Health-related Quality of Life and Pain Outcomes
177-Lu-PSMA in Metastatic Castration-Resistant Prostate Cancer
VISION Trial: Phase 3 Randomized Study
Fizazi K, et al. Lancet Oncol 2023
Background
- mCRPC remains an incurable disease despite therapeutic advances
- Patients have high risk of impaired health-related quality of life (HRQOL) and increased mortality
- Up to 90% have bone metastases which significantly impact HRQOL
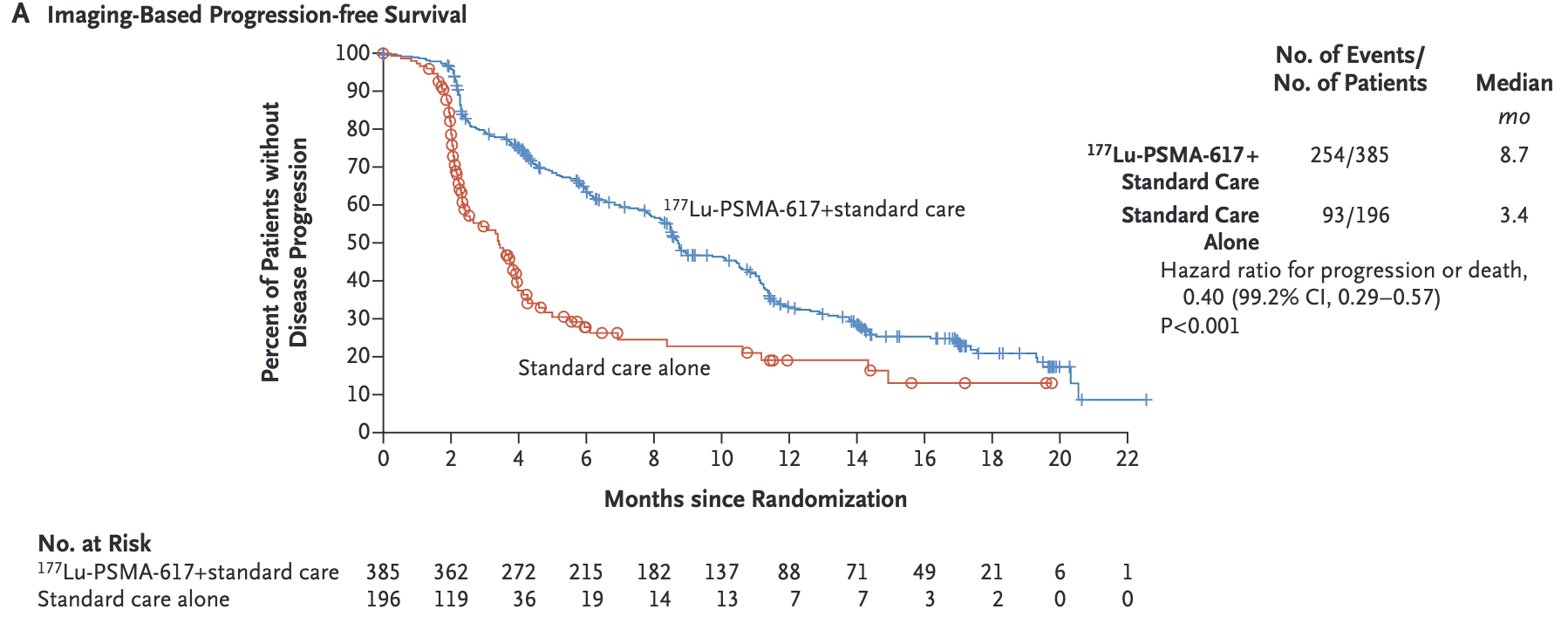
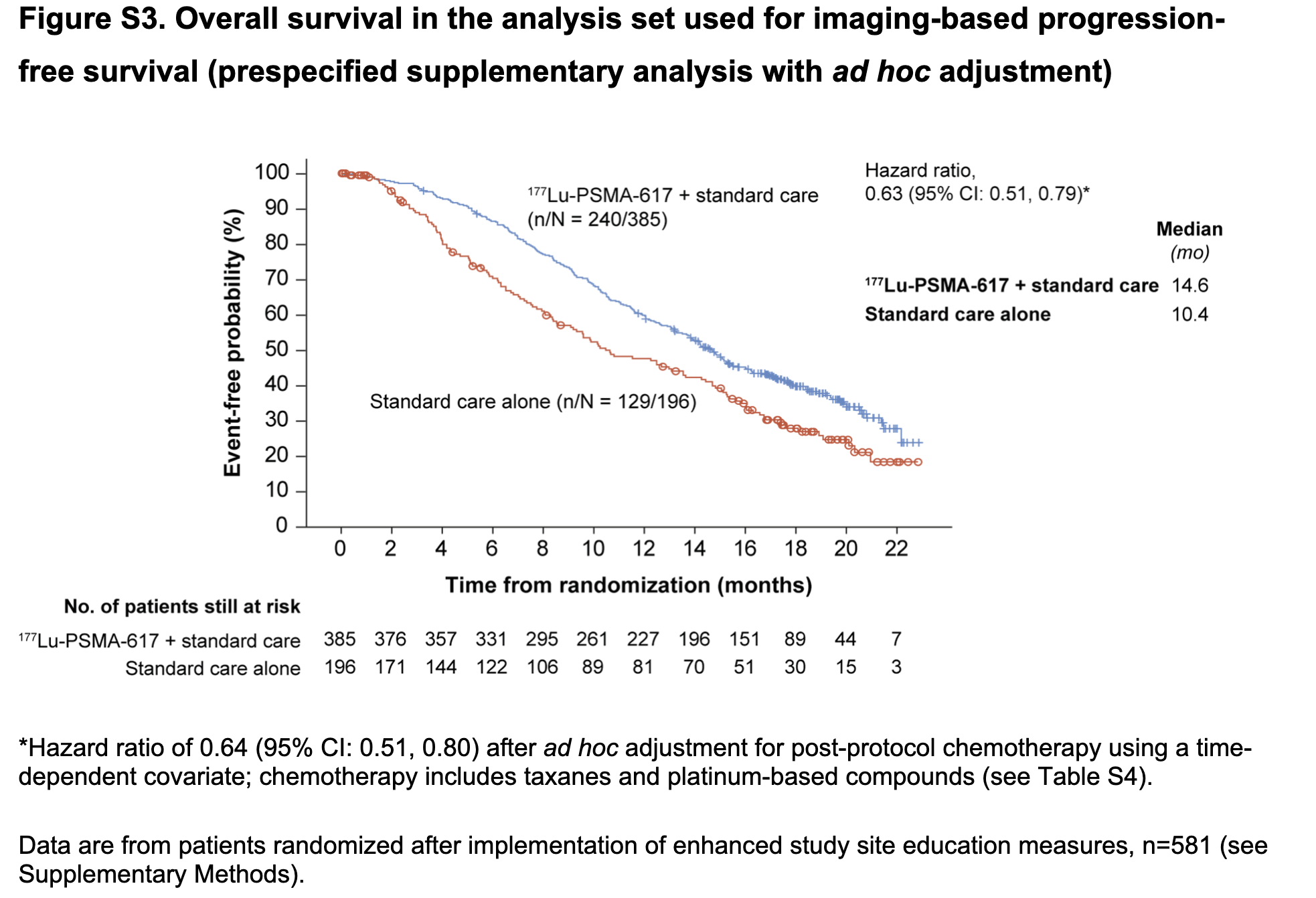
- [177Lu]Lu-PSMA-617 previously showed improved rPFS and OS in VISION trial
- This analysis examines impact on HRQOL, pain and symptomatic skeletal events
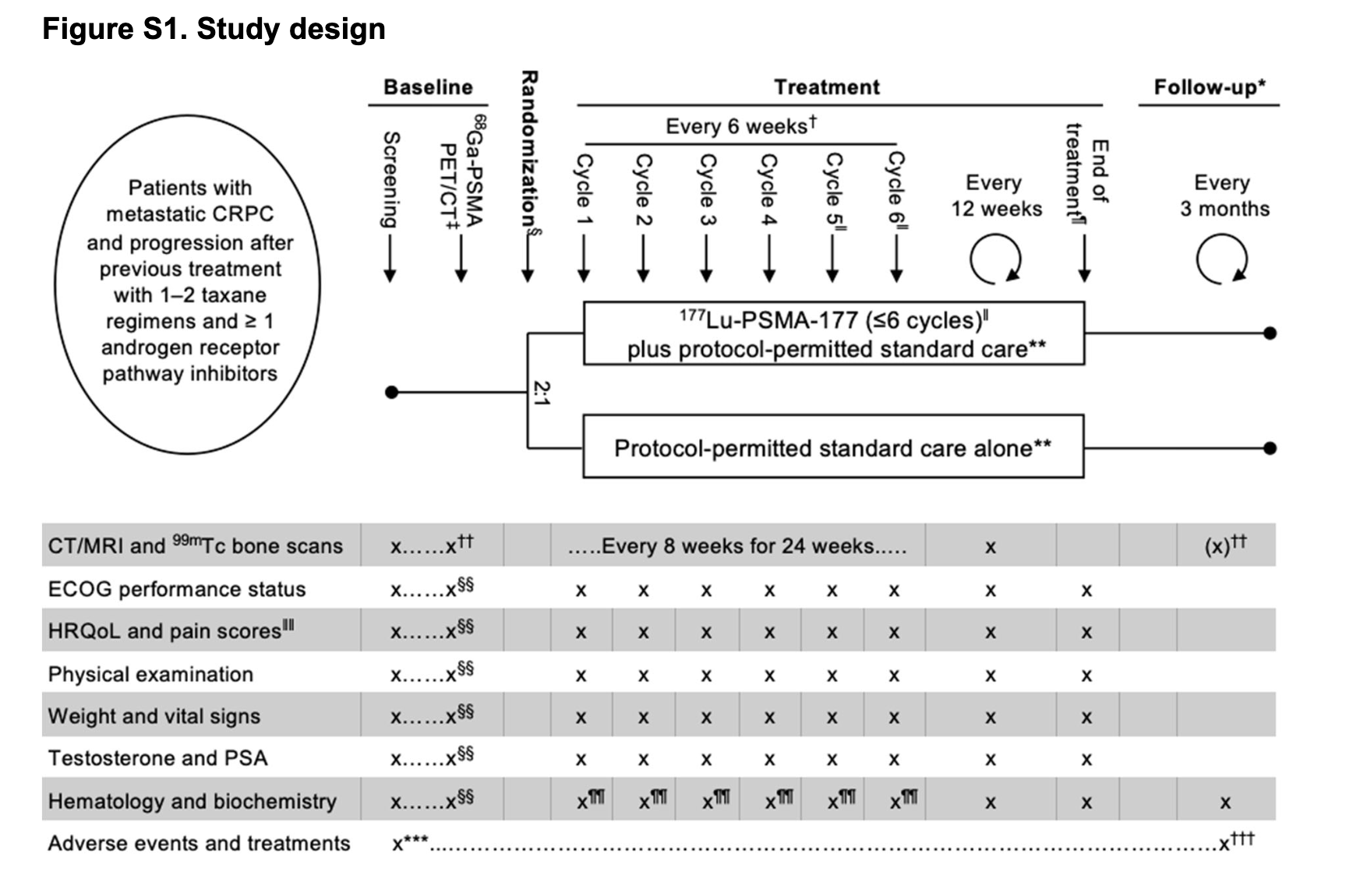
- Phase 3, open-label, randomized trial (2:1)
- 84 Centers
- N=581 enrolled after March 2019:
- 385 [177Lu]Lu-PSMA-617 + standard of care
- 196 standard of care alone
- Key Eligibility:
- Progressive PSMA+ mCRPC
- Prior AR pathway inhibitor and 1-2 taxanes
- ECOG PS 0-2
- PSMA-positive tumors on Ga-68 PSMA-11 PET/CT
- No eligibility for additional chemotherapy
- Life expectancy ≥6 months
- Treatment:
- 177Lu-PSMA-617: 7.4 GBq IV q6wk x 4-6 cycles
- Standard of care: hormonal therapy, bone-targeted agents, supportive care
VISION Trial Design
Screening
Assess eligibility, 68Ga-PSMA-11 PET/CT scan
↓
PSMA-positive cancer?
No
↓
Screen Failure
Yes
↓
Randomization (2:1)
Investigational Arm
177Lu-PSMA-617 + Best Supportive/Standard Care
7.4 GBq every 6 weeks, max 6 cycles
Control Arm
Best Supportive/Standard Care Alone
↓
End of Treatment Visit
30 days after last treatment
↓
Long-term Follow-up
Every 3 months for 24 months
Patient Population
Primary Outcomes
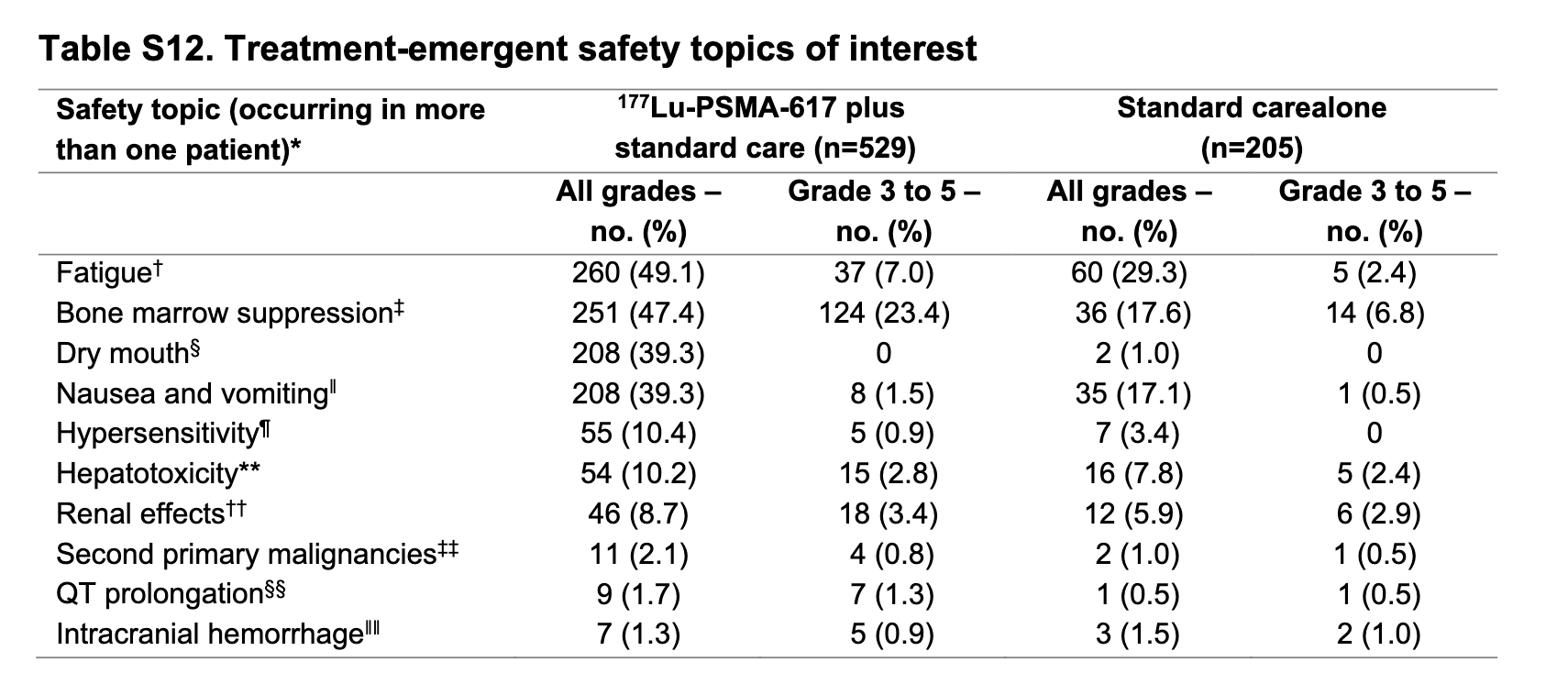
Safety
- Higher grade 3-4 hematologic adverse events with Lu-PSMA-617:
- Anemia: 15% vs 6%
- Thrombocytopenia: 9% vs 2%
- Lymphopenia: 51% vs 19%
- 5 treatment-related deaths in Lu-PSMA-617 arm:
- Pancytopenia (n=2)
- Bone marrow failure (n=1)
- Subdural hematoma (n=1)
- Intracranial hemorrhage (n=1)
- Renal function remained stable
Safety
Safety
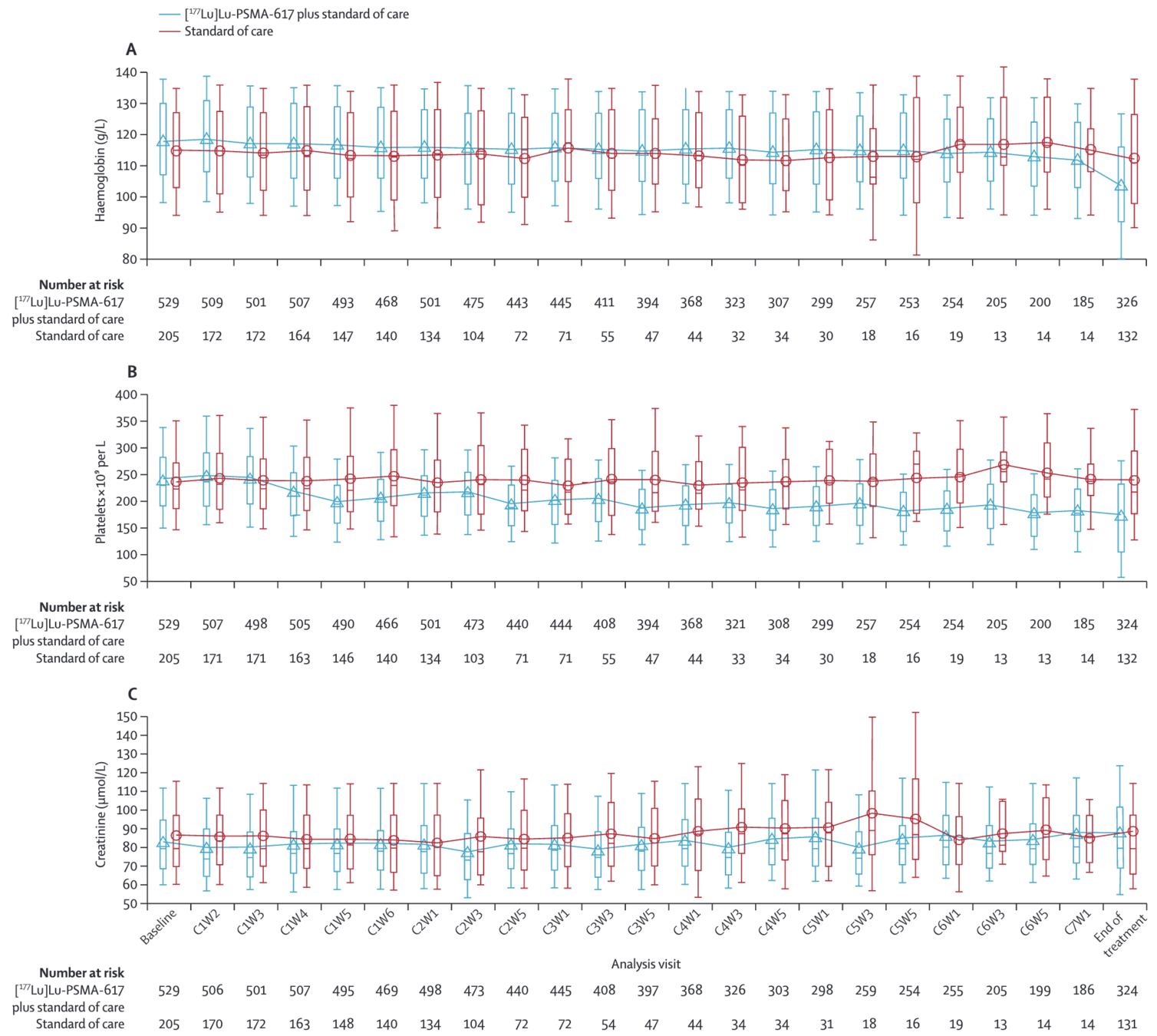
Hemaglobin
Platelets
Creatinine
Symptomatic Skeletal Events
- Median time to first SSE or death:
- Lu-PSMA: 11.5 months
- Control: 6.8 months
- HR 0.50 (95% CI 0.40-0.62)
- SSE events:
- Lu-PSMA: 60/385 (16%)
- Control: 34/196 (17%)
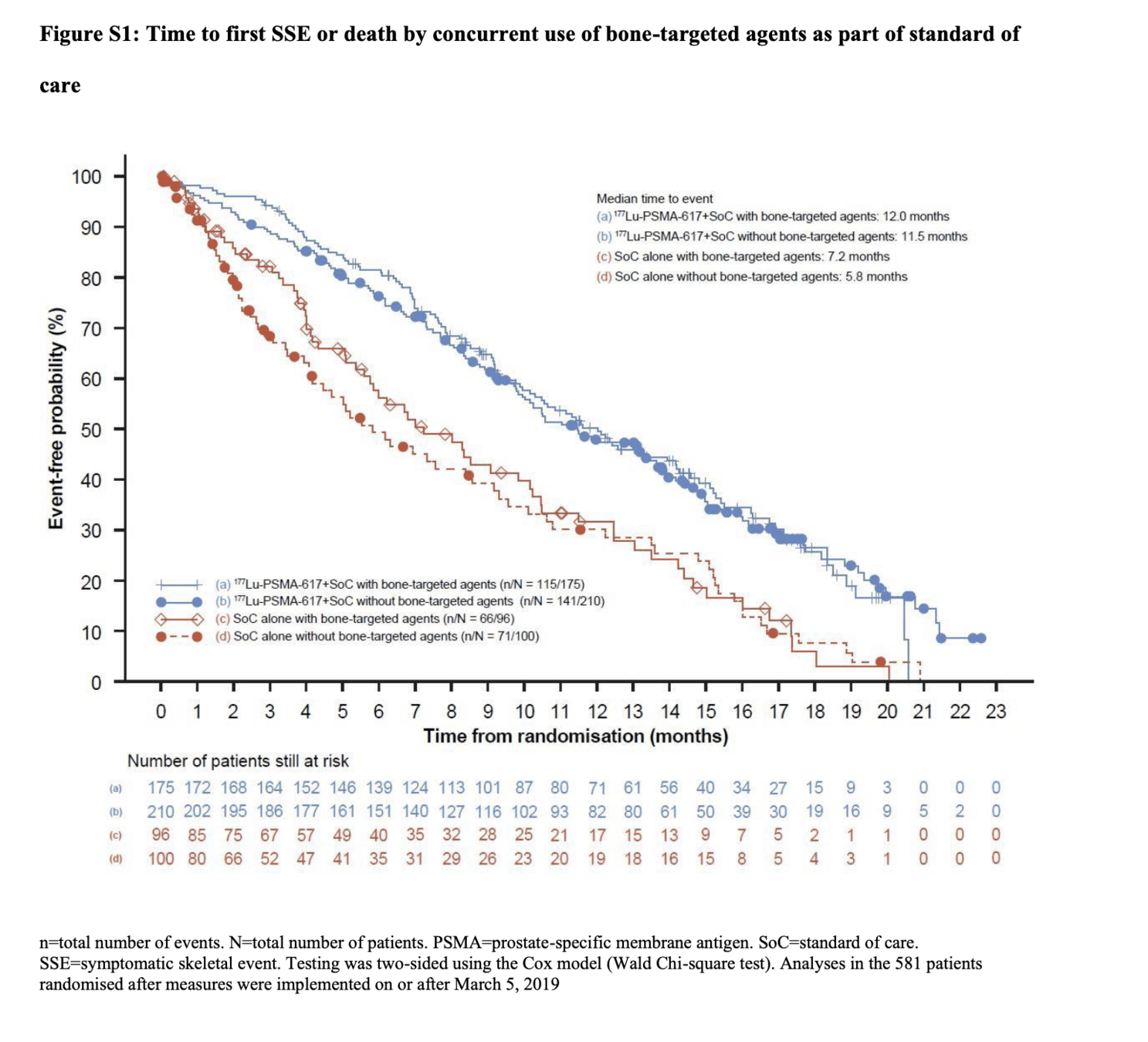
- Benefit seen regardless of bone-targeted therapy use
- Most common SSE: radiation for bone pain
Symptomatic Skeletal Events
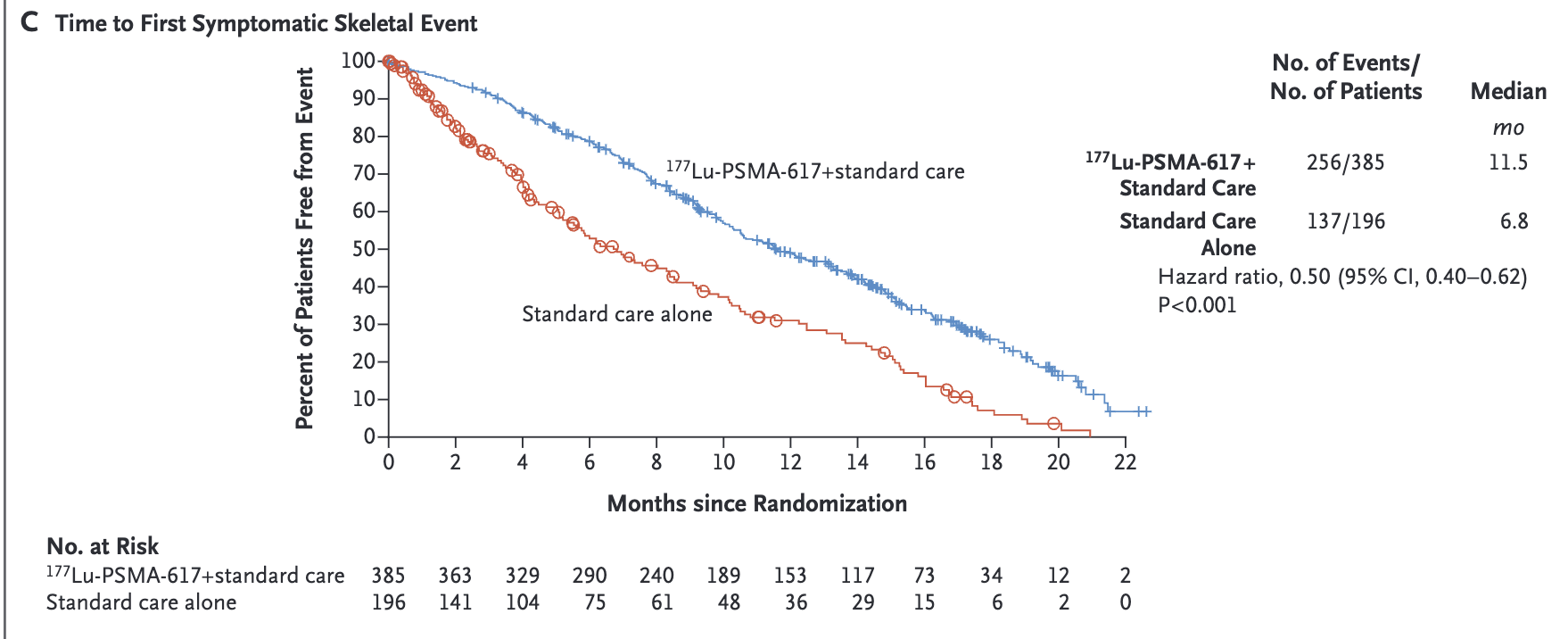
Symptomatic Skeletal Events
Quality of life Endpoints
- Patient-Reported Outcomes (PROs):
- FACT-P (Functional Assessment of Cancer Therapy-Prostate)
- EQ-5D-5L utility score
- Brief Pain Inventory-Short Form (BPI-SF)
- Time to first symptomatic skeletal event
- PROs assessed at:
- Baseline
- Day 1 of each cycle
- End of treatment
- Safety/adverse events also evaluated

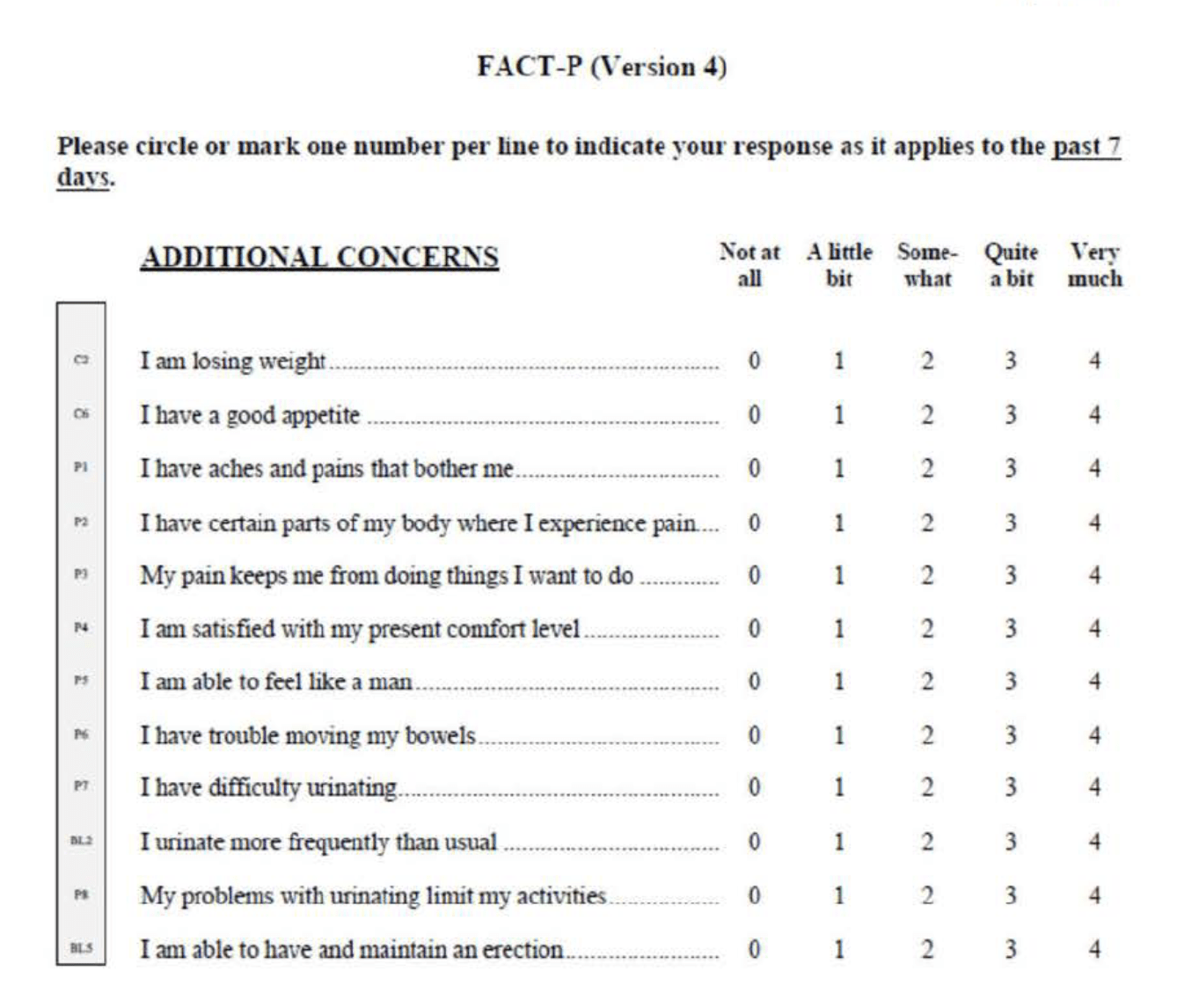
Endpoints
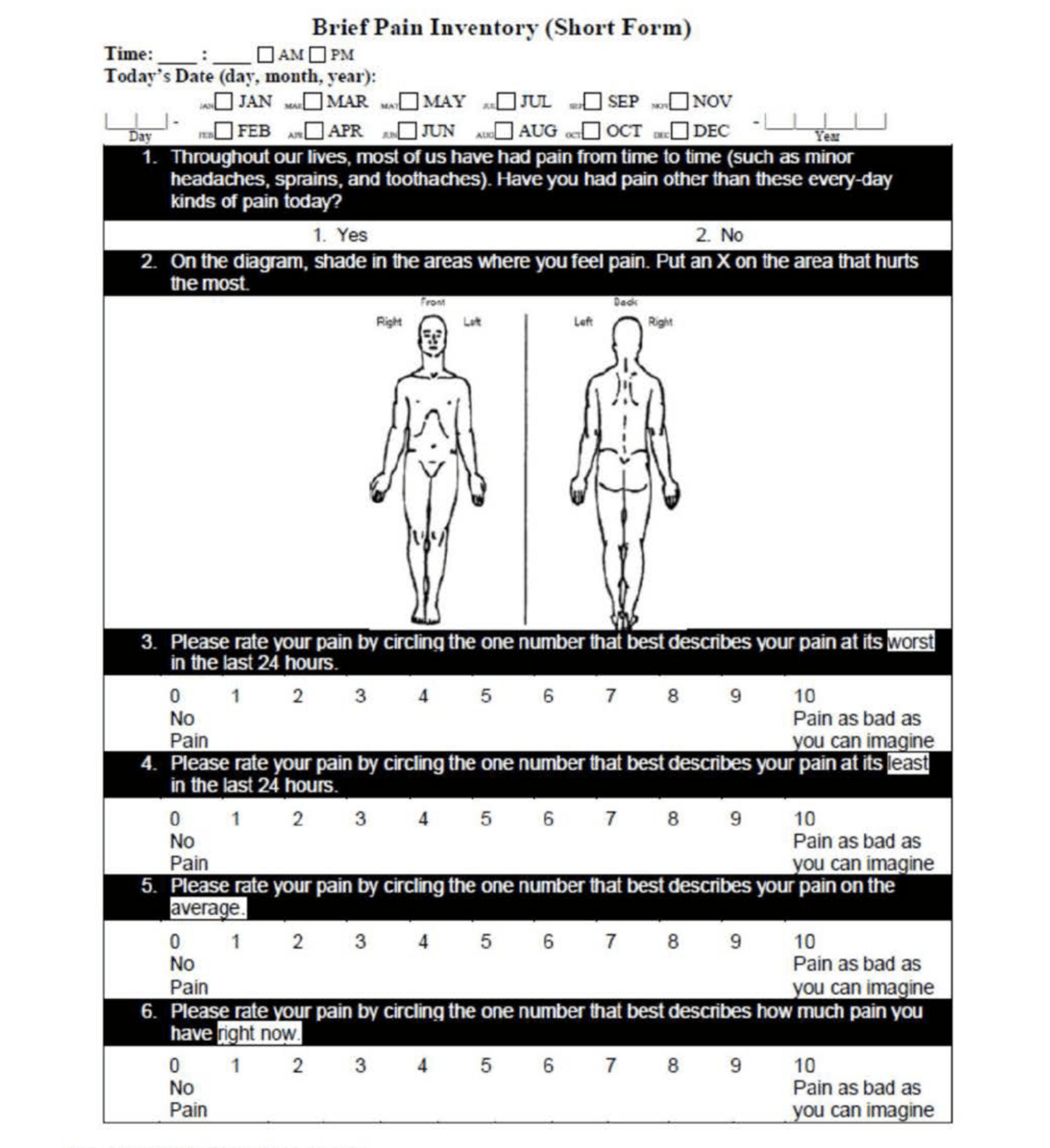


EQ-5D-5L (European Quality of Life (EuroQol) – 5 Domain 5Level scale)
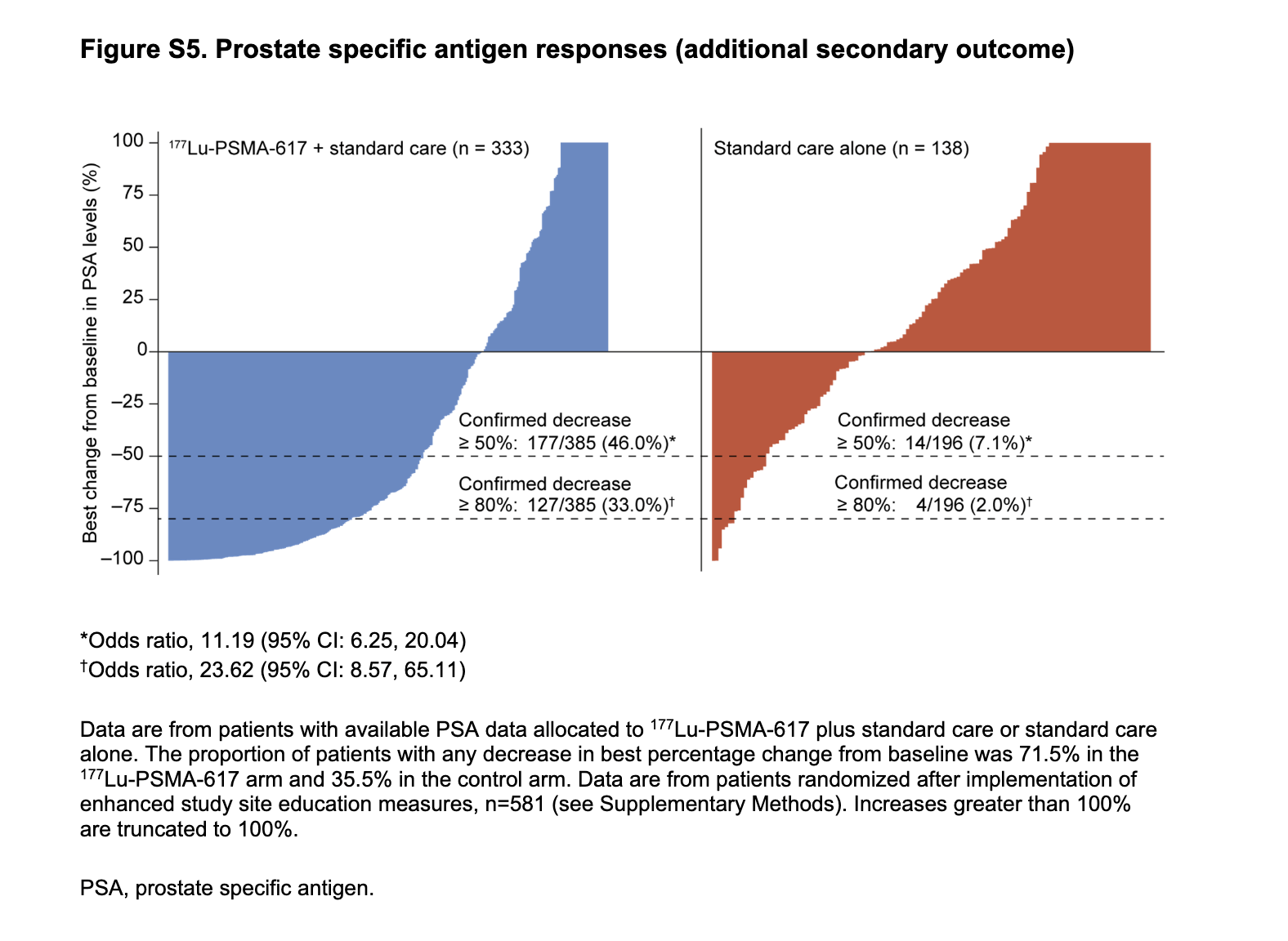
Key Results - HRQOL
- Delayed time to worsening with Lu-PSMA-617 vs SOC for:
- FACT-P total score
- Lu-PSMA: 5.7 vs Control: 2.2 (HR 0.54; 95% CI 0.45-0.66)
- BPI-SF pain intensity
- 6.9 vs Control: 2.6 (HR 0.52; 95% CI 0.42-0.63)
- EQ-5D-5L utility score
- 1.0 vs Control: 0.5 (HR 0.65; 95% CI 0.54-0.78)
- FACT-P total score
- Benefits seen across all FACT-P domains
- Similar results in prespecified and post-hoc analyses
- Consistent effects with/without including disease progression/death
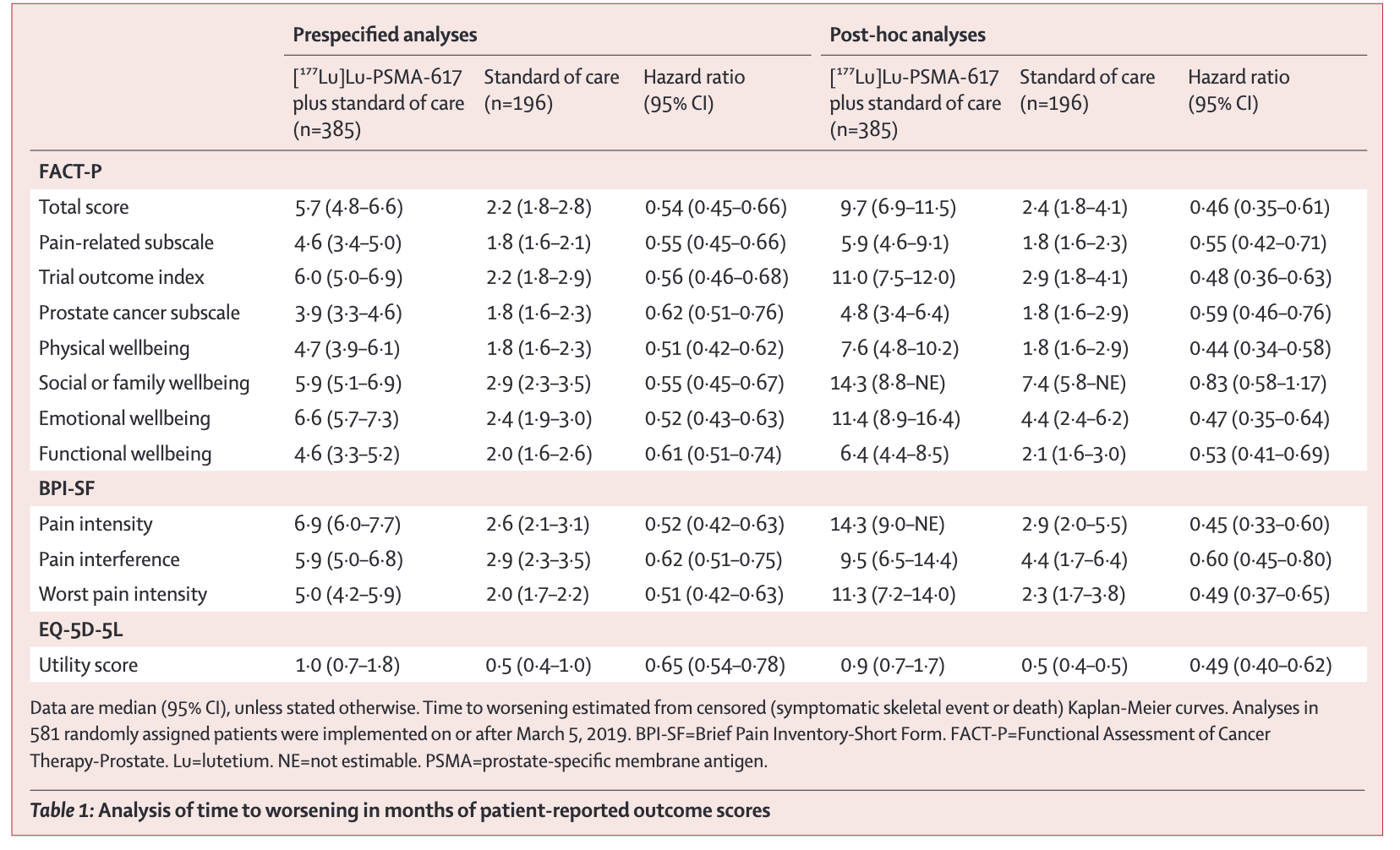
Key Results - FACT-P
Time to worsening of 10 points or more in the FACT-P score, clinical disease progression, or death
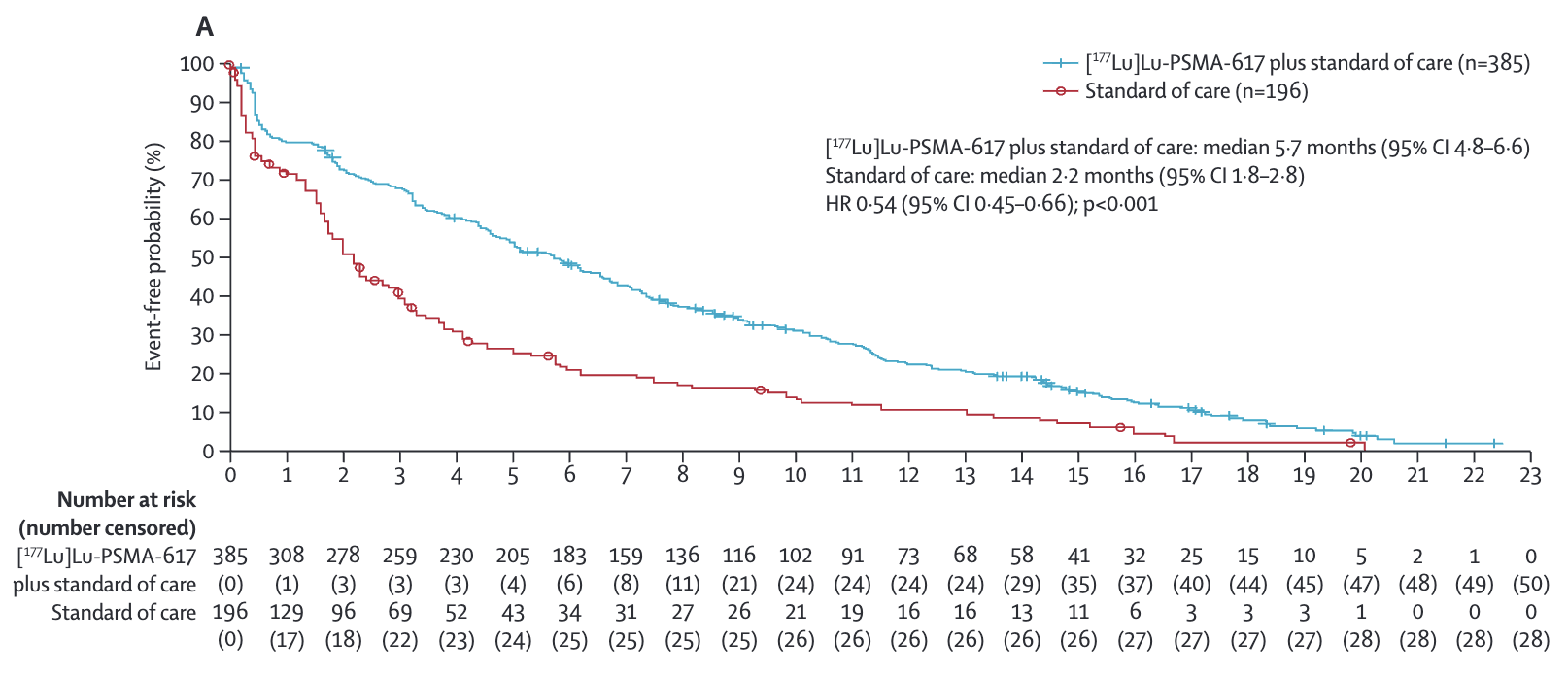
Key Results -
Time to worsening of at least 30% or at least 2 points in the BPI-SF scale,
clinical disease progression, or death
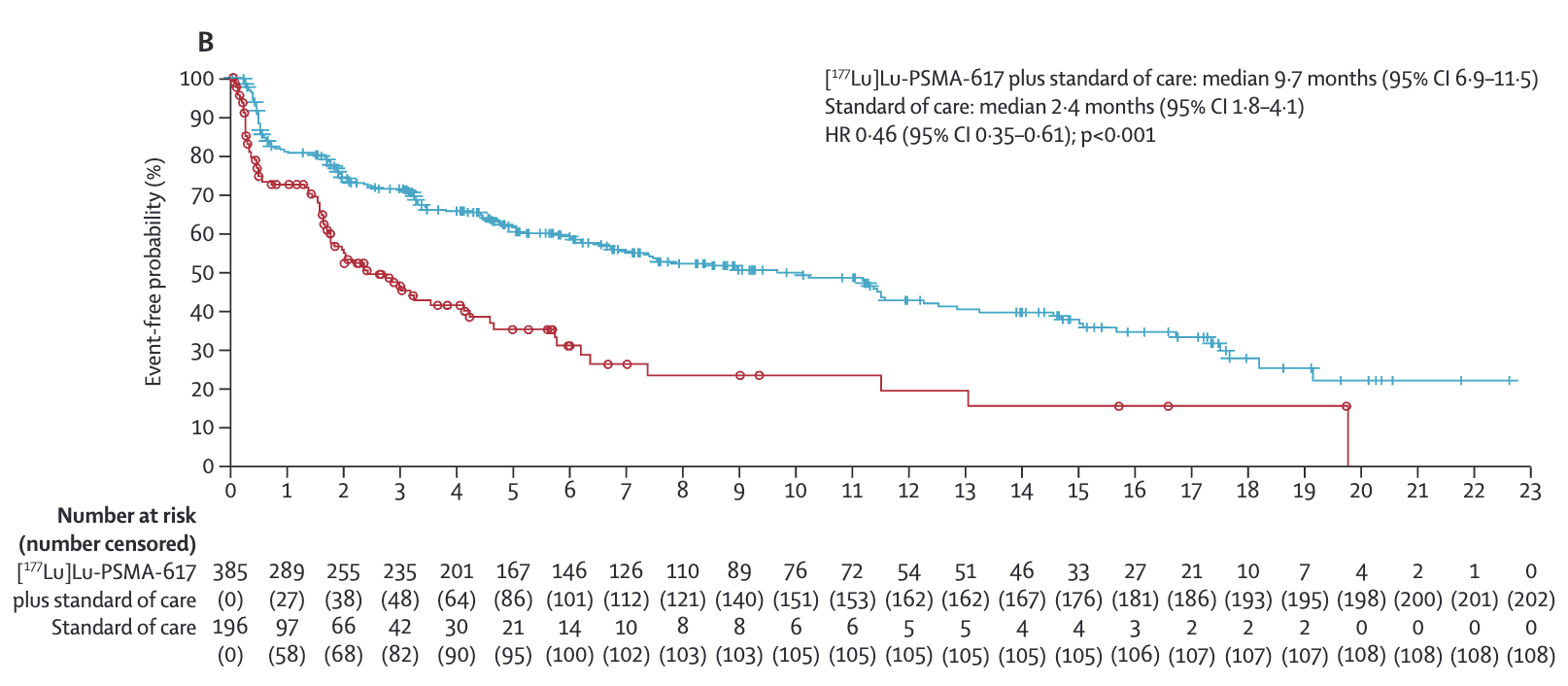
Key Results -
C) or anincrease of at least 2 points inthe BPI-SF scale
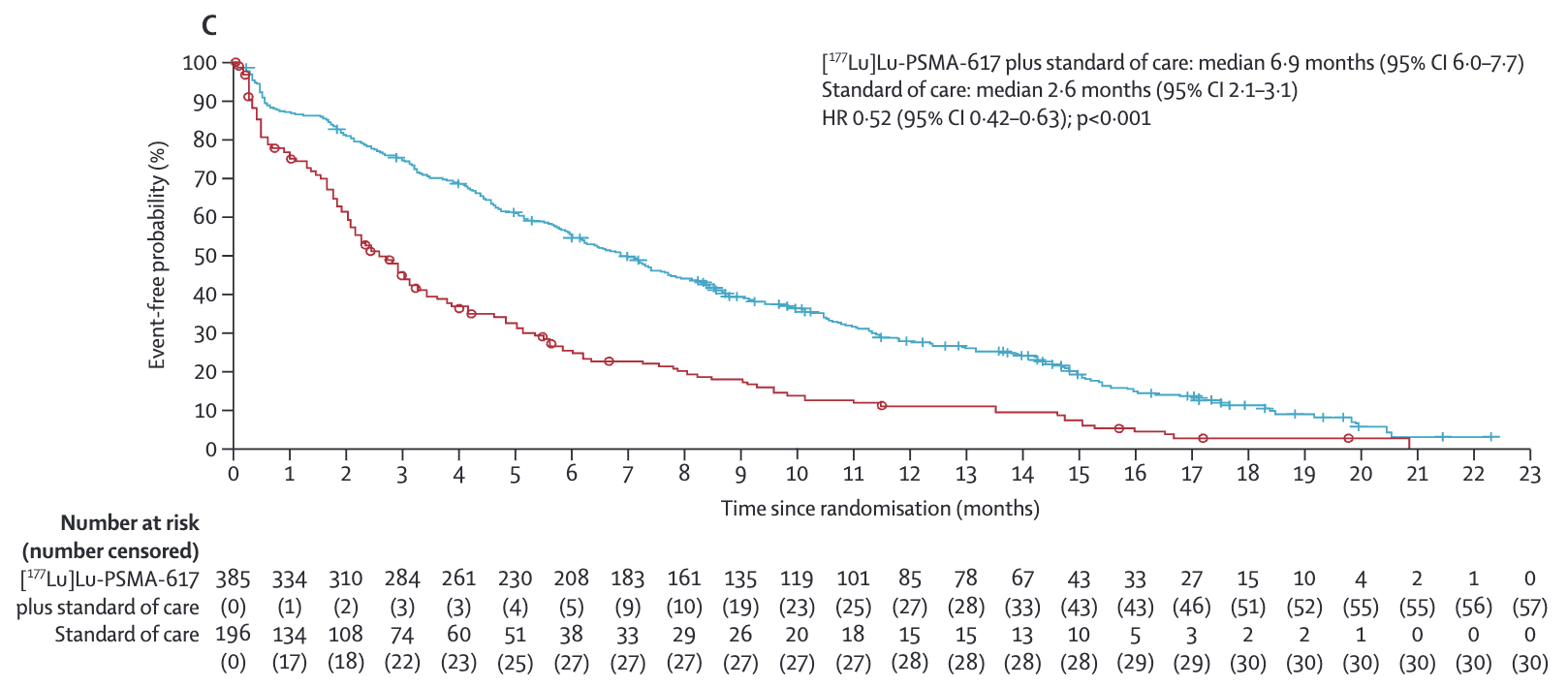
Key Results -
(post hoc; D).Time to worsening inEQ-5D-5L utility score, defined as the time to any decrease inutility score relative to baseline; or no change in score from baseline
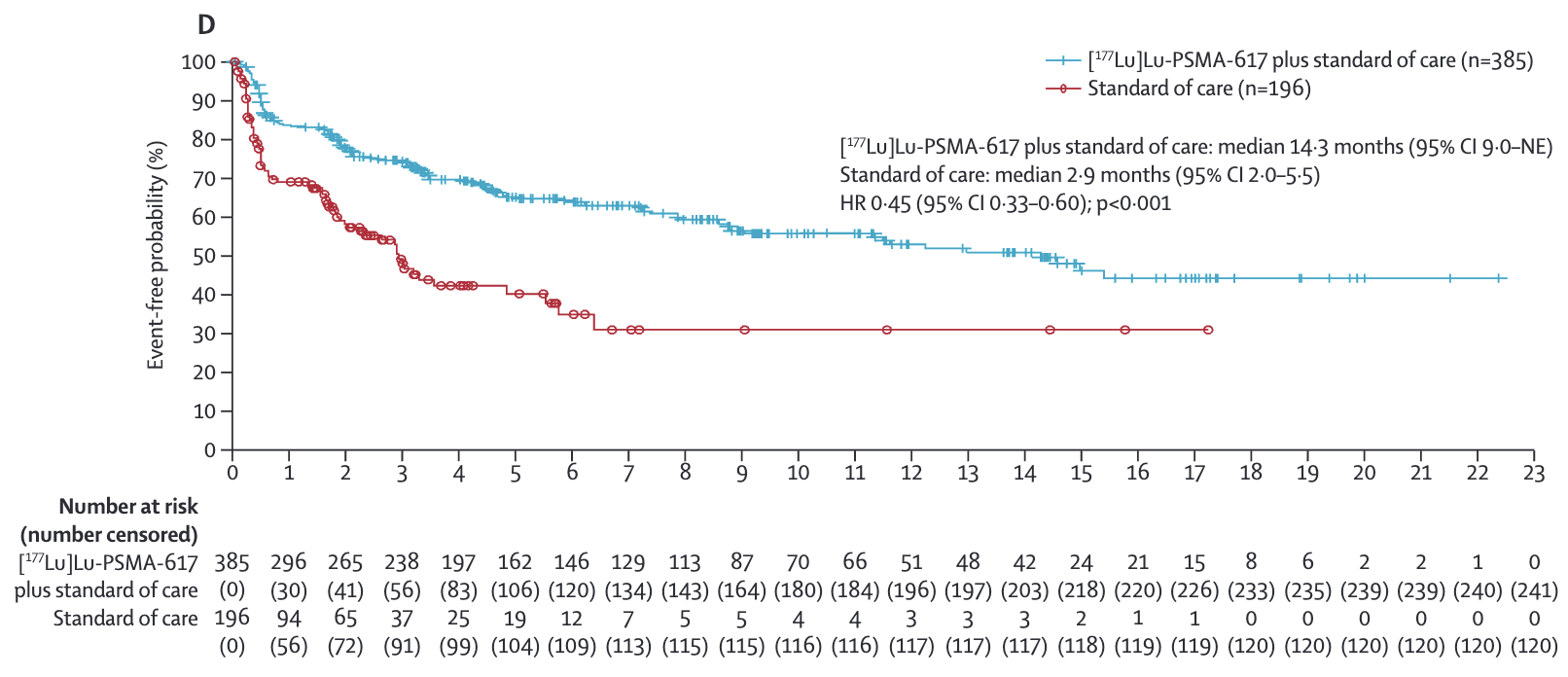
Key Results -
prespecified; E) Time to worsening inEQ-5D-5L utility score, defined as the time to any decrease inutility score relative to baseline; or no change in score from baseline
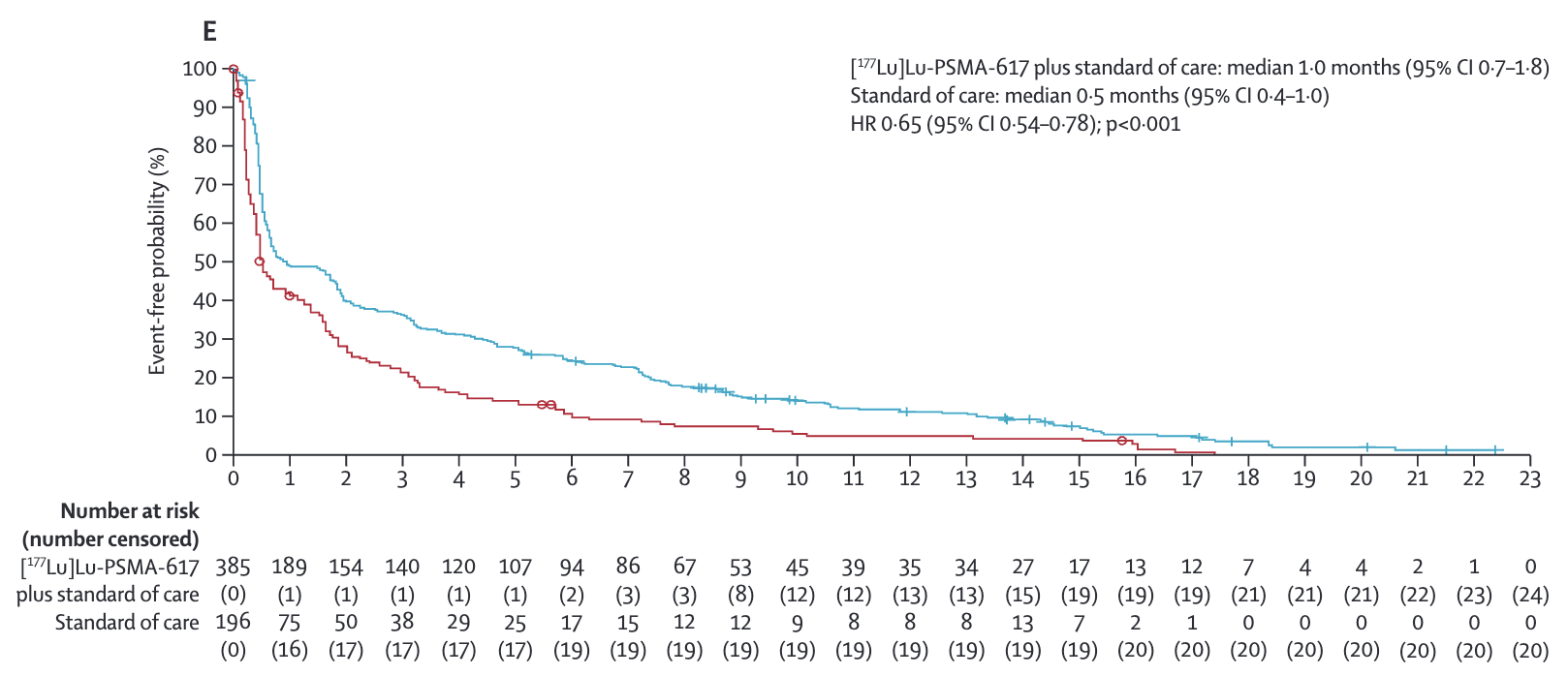
Key Results -
decrease in score of at least 0·10 points from baseline(post hoc; F)
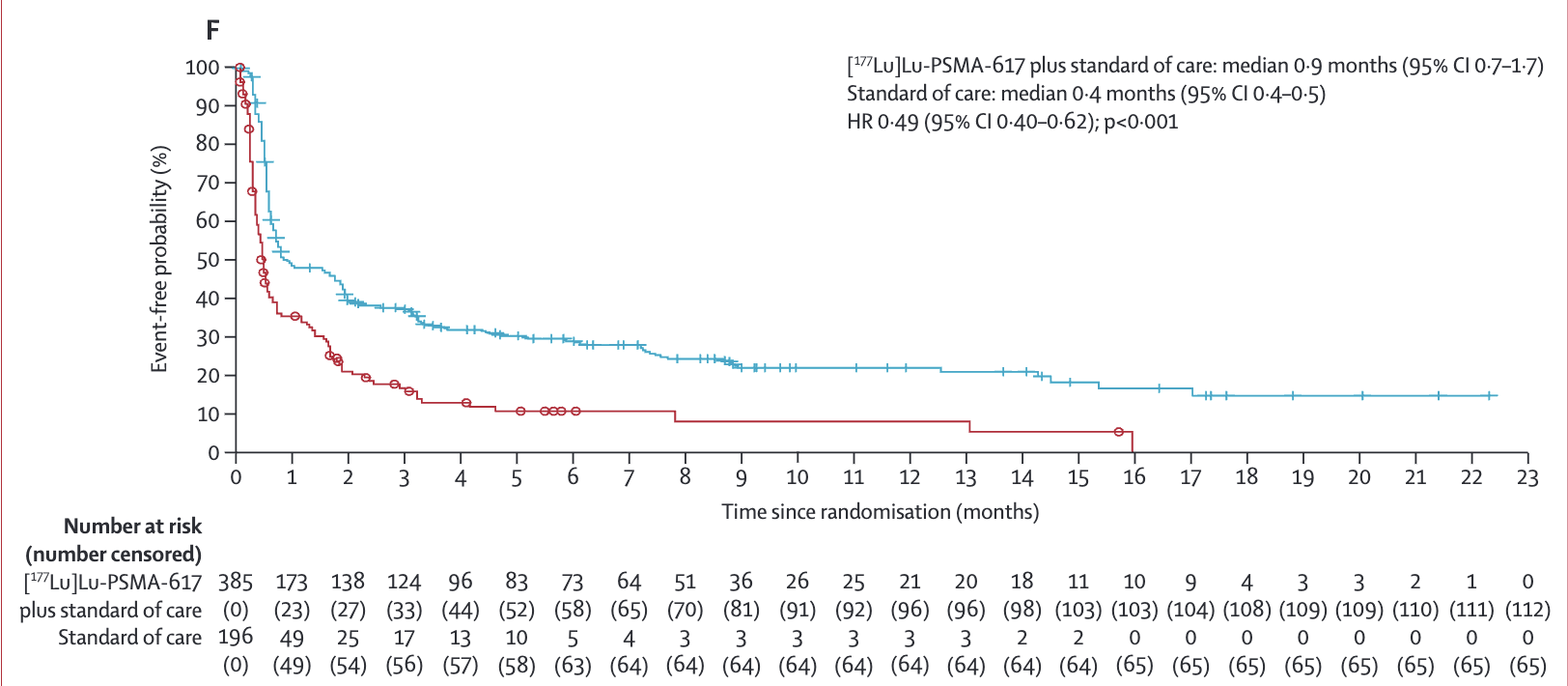
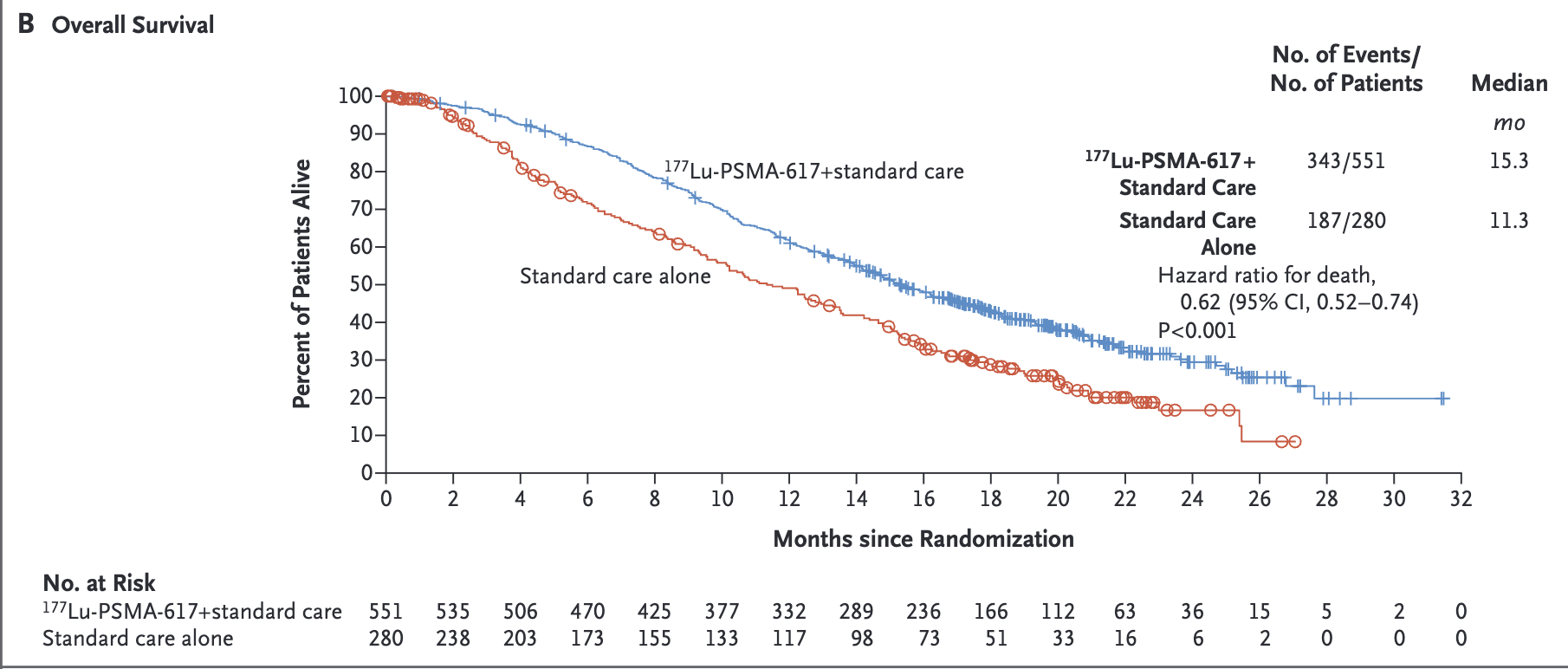
Conclusions
- [177Lu]Lu-PSMA-617 + standard of care improved multiple aspects of HRQOL:
- Delayed time to worsening of FACT-P scores
- Delayed pain progression
- Delayed symptomatic skeletal events
- Manageable safety profile with expected hematologic toxicity
- Results support use in mCRPC patients previously treated with AR pathway inhibitors and taxanes
Discussion Points
- How do these QOL outcomes impact the positioning of Lu-PSMA-617 in the treatment landscape?
- What are implications of improved skeletal-related events for patient management?
- How should the hematologic toxicity profile inform patient selection?
- What are key considerations for implementing Lu-PSMA-617 in clinical practice?
- What questions remain about optimal use of Lu-PSMA-617?