Metastasis-directed radiotherapy without systemic therapy for oligometastatic clear-cell renal-cell carcinoma
Primary efficacy analysis of a single-arm, single-centre, phase 2 trial
Tang C, Sherry AD, Seo A, et al.
Lancet Oncology 2025; 26: 1289–99
Study design
- Single-arm, single-centre, phase 2 trial (MD Anderson Cancer Center)
- Enrolled July 2018 – May 2023; 121 patients
- Co-primary endpoints:
- Progression-free survival (RECIST 1.1) — per-protocol population
- Systemic therapy-free survival (STFS) — intention-to-treat population
- Prespecified threshold for success: median STFS ≥ 24 months
- Benchmark: Rini et al. surveillance study — median time to systemic therapy 14.9 months
- Sample size: 100 patients (84% power); increased to 120 for censoring
Eligibility criteria
- Inclusion:
- Age ≥ 18 years
- Histologically confirmed renal-cell carcinoma
- 1–5 sites of metastatic disease
- ECOG performance status 0–2
- Candidate for definitive radiotherapy to all disease sites
- Adequate organ function (ANC ≥ 1000, Plt ≥ 50k, Hgb ≥ 9, Tbili ≤ 1.5, ALT/AST ≤ 3× ULN)
- Off systemic therapy ≥ 1 month or systemic therapy-naïve
- Exclusion:
- Pregnancy
- Comorbidities contraindicating RT (e.g. active scleroderma)
- Diffuse metastatic processes (leptomeningeal, diffuse bone marrow, peritoneal carcinomatosis)
- RECIST 1.1 measurable disease was not required
Trial schema
Enrollment: 1–5 metastatic lesions, ccRCC, ECOG 0–2
Staging: CT chest + MRI abd/pelvis, CT CAP, or PET-CT
↓
MDT to all sites of disease (SBRT preferred: ≤ 5 fx, ≥ 7 Gy/fx)
Surgery or IR ablation permitted if ≥ 1 site treated with RT
↓
Surveillance off systemic therapy
Imaging q12 weeks × 1 year, then q18 weeks
↓
Disease progression?
↓
Oligoprogression (≤ 3 new/progressing sites)
↓
Additional round of MDT
Reset surveillance schedule
Polymetastatic progression (> 3 sites)
↓
Initiate systemic therapy
Also if: RT toxicity precludes MDT, local progression, or physician/patient choice
Patient characteristics
- 121 patients enrolled (ITT); 120 received RT (per-protocol)
- Median age: 66 years (IQR 61–72); 74% male
- 98% had prior nephrectomy; median time nephrectomy → enrollment: 71 months
- Median time metastasis diagnosis → enrollment: 10 months (IQR 2–29)
- ECOG PS: 0 (68%), 1 (29%), 2 (3%)
- IMDC risk: Favorable 52%, Intermediate 47%, Poor 1%
- Previous systemic therapy: 30% (most common: pazopanib n=11, nivo/ipi n=10)
- Prior local therapy: Metastasectomy 32%, RT 16%, IR ablation 8%
- Lesions treated (round 1): 1 lesion (59%), 2 lesions (31%), 3–5 lesions (10%)
- Most common met sites: Lung (n=76), lymph nodes (n=16)
- Median GTV of all irradiated sites: 5.7 cm³ (IQR 3.1–19.2)
Radiation and surveillance details
SBRT protocol
- ≤ 5 fractions at ≥ 7 Gy/fx to all disease sites
- Most common: 40–50 Gy / 4 fx (n=59)
- Alternative RT if location precluded SBRT
- Surgery/IR ablation if ≥ 1 site received RT
Treatment details
- Median GTV: 5.7 cm³ (IQR 3.1–19.2)
- Round 1: 1 lesion (59%), 2 (31%), 3–5 (10%)
- Most common sites: lung (n=76), LN (n=16)
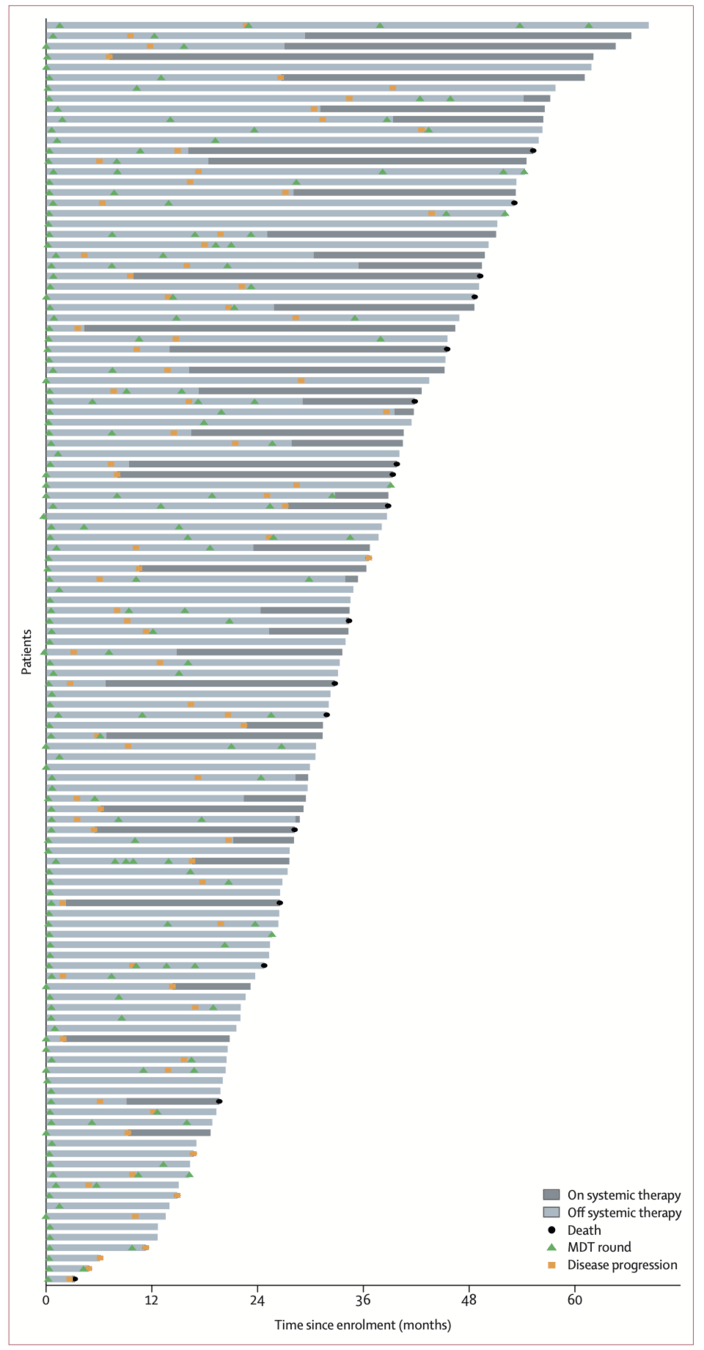
- 59% received ≥ 2 MDT rounds; up to 6 total
- Only 7% local progression in irradiated sites
Surveillance protocol
- Imaging q12 weeks × 1 year, then q18 weeks
- AE review + CBC with differential at each visit
- RECIST 1.1 with central radiology review
- Schedule resets after each MDT round
Oligoprogression → more MDT
- ≤ 3 new or progressing metastases
- Additional round of MDT, reset surveillance
Triggers for systemic therapy
- Polymetastatic progression (>3 sites)
- RT toxicity precluding further MDT
- Local progression after RT
- Physician or patient choice
51/121 (42%) ultimately started systemic Tx
Primary outcomes
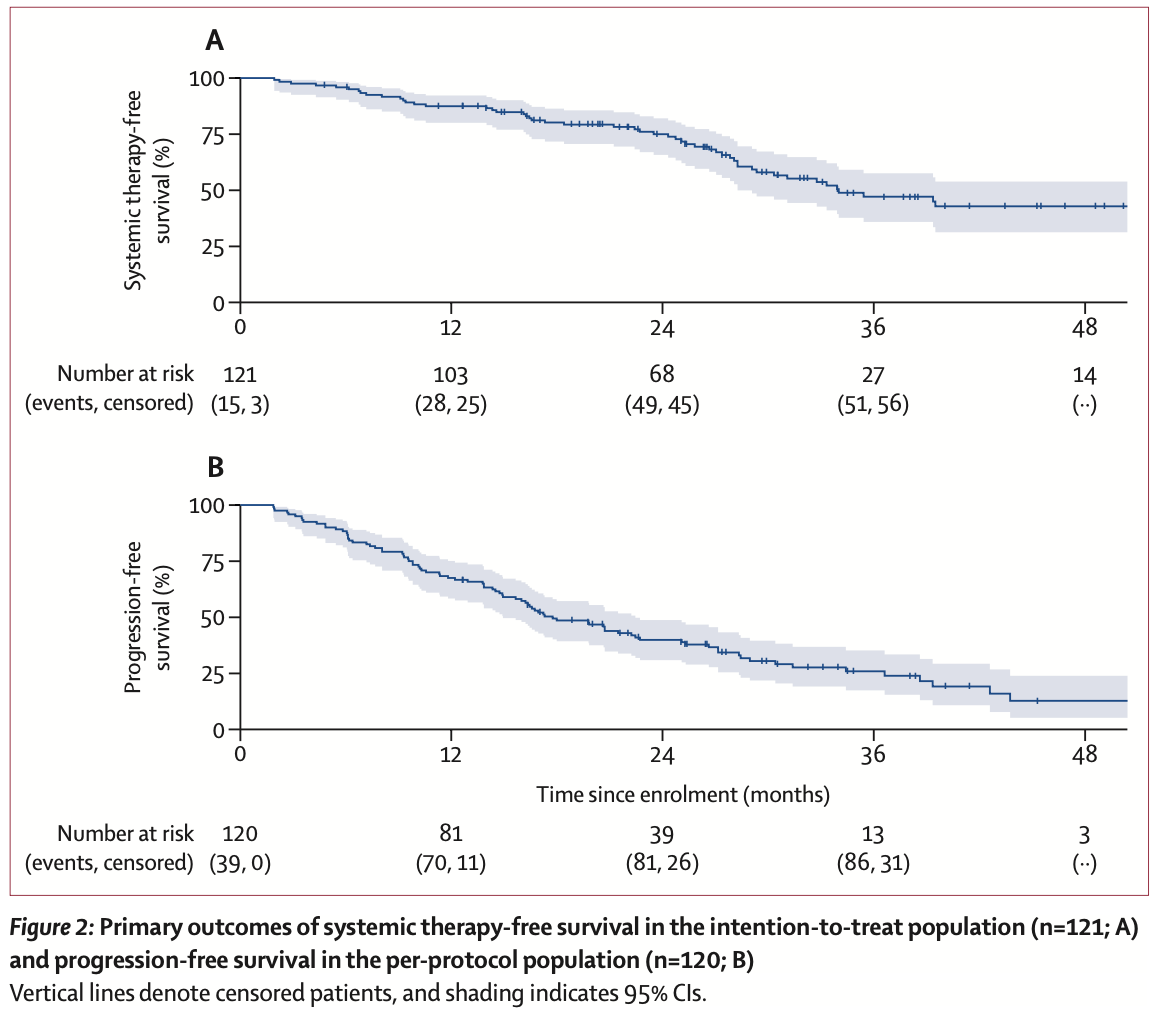
Median follow-up: 36.3 months (IQR 26.5–51.1)
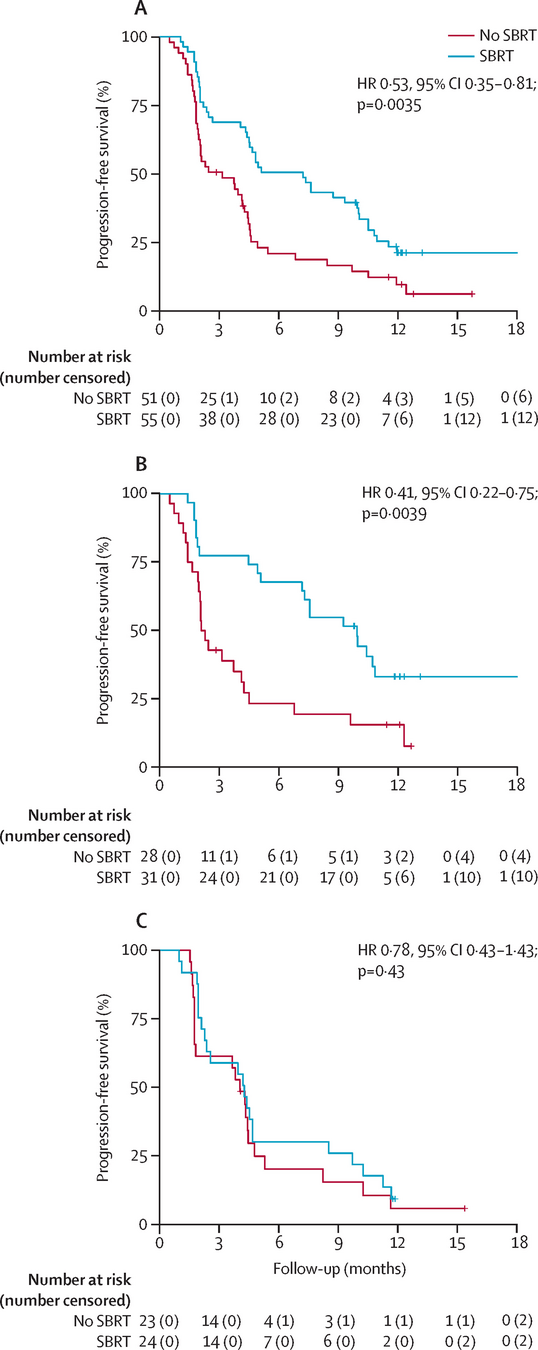
Systemic therapy-free survival (co-primary endpoint, intention-to-treat, n=121)
Time from enrollment to initiation of systemic therapy or death from ccRCC
| Median | 34.0 months (95% CI 28.3–54.1) |
| 1-year rate | 87.5% (95% CI 80.0–92.2) |
| Prespecified target | Median ≥ 24 months — exceeded ✓ (lower 95% CI also exceeded) |
| Events | 51 (42%) started systemic therapy; 2 (2%) died from disease |
| Benchmark | Rini et al. surveillance: median 14.9 months (95% CI 10.6–25.0) |
Progression-free survival (co-primary endpoint, per-protocol, n=120)
Time from enrollment to RECIST 1.1 progression, clinical progression, or death from any cause
| Median | 17.7 months (95% CI 14.9–22.4) |
| 1-year rate | 67.5% (95% CI 58.3–75.1) |
| Events | 86/120 (72%) — 73 RECIST progression, 7 clinical progression, 6 death |
| Comparators | Rini surveillance: 9.4 mo; RAPPORT (MDT + pembrolizumab): 15.6 mo |
Secondary outcomes and safety
Overall survival
| Median OS | Not reached |
| 1-year OS | 96.7% (95% CI 91.4–98.7) |
| 3-year OS (post hoc) | 86.5% (95% CI 77.5–92.1) |
| Deaths | 23 total: 14 disease, 9 other causes |
Patterns of progression
| Freedom from new lesions (12-mo) | 72.7% |
| Median time to new lesion | 22.7 months |
| First event: new lesions | 57% of patients |
| First event: local progression | Only 7% — high local control |
Toxicity (per-protocol, n=120)
| Grade ≥ 2 AEs | 25 patients (21%) |
| Grade 3–4 AEs | 8 patients (7%) |
| Treatment-related deaths | 0 (none) |
| Most common grade 3 | Pain (4 events), leukocytosis (2) |
| Grade 4 event | Hyperglycemia (RT to pancreas → insulin-dependent) |
| Grade 2 pneumonitis | 5 patients (4%) |
Key safety takeaway: 7% grade 3+ with no treatment-related deaths — compare to IO combination toxicity (grade 3+ in 46–73% across CheckMate 214, KEYNOTE-426, CLEAR) and metastasectomy complications (45.7% overall, Meyer J Urol 2017)
treatment course of all patients
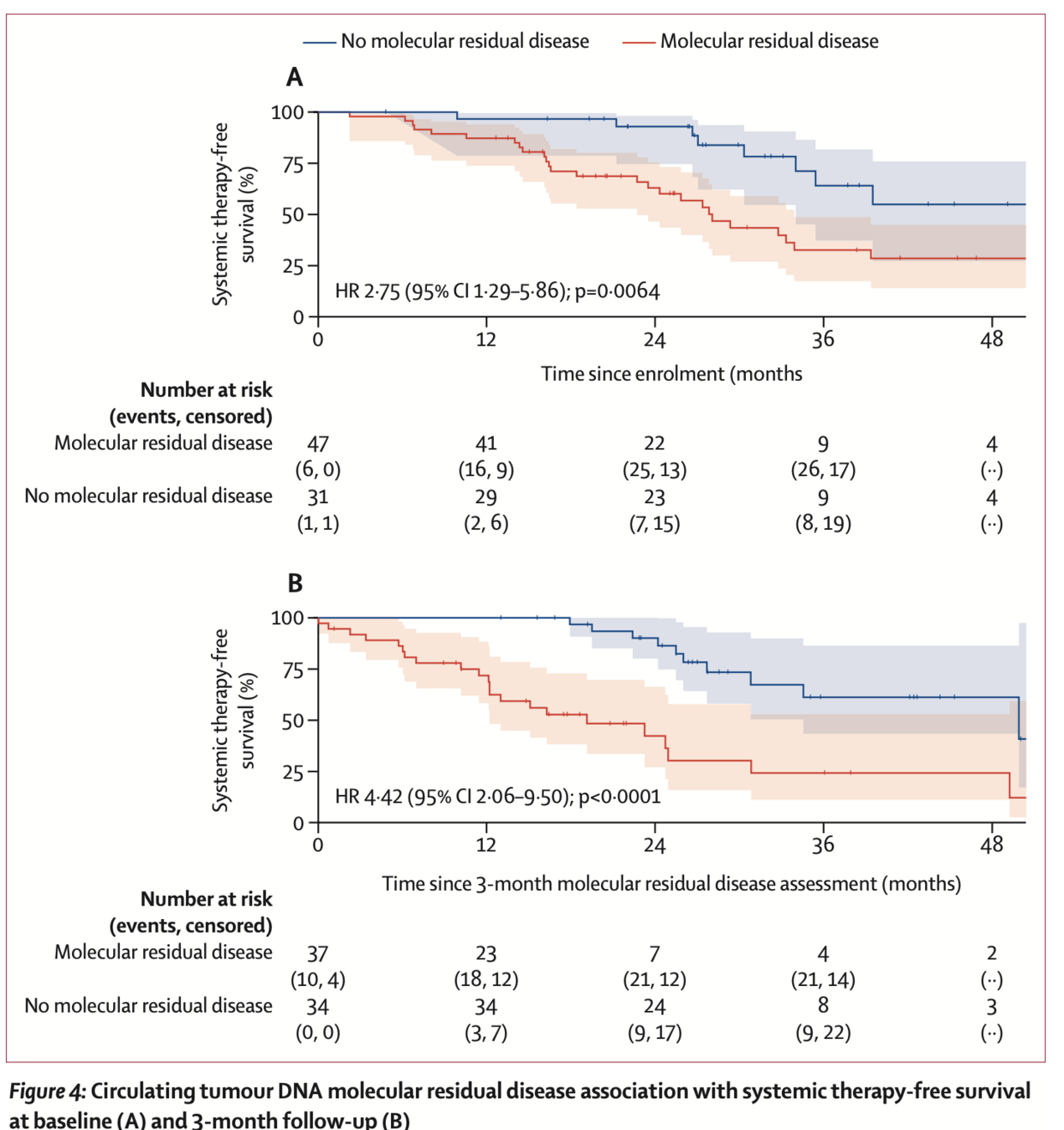
Exploratory: ctDNA molecular residual disease
- Assay: Tumor-informed, patient-specific ctDNA panels via WGS (up to 2000 somatic variants per patient; Precise MRD, Myriad Genetics)
- 87/89 patients had successful panel creation (98%); 149/168 plasma samples analyzed (89%)
- Baseline MRD: 47/78 patients (60%) were ctDNA MRD-positive
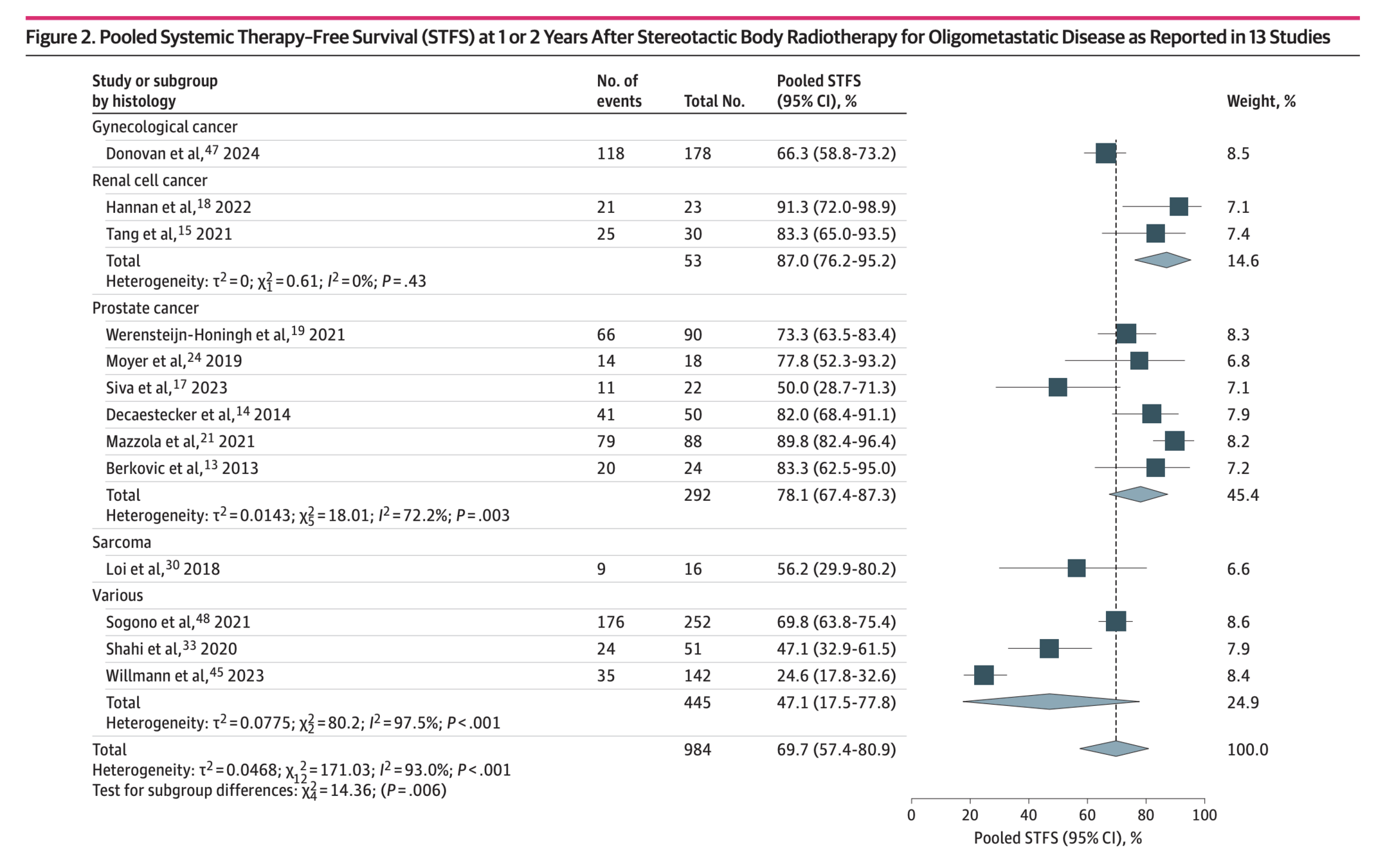
- MRD+ vs MRD−: median STFS 28.1 vs 54.1 months (HR 2.75, p=0.0064)
- 3-month MRD (landmark analysis): 37/71 patients (52%) MRD-positive
- MRD+ vs MRD−: median STFS 19.1 vs 49.9 months (HR 4.42, p<0.0001)
- 25% of baseline MRD+ patients converted to MRD− at 3 months
- Multivariable model: ctDNA MRD status remained independently prognostic after adjusting for # lesions and prior systemic therapy lines (all p<0.05)
ctDNA MRD and systemic therapy-free survival
A) Baseline
ctDNA MRD+ vs MRD− and STFS (HR 2.75, p=0.0064)
B) 3-month
ctDNA MRD+ vs MRD− and STFS (HR 4.42, p<0.0001)]
Prognostic factors for STFS
- Prespecified baseline clinical factors associated with STFS:
- Number of metastatic lesions at enrollment: HR 1.53 per lesion (95% CI 1.14–2.07, p=0.0044)
- Number of previous lines of systemic therapy: HR 1.50 per line (95% CI 1.11–2.04, p=0.0078)
- ctDNA MRD status independently prognostic on multivariable analysis (all p<0.05 with lesion count and prior systemic therapy)
- Factors associated with baseline MRD positivity (not adjusted for multiple testing):
- IMDC risk group (p=0.0034)
- Bone metastases (p=0.038)
- Total irradiated GTV (p=0.0045)
- Notably: factors associated with MRD status were not the same as those associated with STFS, suggesting independent prognostic value
Prostate cancer: the most mature MDT evidence — and model for RCC
Two key questions answered by randomized data: (1) Does MDT beat observation? (2) Should MDT be combined with systemic therapy?
Q1: MDT alone vs observation
| STOMP Ost, JCO 2018; 5-yr ASCO GU 2020 |
Median ADT-free survival: 21 vs 13 mo 5-yr ADT-free: 34% vs 8% (HR 0.57) 5-yr CRPC-free: 76% vs 53% |
| ORIOLE Phillips, JAMA Oncol 2020 |
6-mo progression: 19% vs 61% (p=0.005) PFS: not reached vs 5.8 mo (HR 0.30) |
| Pooled analysis Deek, JCO 2022 |
PFS: 11.9 vs 5.9 mo (HR 0.44, p<0.001) 15–20% durable PFS beyond 4–5 yrs High-risk mutations (ATM/BRCA/Rb1/TP53): HR 0.05 |
Q2: MDT + ADT vs ADT alone
| EXTEND Tang, JAMA Oncol 2023 |
PFS: NR vs 15.8 mo (HR 0.25, p<0.001) Eugonadal PFS: NR vs 6.1 mo (HR 0.32, p=0.03) MDT + intermittent ADT preserves testosterone |
| RADIOSA Marvaso, Lancet Oncol 2025 |
PFS: 32.2 vs 15.1 mo (HR 0.43, p=0.001) SBRT + 6-mo ADT vs SBRT alone First RCT showing ADT adds to SBRT |
| WOLVERINE X-MET, ASCO GU 2025 |
Pooled IPD from 5 RCTs (n=472) 48-mo OS: 87% vs 75% (HR 0.64, p=0.057) First signal of OS benefit with MDT |
The parallel to RCC: Both prostate and RCC have indolent biology in the oligometastatic subset. Both show SBRT can defer systemic therapy. Prostate has randomized data; RCC now has the Tang expansion cohort (n=121, STFS 34 mo). The next step for RCC is what prostate already has — randomized trials (SOAR, ASTROs).
Key difference: In prostate, the systemic therapy being deferred is ADT — with known QoL effects (sexual dysfunction, metabolic syndrome, osteoporosis). In RCC, the deferred therapy is IO combinations — with different but significant toxicity.
Conclusions
- Serial MDT without systemic therapy in oligometastatic ccRCC achieved a median STFS of 34.0 months, exceeding the 24-month prespecified threshold
- Favorable PFS (median 17.7 months)
- OS (median not reached; 3-year OS 86.5%)
- Compared favorably to surveillance alone (Rini: PFS 9.4 mo) and MDT + pembrolizumab (RAPPORT: PFS 15.6 mo, OS 20 mo)
- Modest toxicity profile: grade ≥ 3 in only 7%; no treatment-related deaths
- 59% of patients received ≥ 2 rounds of MDT, demonstrating feasibility of serial approach
- ctDNA MRD status at baseline and 3 months was an independent prognostic biomarker for STFS
- Number of metastatic lesions and prior systemic therapy lines also prognostic
- MDT can enable meaningful treatment de-escalation, potentially improving QoL and reducing costs
Strengths and limitations
- Strengths:
- Largest prospective trial of serial MDT without systemic therapy for oligometastatic ccRCC (n=121)
- Longest follow-up in this setting (median 36.3 months)
- Central radiology review (RECIST 1.1) by subspecialized radiologist
- Novel integration of second-generation ultrasensitive ctDNA assay
- 60% baseline ctDNA detection rate (vs 25% with first-generation assays)
- Prespecified endpoints, futility analyses, and exploratory biomarker analyses
- Limitations:
- Single-arm, single-institution — no randomized comparator
- No quality-of-life data collected
- Systemic therapy initiation partly physician/patient discretion
- RECIST 1.1 developed for systemic therapy assessment — may not optimally capture oligomet MDT response
- ctDNA analysis exploratory and in a subset only; tissue procurement biases
- Predominantly favorable/intermediate IMDC risk and predominantly lung metastases — limits generalizability
Discussion points
- How should radiation oncologists select patients for MDT-only strategies? Should ctDNA MRD testing be integrated into routine practice, and at what time points?
- What are the implications for non-clear-cell histologies, which have fewer effective systemic therapy options? Could MDT have an even greater role?
- In what other histologies can metastasis directed therapy be used to delay systemic therapy vs used to maintain efficacy of systemic therapy?