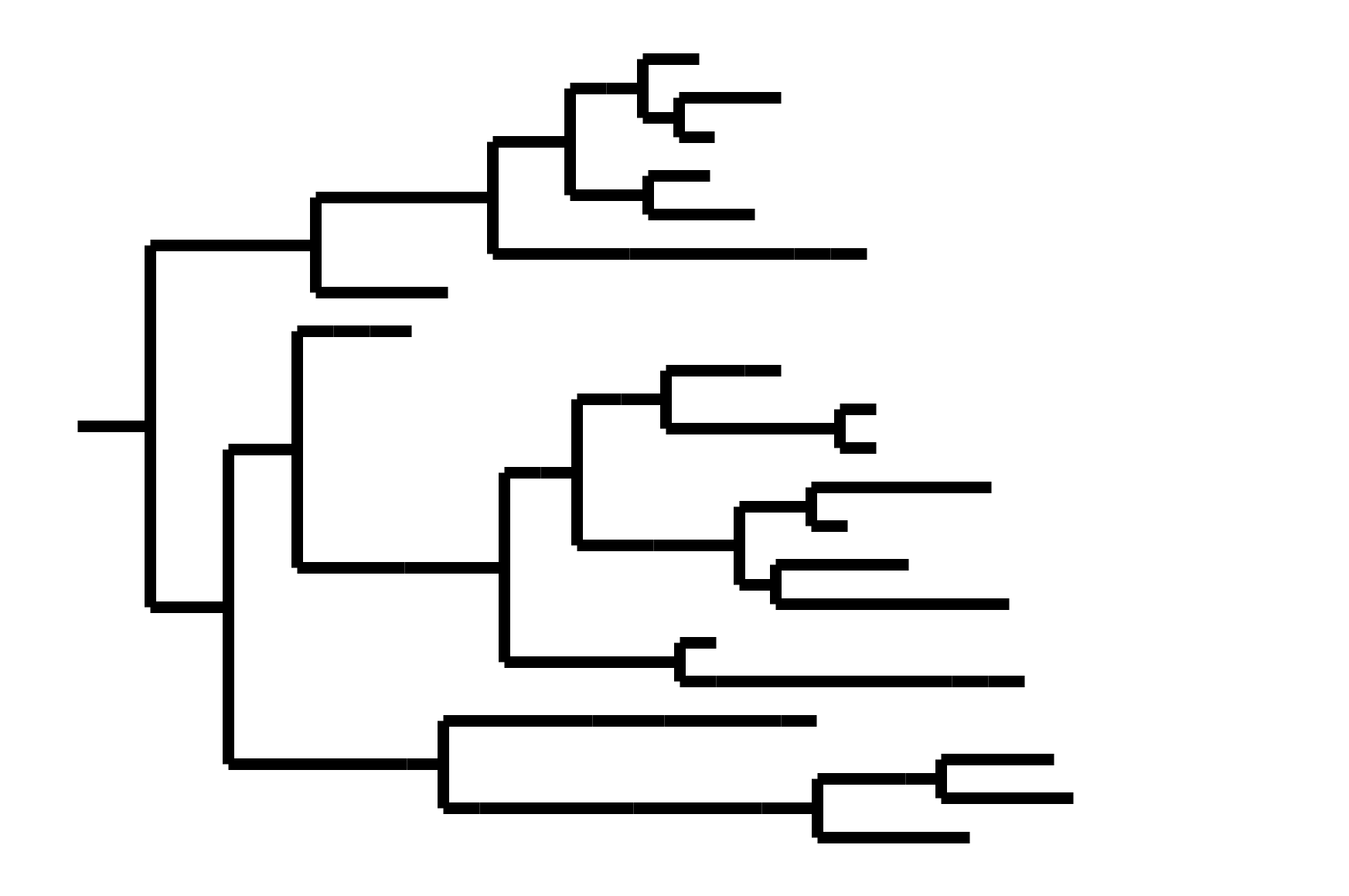
CoV-OC43 split into two ladder-like lineages. Influenza B evolves this way too. It's theoretically possible to pick well-matched bivalent vaccine.
Phylogenetic tree shape and vaccine strategy


CoV-229E has ladder-like tree:
- new variants displace old ones
- new variants descend from recent successful ones
Human influenza A evolves this way too. It's theoretically possible to pick single well-matched vaccine strain.
CoV-OC43 split into two ladder-like lineages. Influenza B evolves this way too. It's theoretically possible to pick well-matched bivalent vaccine.
In non-ladder-like tree, next variant not descended from recent successful one. Makes picking vaccine strains difficult.
After early fixation of D614G, the SARS-CoV-2 tree has not been ladder-like

Take-aways on general principles
Other antigenically evolving human respiratory viruses have ladder-like phylogenetic trees, which is key to enabling vaccine updates as it provides some level of predictability (the next variant usually descended from the last one).
So far SARS-CoV-2 has defied ladder-like paradigm: Omicron not descended from Delta; Delta not descended from Alpha. However, it's still early and we are seeing combined selection for antibody escape and increased transmissibility--presumably transmissibility will eventually plateau. Despite having been wrong in thinking next variant would come from Delta, I still think evolution will eventually become ladder-like.
Outline
- Principles of viral antigenic evolution and emergence of Omicron
- Importance of the RBD
- Antibody escape in Omicron
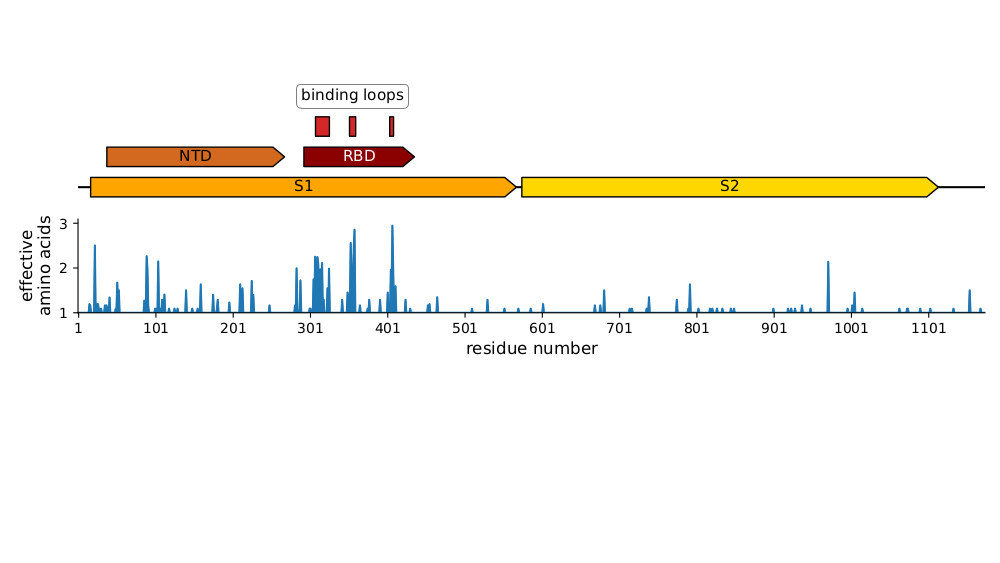
Strongest evolutionary selection is in RBD
Sites of evolutionary change in the spike of CoV-229E over the last four decades
Strongest evolutionary selection is in RBD
Sites of evolutionary change in the spike of CoV-229E over the last four decades

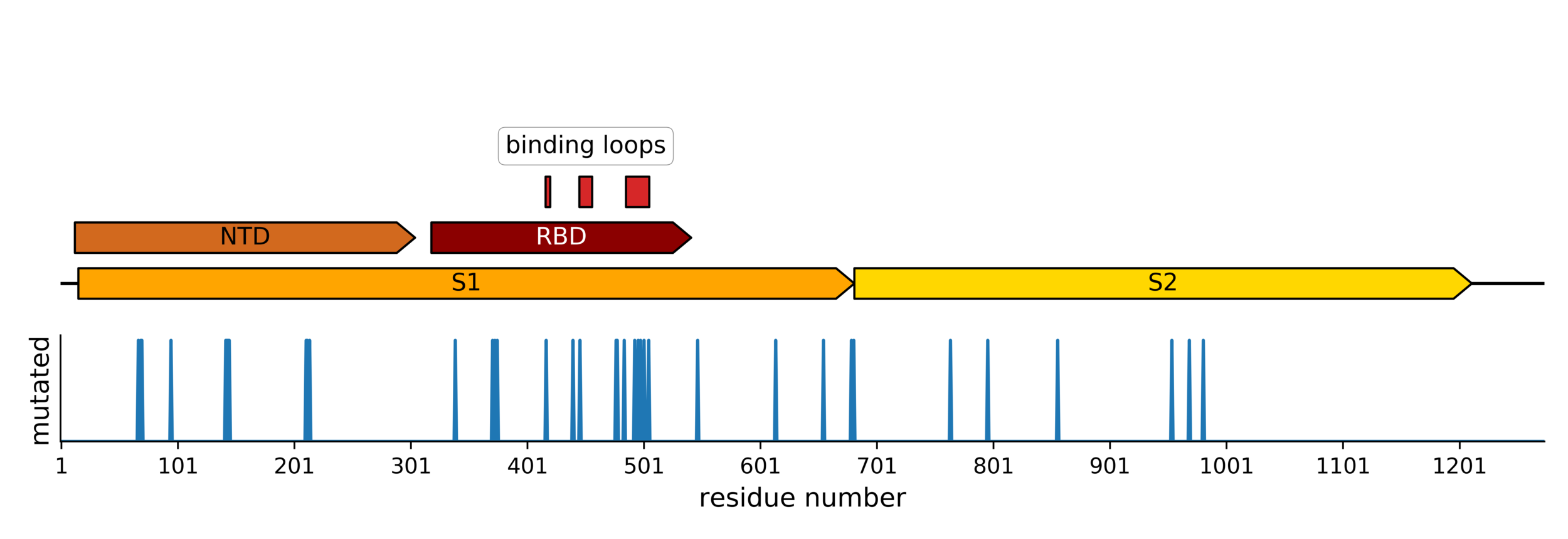
Sites of mutations in SARS-CoV-2 Omicron (BA.1) spike relative to Wuhan-Hu-1
Main difference is SARS-CoV-2 also fixing transmissibility-enhancing spike mutations that affect proteolytic processing and stabilize defects cause by furin-cleavage site
Majority of neutralizing antibody response in vaccinated/infected humans targets RBD
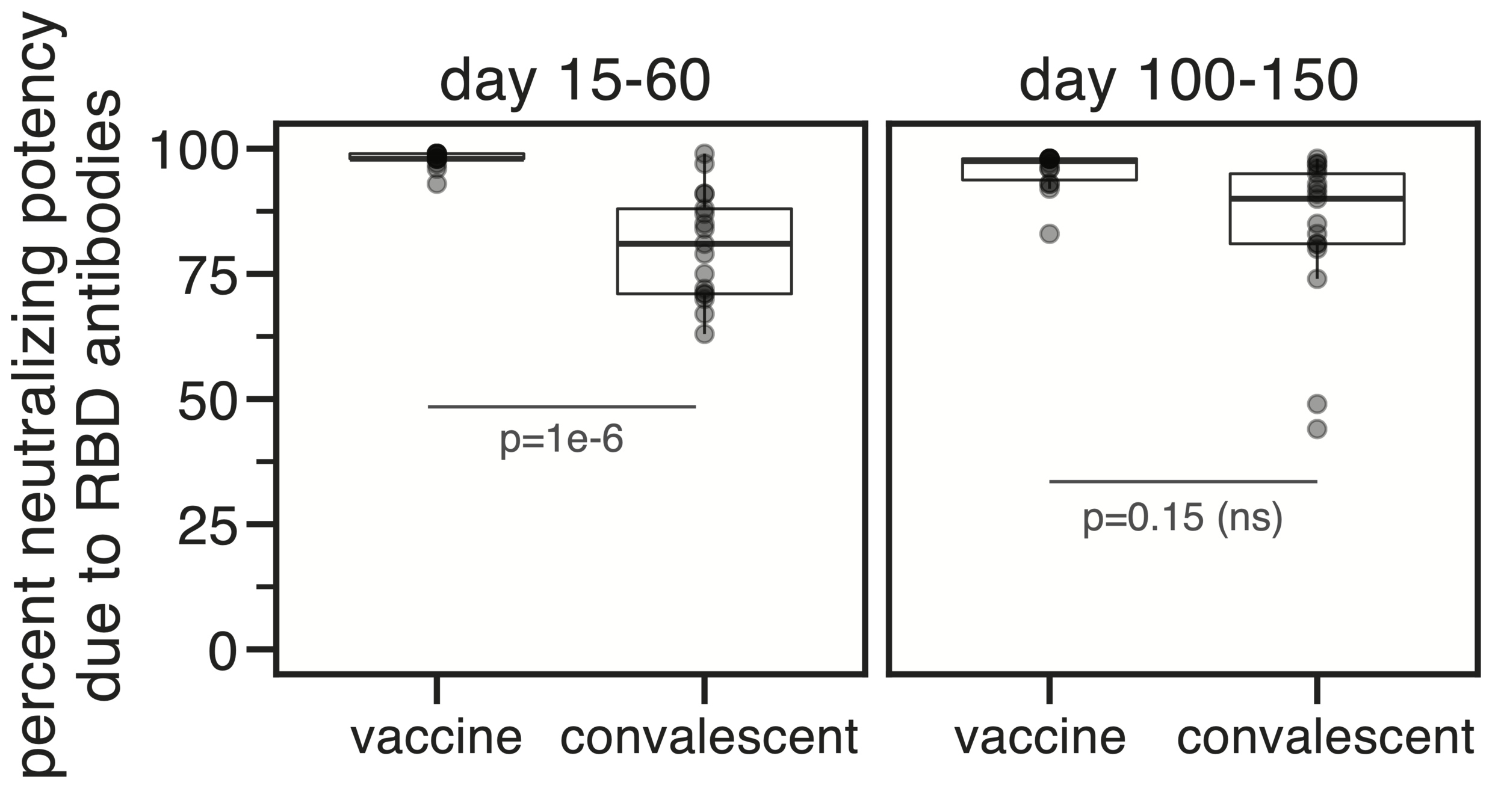
Importance of RBD
Human CoVs, which evolve to escape transmission-blocking immunity, show strongest selection in RBD. So virus is telling us RBD antibodies matter most for blocking transmission. But non-RBD antibodies and T-cells still matter, especially for reducing disease severity while putting less selection on virus.
Most neutralizing activity from RBD antibodies (although antibodies to other domains such as NTD can also be neutralizing).
However, a minority of all anti-spike antibodies elicited by current vaccines target RBD. Given these facts, we should continue to weigh pros / cons of RBD-only vaccines.
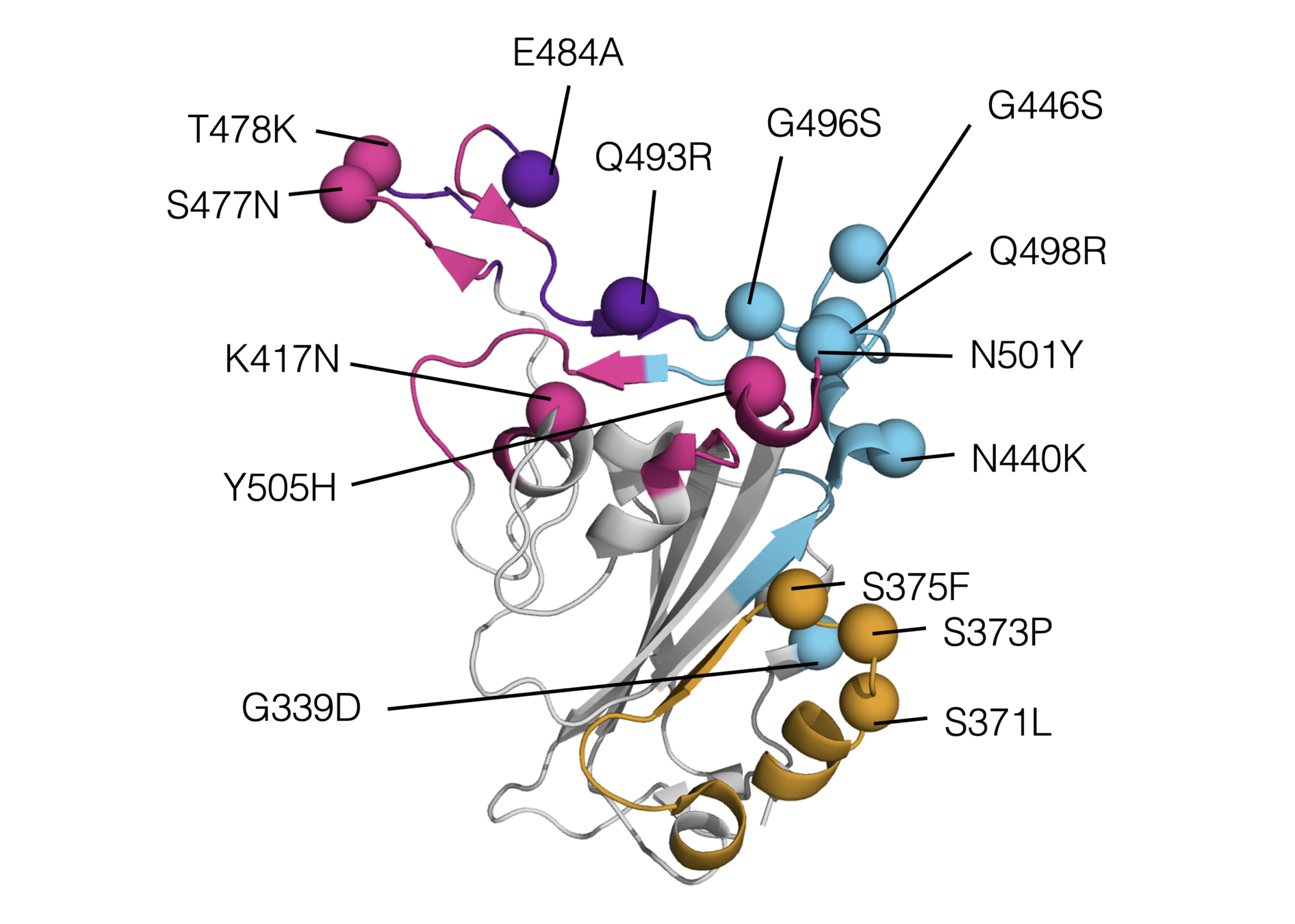
Omicron has a lot of mutations, especially in the receptor binding domain (RBD)
How does Omicron bind ACE2 with so many mutations?
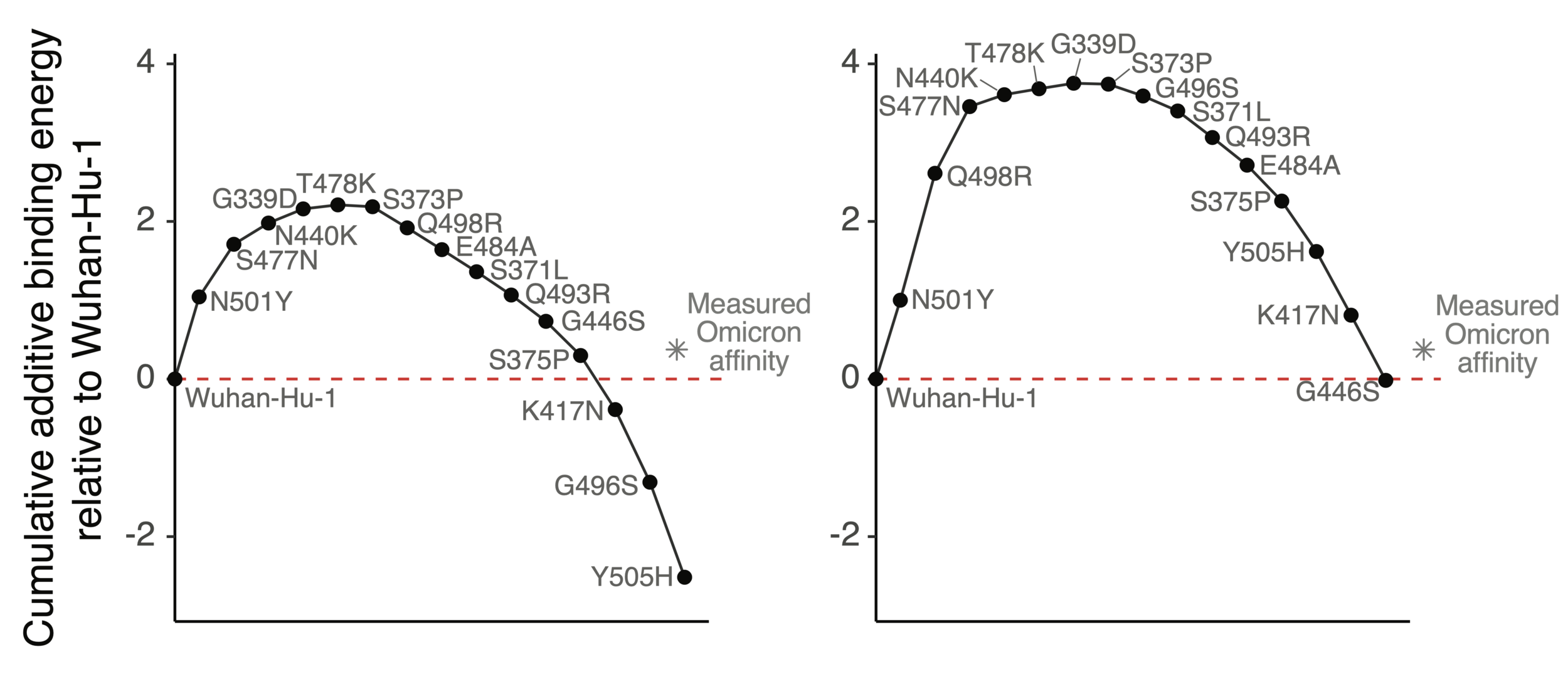
Mutations in Omicron have net negative effect on ACE2 binding if summed as single mutants
How does Omicron bind ACE2 with so many mutations?


Mutations in Omicron have net negative effect on ACE2 binding if summed as single mutants
However, two mutations (Q498R & N501Y) work together so net effect ~zero when both present
ACE2 affinity-enhancing mutations buffer antibody-escape mutations
RBD will not run out of evolutionary space
25 of 31 residues in CoV-229E RBD that contact receptor varied during virus's evolution in humans over last ~50 years (Li et al, eLife, 2019)
There are lots of mutations to SARS-CoV-2 RBD that retain (and sometimes even enhance) ACE2 affinity (Starr et al, 2020)
Outline
- Principles of viral antigenic evolution and emergence of Omicron
- Importance of the RBD
- Antibody escape in Omicron
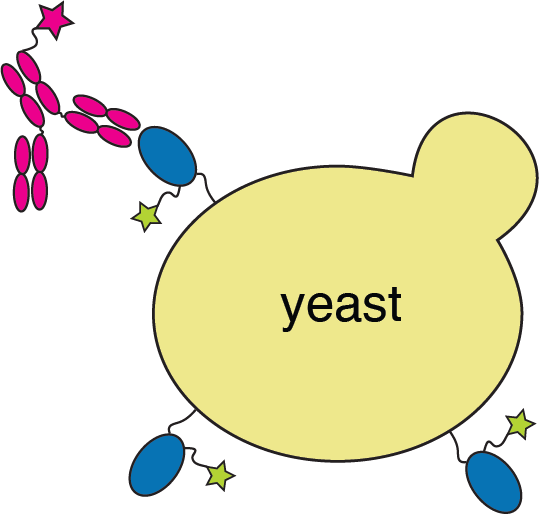
We map escape mutations by sorting for RBD variants that don't bind antibody
RBD
fluorescently labeled antibody
yeast
fluorescent tag on RBD
Escape map from a single antibody
Escape maps from lots of antibodies
Infection / vaccination elicit polyclonal antibodies
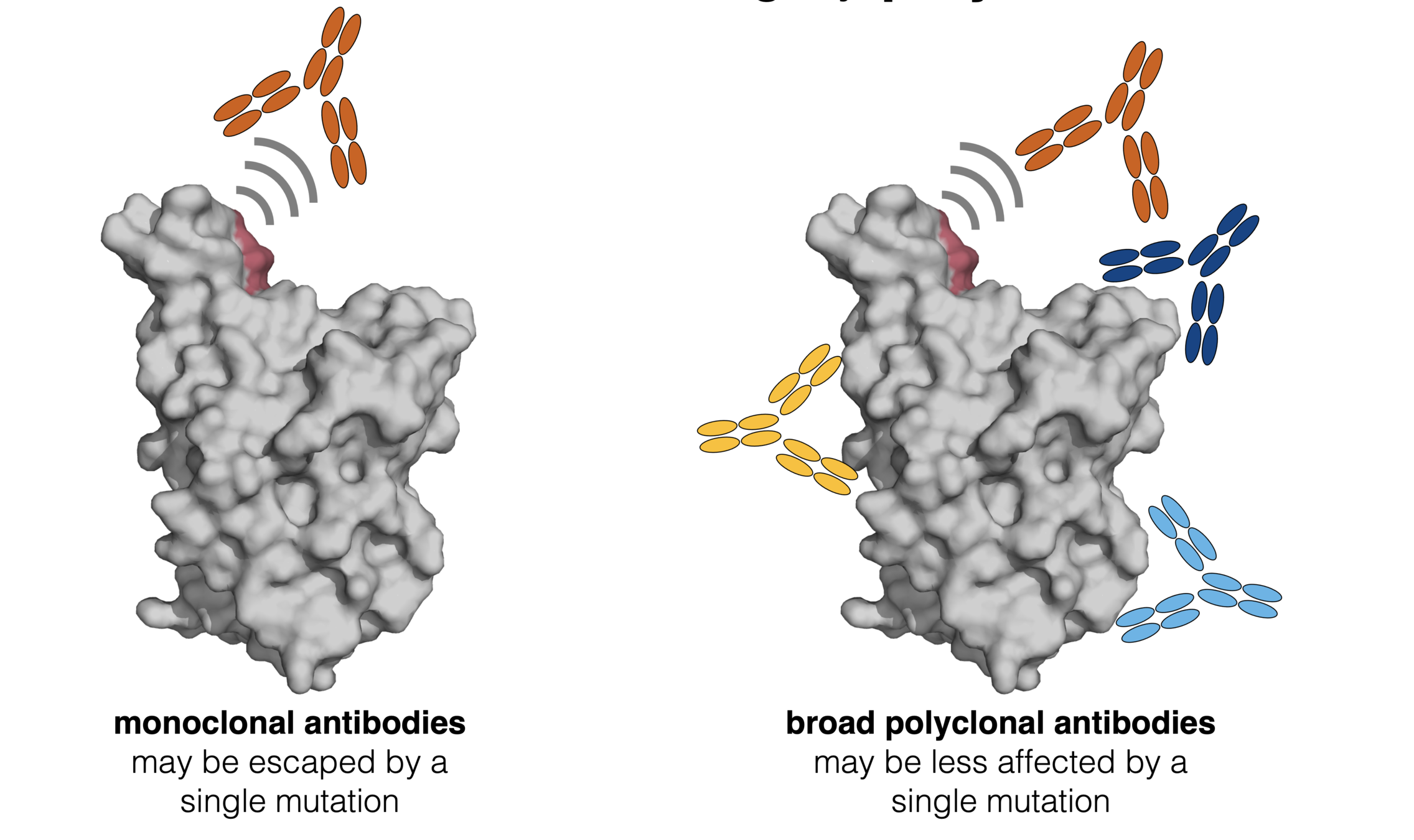
How do mutations affect polyclonal antibodies? First, consider an equal mix of three monoclonal antibodies.

Interactive version of this mini example is at https://jbloomlab.github.io/SARS2_RBD_Ab_escape_maps/mini-example-escape-calc/
LY-CoV555 is escaped at both sites 484 and 490, so mutating either site has same overall effect
Average escape across all antibodies

Mutating site 484 or 490 eliminates neutralization by antibody LY-CoV555, as reflected in thick black line showing average
Interactive version of this mini example is at https://jbloomlab.github.io/SARS2_RBD_Ab_escape_maps/mini-example-escape-calc/
Antibody-escape calculator extends this principle to deep mutational scanning data for ~1,500 different human antibodies
Escape calculator is described in Greaney et al (2022), and is available at https://jbloomlab.github.io/SARS2_RBD_Ab_escape_maps/escape-calc/
36 antibodies mapped by Tyler Starr & Allie Greaney in Bloom lab, from early SARS-CoV-2 strains
Antibody-escape calculator extends this principle to deep mutational scanning data for ~1,500 different human antibodies
Escape calculator is described in Greaney et al (2022), and is available at https://jbloomlab.github.io/SARS2_RBD_Ab_escape_maps/escape-calc/
36 antibodies mapped by Tyler Starr & Allie Greaney in Bloom lab, from early SARS-CoV-2 strains
1,522 (!) antibodies mapped by Sunney Xie, Richard Cao, Fanchong Jian, et al at Peking University. From early strains, BA.1, & patients with prior SARS-CoV-1 infection. See here.
What does escape calculator using these data tell us about evolution that has already happened?
Antibodies elicited by early SARS-CoV-2 that neutralize Wuhan-Hu-1 heavily focus on sites like 484, 417, 446

417
446
484
417
446
484
490 not mutated, but antibodies that bind there are escaped by mutation at 484 which is in same epitope.

Note Omicron has additional escape not modeled here due to mutations that put RBD more in down conformation and cause N343 glycan displacement: Cao et al (2022), Gobeil et al (2022), and explanation here.
Omicron BA.1 is mutated at many of these sites, which is why it is neutralized substantially less well by current vaccines
Omicron BA.2 has some different RBD mutations than BA.1, but similar overall escape from antibodies from early SARS-CoV-2

Note Omicron has additional escape not modeled here due to mutations that put RBD more in down conformation and cause N343 glycan displacement: Cao et al (2022), Gobeil et al (2022), and explanation here.
Can identify mutations that mediate further escape. In Dec 2021 we predicted 486 as likely site of future evolution--in April 2022, mutation F486V was identified in Omicron BA.4 and BA.5.

486 is largest site of escape for antibodies not already escaped by mutations in BA.2
What are likely next steps in antigenic evolution of SARS-CoV-2?

Sites of escape from antibodies elicited by early (pre-Omicron) infection/vaccination that still neutralize Omicron BA.2
346
356
444-446
452
450
462
468
486
499
mutated in BA.4/BA.5 or BA.2.12.1
Sites of escape are somewhat different for antibodies from people who had an Omicron BA.1 breakthrough infection

346-348
444-446
452
486
468
mutated in BA.4/BA.5 or BA.2.12.1
356
490
The differences in antibody-escape mutations between people who have / have-not had BA.1 breakthrough infections is consistent with prior studies on other viruses showing that serum antibodies from people with different exposure histories have different viral escape mutations.
Averaging across early SARS-CoV-2 & BA.1 breakthrough exposures, sites of escape in BA.2. Watch these sites!
346-348
444-446
452
486
468
mutated in BA.4/BA.5 or BA.2.12.1
356

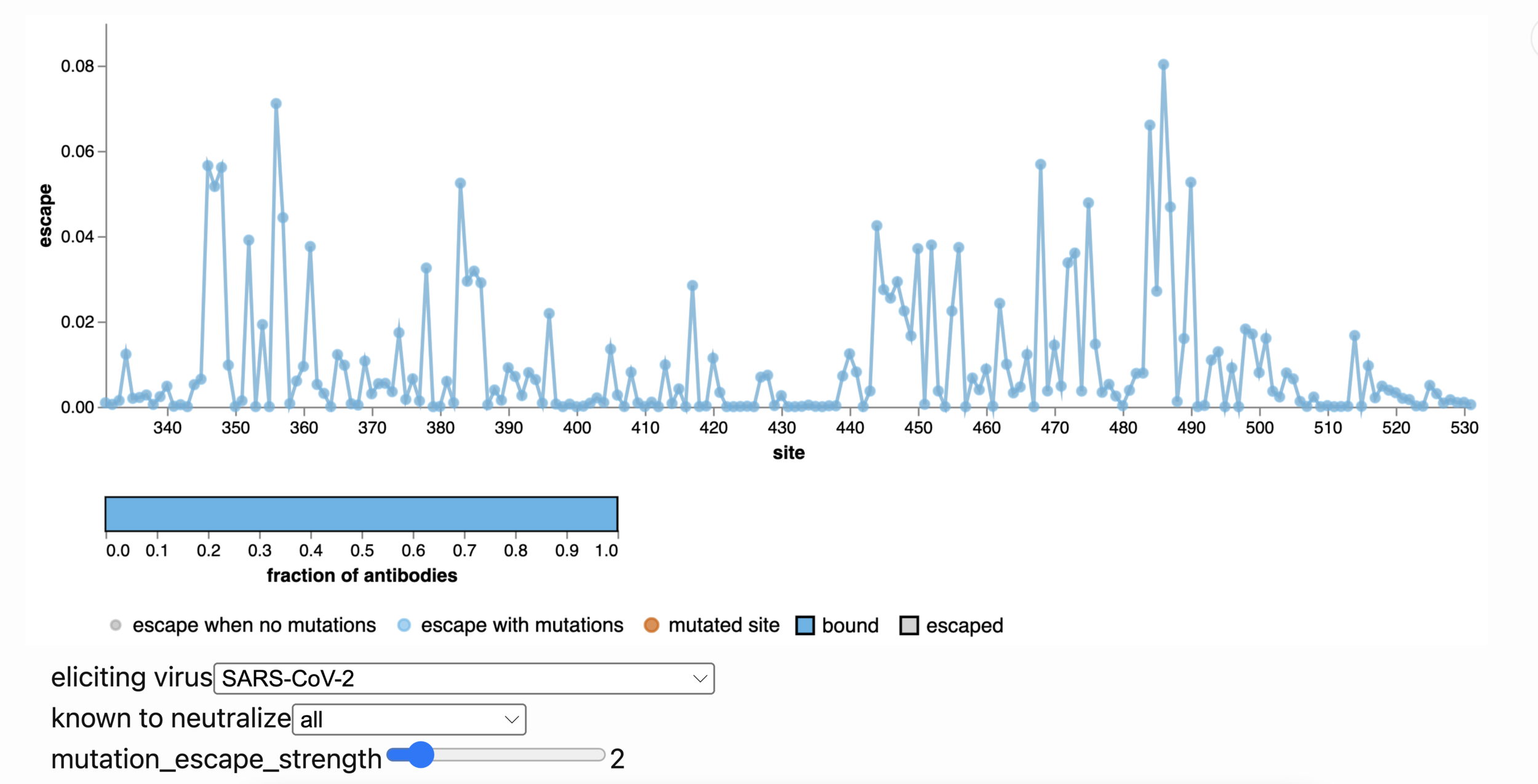


Explore yourself with online escape calculator: select antibodies by eliciting virus or strains they neutralize, click sites to mutate
https://jbloomlab.github.io/SARS2_RBD_Ab_escape_maps/escape-calc/
Crowe lab (Vanderbilt)
Chu lab (Univ Wash)
Veesler lab (Univ Wash)
King lab (Univ Wash)
Li lab (Brigham & Women's)
Boeckh lab (Fred Hutch)
Alex Greninger (Univ Wash)
Nussenzweig lab (Rockefeller)
Bjorkman lab (Caltech)






Tyler Starr
Allie Greaney
Rachel Eguia
Bloom lab (Fred Hutch)


Sarah Hilton
Kate Crawford


Andrea Loes