Open PHACTS architecture
Stian Soiland-Reyes
eScience lab, University of Manchester
@soilandreyes
http://orcid.org/0000-0001-9842-9718
2015-09-18
This work is licensed under a
Creative Commons Attribution 4.0 International License.

Linked Data for pharma
Multiple domains:
Bioinformatics (genes, proteins, pathways)
Chemistry (molecules)
Pharmaceutical (compounds, patents)
Medical (diseases, trials)
Loads of Linked Data available
Uniprot
Chembl
DrugBank
DisGeNet
ChemSpider
GeneOntology
WikiPathways
...
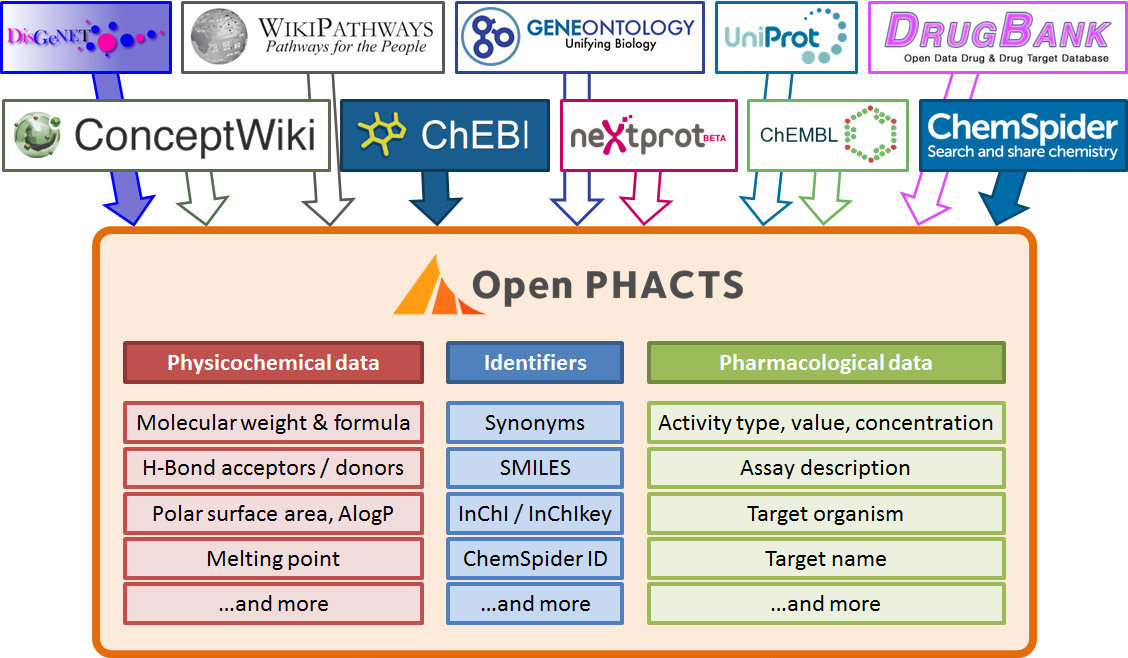
Integrating data mirror
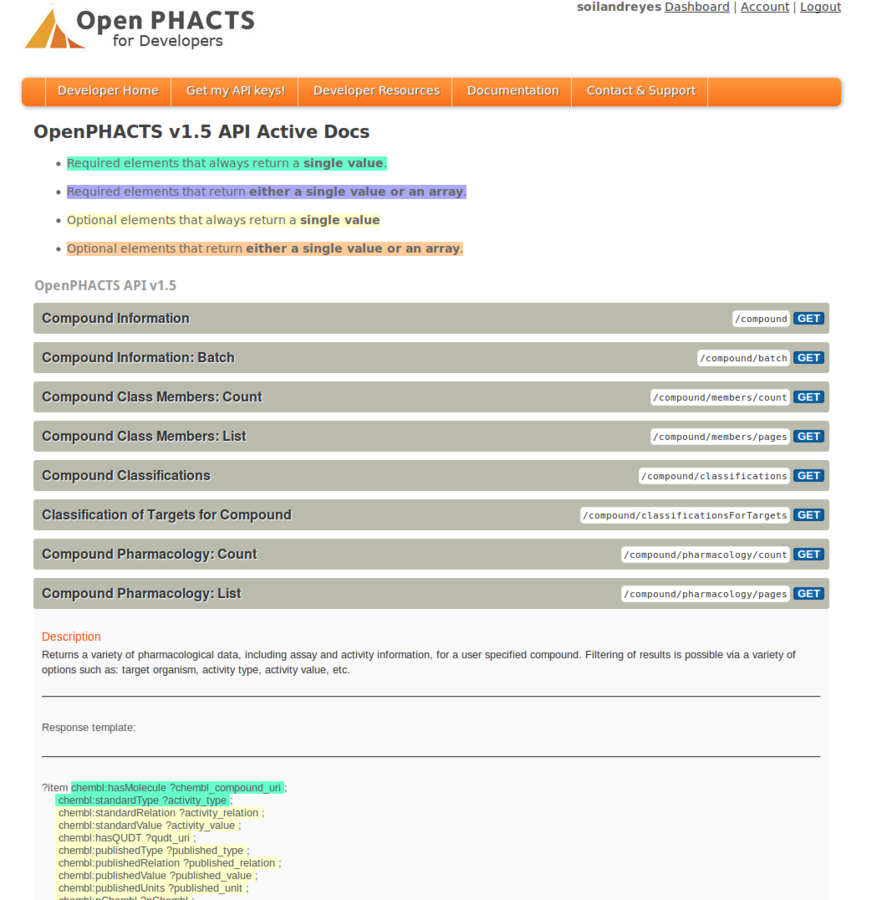
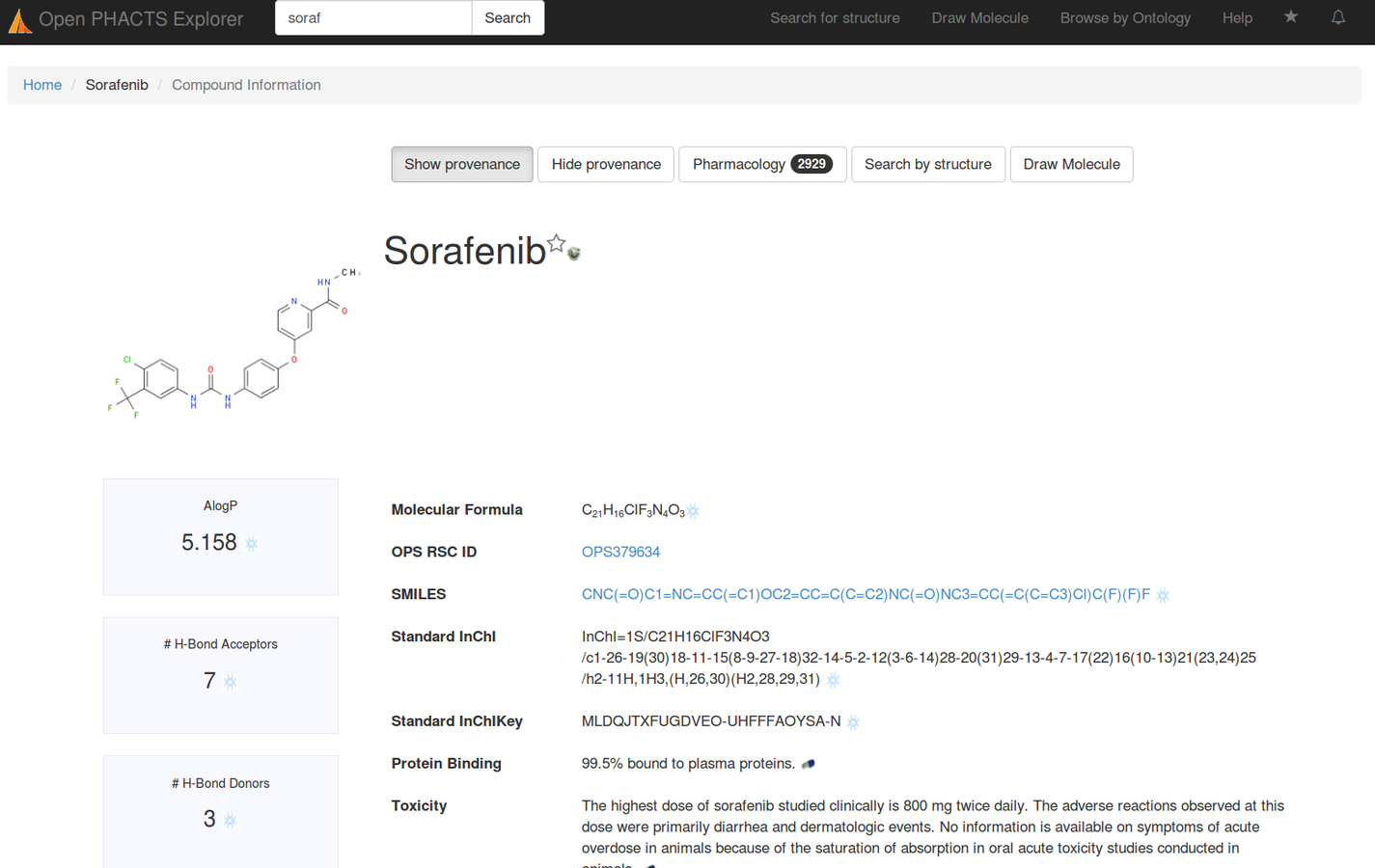
Re-exposed as public APIs
{
"format": "linked-data-api",
"version": "1.5",
"result": {
"_about": "https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=json",
"definition": "https://beta.openphacts.org/api-config",
"extendedMetadataVersion": "https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=json&_metadata=all%2Cviews%2Cformats%2Cexecution%2Cbindings%2Csite",
"linkPredicate": "http://www.w3.org/2004/02/skos/core#exactMatch",
"activeLens": "Default",
"primaryTopic": {
"_about": "http://www.conceptwiki.org/concept/38932552-111f-4a4e-a46a-4ed1d7bdf9d5",
"inDataset": "http://www.conceptwiki.org",
"exactMatch": [
{
"_about": "http://bio2rdf.org/drugbank:DB00398",
"description_en": "Sorafenib (rINN), marketed as Nexavar by Bayer, is a drug approved for the treatment of advanced renal cell carcinoma (primary kidney cancer). It has also received \"Fast Track\" designation by the FDA for the treatment of advanced hepatocellular carcinoma (primary liver cancer), and has since performed well in Phase III trials.\nSorafenib is a small molecular inhibitor of Raf kinase, PDGF (platelet-derived growth factor), VEGF receptor 2 & 3 kinases and c Kit the receptor for Stem cell factor. A growing number of drugs target most of these pathways. The originality of Sorafenib lays in its simultaneous targeting of the Raf/Mek/Erk pathway.",
"description": "Sorafenib (rINN), marketed as Nexavar by Bayer, is a drug approved for the treatment of advanced renal cell carcinoma (primary kidney cancer). It has also received \"Fast Track\" designation by the FDA for the treatment of advanced hepatocellular carcinoma (primary liver cancer), and has since performed well in Phase III trials.\nSorafenib is a small molecular inhibitor of Raf kinase, PDGF (platelet-derived growth factor), VEGF receptor 2 & 3 kinases and c Kit the receptor for Stem cell factor. A growing number of drugs target most of these pathways. The originality of Sorafenib lays in its simultaneous targeting of the Raf/Mek/Erk pathway.",
"drugType_en": [
"investigational",
"approved"
],
"drugType": [
"investigational",
"approved"
],
"genericName_en": "Sorafenib",
"genericName": "Sorafenib",
"metabolism_en": "Sorafenib is metabolized primarily in the liver, undergoing oxidative metabolism, mediated by CYP3A4, as well as glucuronidation mediated by UGT1A9. Sorafenib accounts for approximately 70-85% of the circulating analytes in plasma at steady- state. Eight metabolites of sorafenib have been identified, of which five have been detected in plasma. The main circulating metabolite of sorafenib in plasma, the pyridine N-oxide, shows in vitro potency similar to that of sorafenib. This metabolite comprises approximately 9-16% of circulating analytes at steady-state.",
"metabolism": "Sorafenib is metabolized primarily in the liver, undergoing oxidative metabolism, mediated by CYP3A4, as well as glucuronidation mediated by UGT1A9. Sorafenib accounts for approximately 70-85% of the circulating analytes in plasma at steady- state. Eight metabolites of sorafenib have been identified, of which five have been detected in plasma. The main circulating metabolite of sorafenib in plasma, the pyridine N-oxide, shows in vitro potency similar to that of sorafenib. This metabolite comprises approximately 9-16% of circulating analytes at steady-state.",
"proteinBinding_en": "99.5% bound to plasma proteins.",
"proteinBinding": "99.5% bound to plasma proteins.",
"toxicity_en": "The highest dose of sorafenib studied clinically is 800 mg twice daily. The adverse reactions observed at this dose were primarily diarrhea and dermatologic events. No information is available on symptoms of acute overdose in animals because of the saturation of absorption in oral acute toxicity studies conducted in animals.",
"toxicity": "The highest dose of sorafenib studied clinically is 800 mg twice daily. The adverse reactions observed at this dose were primarily diarrhea and dermatologic events. No information is available on symptoms of acute overdose in animals because of the saturation of absorption in oral acute toxicity studies conducted in animals.",
"inDataset": "http://www.openphacts.org/bio2rdf/drugbank",
"drugInteraction": [
{
"_about": "http://bio2rdf.org/drugbank_resource:DB00398_DB00755",
"text_en": "DDI between Sorafenib and Tretinoin - The strong CYP2C8 inhibitor, Sorafenib, may decrease the metabolism and clearance of oral Tretinoin. Consider alternate therapy or monitor for changes in Tretinoin effectiveness and adverse/toxic effects if Sorafenib is initiated, discontinued to dose changed.",
"text": "DDI between Sorafenib and Tretinoin - The strong CYP2C8 inhibitor, Sorafenib, may decrease the metabolism and clearance of oral Tretinoin. Consider alternate therapy or monitor for changes in Tretinoin effectiveness and adverse/toxic effects if Sorafenib is initiated, discontinued to dose changed.",
"inDataset": "http://www.openphacts.org/bio2rdf/drugbank",
"interactingDrug": "http://bio2rdf.org/drugbank:DB00755"
},
{
"_about": "http://bio2rdf.org/drugbank_resource:DB00398_DB00958",
"text_en": "DDI between Sorafenib and Carboplatin - Sorafenib may enhance the adverse/toxic effect of carboplatin. Concurrent use of sorafenib with carboplatin and placlitaxel in patients with squamous cell lung cancer is contraindicated. The use of this combination in other settings is not specifically contraindicated, but any such use should be approached with added caution.",
"text": "DDI between Sorafenib and Carboplatin - Sorafenib may enhance the adverse/toxic effect of carboplatin. Concurrent use of sorafenib with carboplatin and placlitaxel in patients with squamous cell lung cancer is contraindicated. The use of this combination in other settings is not specifically contraindicated, but any such use should be approached with added caution.",
"inDataset": "http://www.openphacts.org/bio2rdf/drugbank",
"interactingDrug": "http://bio2rdf.org/drugbank:DB00958"
},
{
"_about": "http://bio2rdf.org/drugbank_resource:DB00398_DB06414",
"text_en": "DDI between Sorafenib and Etravirine - Sorafebib, when used concomitantly with etravirine, may experience a decrease in serum concentration. It is recommended to avoid concurrent therapy.",
"text": "DDI between Sorafenib and Etravirine - Sorafebib, when used concomitantly with etravirine, may experience a decrease in serum concentration. It is recommended to avoid concurrent therapy.",
"inDataset": "http://www.openphacts.org/bio2rdf/drugbank",
"interactingDrug": "http://bio2rdf.org/drugbank:DB06414"
},
{
"_about": "http://bio2rdf.org/drugbank_resource:DB00072_DB00398",
"text_en": "DDI between Trastuzumab and Sorafenib - Trastuzumab may increase the risk of neutropenia and anemia. Monitor closely for signs and symptoms of adverse events.",
"text": "DDI between Trastuzumab and Sorafenib - Trastuzumab may increase the risk of neutropenia and anemia. Monitor closely for signs and symptoms of adverse events.",
"inDataset": "http://www.openphacts.org/bio2rdf/drugbank",
"interactingDrug": "http://bio2rdf.org/drugbank:DB00072"
},
{
"_about": "http://bio2rdf.org/drugbank_resource:DB00112_DB00398",
"text_en": "DDI between Bevacizumab and Sorafenib - Monitor therapy due to increased adverse effects of sorafenib, especially hand-foot skin reaction.",
"text": "DDI between Bevacizumab and Sorafenib - Monitor therapy due to increased adverse effects of sorafenib, especially hand-foot skin reaction.",
"inDataset": "http://www.openphacts.org/bio2rdf/drugbank",
"interactingDrug": "http://bio2rdf.org/drugbank:DB00112"
}
]
},
{
"_about": "http://aers.data2semantics.org/resource/drug/SORAFENIB",
"inDataset": "http://aers.data2semantics.org/",
"reportedAdverseEvent": [
{
"_about": "http://aers.data2semantics.org/resource/diagnosis/CARDIAC_FAILURE_ACUTE",
"inDataset": "http://aers.data2semantics.org/",
"prefLabel": "CARDIAC FAILURE ACUTE"
},
{
"_about": "http://aers.data2semantics.org/resource/diagnosis/RENAL_IMPAIRMENT",
"inDataset": "http://aers.data2semantics.org/",
"prefLabel": "RENAL IMPAIRMENT"
},
{
"_about": "http://aers.data2semantics.org/resource/diagnosis/HYPERURICAEMIA",
"inDataset": "http://aers.data2semantics.org/",
"prefLabel": "HYPERURICAEMIA"
},
{
"_about": "http://aers.data2semantics.org/resource/diagnosis/TUMOUR_LYSIS_SYNDROME",
"inDataset": "http://aers.data2semantics.org/",
"prefLabel": "TUMOUR LYSIS SYNDROME"
},
{
"_about": "http://aers.data2semantics.org/resource/diagnosis/LEFT_VENTRICULAR_DYSFUNCTION",
"inDataset": "http://aers.data2semantics.org/",
"prefLabel": "LEFT VENTRICULAR DYSFUNCTION"
},
{
"_about": "http://aers.data2semantics.org/resource/diagnosis/METABOLIC_ACIDOSIS",
"inDataset": "http://aers.data2semantics.org/",
"prefLabel": "METABOLIC ACIDOSIS"
}
],
},
{
"_about": "http://rdf.ebi.ac.uk/resource/chembl/molecule/CHEMBL1336",
"mw_freebase": 464.82,
"inDataset": "http://www.ebi.ac.uk/chembl",
"type": "http://rdf.ebi.ac.uk/terms/chembl#SmallMolecule"
},
{
"_about": "http://ops.rsc.org/OPS379634",
"inDataset": "http://ops.rsc.org",
"hba": 7,
"hbd": 3,
"inchi": "InChI=1S/C21H16ClF3N4O3/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25/h2-11H,1H3,(H,26,30)(H2,28,29,31)",
"inchikey": "MLDQJTXFUGDVEO-UHFFFAOYSA-N",
"logp": 5.158,
"molformula": "C21H16ClF3N4O3",
"molweight": 464.825,
"psa": 92.35,
"ro5_violations": 1,
"rtb": 5,
"smiles": "CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F"
}
],
"prefLabel_en": "Sorafenib",
"prefLabel": "Sorafenib",
"isPrimaryTopicOf": "https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=json"
}
}
}<?xml version="1.0" encoding="utf-8"?>
<result format="linked-data-api" version="1.5" href="https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=xml">
<primaryTopic href="http://www.conceptwiki.org/concept/38932552-111f-4a4e-a46a-4ed1d7bdf9d5">
<prefLabel xml:lang="en">Sorafenib</prefLabel>
<exactMatch>
<item href="http://rdf.ebi.ac.uk/resource/chembl/molecule/CHEMBL1336">
<type href="http://rdf.ebi.ac.uk/terms/chembl#SmallMolecule"/>
<inDataset href="http://www.ebi.ac.uk/chembl"/>
<mw_freebase datatype="double">464.82</mw_freebase>
</item>
<item href="http://ops.rsc.org/OPS379634">
<smiles>CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F</smiles>
<rtb datatype="double">5.0</rtb>
<ro5_violations datatype="double">1.0</ro5_violations>
<psa datatype="double">92.35</psa>
<molweight datatype="double">464.825</molweight>
<molformula>C21H16ClF3N4O3</molformula>
<logp datatype="double">5.158</logp>
<inchikey>MLDQJTXFUGDVEO-UHFFFAOYSA-N</inchikey>
<inchi>InChI=1S/C21H16ClF3N4O3/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25/h2-11H,1H3,(H,26,30)(H2,28,29,31)</inchi>
<hbd datatype="double">3.0</hbd>
<hba datatype="double">7.0</hba>
<inDataset href="http://ops.rsc.org"/>
</item>
<item href="http://aers.data2semantics.org/resource/drug/NEXAVAR">
<prefLabel>NEXAVAR</prefLabel>
<reportedAdverseEvent>
<item href="http://aers.data2semantics.org/resource/diagnosis/HEAD_INJURY">
<prefLabel>HEAD INJURY</prefLabel>
<inDataset href="http://aers.data2semantics.org/"/>
</item>
<item href="http://aers.data2semantics.org/resource/diagnosis/SUPRAVENTRICULAR_TACHYCARDIA">
<prefLabel>SUPRAVENTRICULAR TACHYCARDIA</prefLabel>
<inDataset href="http://aers.data2semantics.org/"/>
</item>
<!-- .. -->
</reportedAdverseEvent>
<inDataset href="http://aers.data2semantics.org/"/>
</item>
<item href="http://www.conceptwiki.org/concept/38932552-111f-4a4e-a46a-4ed1d7bdf9d5"/>
<item href="http://bio2rdf.org/drugbank:DB00398">
<drugInteraction>
<item href="http://bio2rdf.org/drugbank_resource:DB00398_DB00755">
<interactingDrug href="http://bio2rdf.org/drugbank:DB00755"/>
<inDataset href="http://www.openphacts.org/bio2rdf/drugbank"/>
<text xml:lang="en">DDI between Sorafenib and Tretinoin - The strong CYP2C8 inhibitor, Sorafenib, may decrease the metabolism and clearance of oral Tretinoin. Consider alternate therapy or monitor for changes in Tretinoin effectiveness and adverse/toxic effects if Sorafenib is initiated, discontinued to dose changed.</text>
</item>
<item href="http://bio2rdf.org/drugbank_resource:DB00398_DB00958">
<interactingDrug href="http://bio2rdf.org/drugbank:DB00958"/>
<inDataset href="http://www.openphacts.org/bio2rdf/drugbank"/>
<text xml:lang="en">DDI between Sorafenib and Carboplatin - Sorafenib may enhance the adverse/toxic effect of carboplatin. Concurrent use of sorafenib with carboplatin and placlitaxel in patients with squamous cell lung cancer is contraindicated. The use of this combination in other settings is not specifically contraindicated, but any such use should be approached with added caution.</text>
</item>
<!-- .. -->
</drugInteraction>
<inDataset href="http://www.openphacts.org/bio2rdf/drugbank"/>
<toxicity xml:lang="en">The highest dose of sorafenib studied clinically is 800 mg twice daily. The adverse reactions observed at this dose were primarily diarrhea and dermatologic events. No information is available on symptoms of acute overdose in animals because of the saturation of absorption in oral acute toxicity studies conducted in animals.</toxicity>
<proteinBinding xml:lang="en">99.5% bound to plasma proteins.</proteinBinding>
<metabolism xml:lang="en">Sorafenib is metabolized primarily in the liver, undergoing oxidative metabolism, mediated by CYP3A4, as well as glucuronidation mediated by UGT1A9. Sorafenib accounts for approximately 70-85% of the circulating analytes in plasma at steady- state. Eight metabolites of sorafenib have been identified, of which five have been detected in plasma. The main circulating metabolite of sorafenib in plasma, the pyridine N-oxide, shows <i>in vitro</i> potency similar to that of sorafenib. This metabolite comprises approximately 9-16% of circulating analytes at steady-state.</metabolism>
<genericName xml:lang="en">Sorafenib</genericName>
<drugType>
<item xml:lang="en">investigational</item>
<item xml:lang="en">approved</item>
</drugType>
<description xml:lang="en">Sorafenib (rINN), marketed as Nexavar by Bayer, is a drug approved for the treatment of advanced renal cell carcinoma (primary kidney cancer). It has also received "Fast Track" designation by the FDA for the treatment of advanced hepatocellular carcinoma (primary liver cancer), and has since performed well in Phase III trials.
Sorafenib is a small molecular inhibitor of Raf kinase, PDGF (platelet-derived growth factor), VEGF receptor 2 & 3 kinases and c Kit the receptor for Stem cell factor. A growing number of drugs target most of these pathways. The originality of Sorafenib lays in its simultaneous targeting of the Raf/Mek/Erk pathway.</description>
</item>
</exactMatch>
<inDataset href="http://www.conceptwiki.org"/>
</primaryTopic>
<activeLens>Default</activeLens>
<linkPredicate href="http://www.w3.org/2004/02/skos/core#exactMatch"/>
<extendedMetadataVersion href="https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=xml&_metadata=all%2Cviews%2Cformats%2Cexecution%2Cbindings%2Csite"/>
<definition href="https://beta.openphacts.org/api-config"/>
</result>
@prefix rdf: <http://www.w3.org/1999/02/22-rdf-syntax-ns#> .
@prefix skos: <http://www.w3.org/2004/02/skos/core#> .
@prefix void: <http://rdfs.org/ns/void#> .
@prefix foaf: <http://xmlns.com/foaf/0.1/> .
@prefix ns0: <http://www.openphacts.org/api#> .
@prefix ns1: <http://bio2rdf.org/> .
@prefix ns2: <http://rdf.ebi.ac.uk/terms/chembl#> .
@prefix chembl1336: <http://rdf.ebi.ac.uk/resource/chembl/molecule/CHEMBL1336#> .
@prefix linked-data: <http://purl.org/linked-data/api/vocab#> .
@prefix msg0: <http://www.openphacts.org/api/> .
<http://www.conceptwiki.org/concept/38932552-111f-4a4e-a46a-4ed1d7bdf9d5>
skos:exactMatch <http://aers.data2semantics.org/resource/drug/NEXAVAR> ;
skos:exactMatch <http://aers.data2semantics.org/resource/drug/SORAFENIB> ;
skos:exactMatch <http://www.conceptwiki.org/concept/38932552-111f-4a4e-a46a-4ed1d7bdf9d5> ;
skos:exactMatch <http://bio2rdf.org/drugbank:DB00398> ;
skos:exactMatch <http://rdf.ebi.ac.uk/resource/chembl/molecule/CHEMBL1336> ;
skos:exactMatch <http://ops.rsc.org/OPS379634> ;
skos:prefLabel "Sorafenib"@en ;
void:inDataset <http://www.conceptwiki.org> ;
foaf:isPrimaryTopicOf <https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=ttl> .
<https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=ttl>
foaf:primaryTopic <http://www.conceptwiki.org/concept/38932552-111f-4a4e-a46a-4ed1d7bdf9d5> ;
linked-data:definition <https://beta.openphacts.org/api-config> ;
msg0:activeLens "Default" ;
void:linkPredicate skos:exactMatch ;
linked-data:extendedMetadataVersion <https://beta.openphacts.org/1.5/compound?uri=http%3A%2F%2Fwww.conceptwiki.org%2Fconcept%2F38932552-111f-4a4e-a46a-4ed1d7bdf9d5&app_id=161aeb7d&app_key=bbcba81896020f0b95e3dd35b55e3345&_format=ttl&_metadata=all%2Cviews%2Cformats%2Cexecution%2Cbindings%2Csite> .
<http://ops.rsc.org/OPS379634>
void:inDataset <http://ops.rsc.org> ;
ns0:smiles "CNC(=O)C1=NC=CC(=C1)OC2=CC=C(C=C2)NC(=O)NC3=CC(=C(C=C3)Cl)C(F)(F)F" ;
ns0:inchi "InChI=1S/C21H16ClF3N4O3/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25/h2-11H,1H3,(H,26,30)(H2,28,29,31)" ;
ns0:inchikey "MLDQJTXFUGDVEO-UHFFFAOYSA-N" ;
ns0:logp "5.158"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:hba "7.0"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:hbd "3.0"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:ro5_violations "1.0"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:psa "92.35"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:rtb "5.0"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:molweight "464.825"^^<http://www.w3.org/2001/XMLSchema#double> ;
ns0:molformula "C21H16ClF3N4O3" .
<http://bio2rdf.org/drugbank_resource:DB00398_DB00755>
void:inDataset <http://www.openphacts.org/bio2rdf/drugbank> ;
ns0:interactingDrug <http://bio2rdf.org/drugbank:DB00755> ;
<http://bio2rdf.org/drugbank_vocabulary:text> "DDI between Sorafenib and Tretinoin - The strong CYP2C8 inhibitor, Sorafenib, may decrease the metabolism and clearance of oral Tretinoin. Consider alternate therapy or monitor for changes in Tretinoin effectiveness and adverse/toxic effects if Sorafenib is initiated, discontinued to dose changed."@en .
<http://bio2rdf.org/drugbank_resource:DB00398_DB00958>
void:inDataset <http://www.openphacts.org/bio2rdf/drugbank> ;
ns0:interactingDrug <http://bio2rdf.org/drugbank:DB00958> ;
<http://bio2rdf.org/drugbank_vocabulary:text> "DDI between Sorafenib and Carboplatin - Sorafenib may enhance the adverse/toxic effect of carboplatin. Concurrent use of sorafenib with carboplatin and placlitaxel in patients with squamous cell lung cancer is contraindicated. The use of this combination in other settings is not specifically contraindicated, but any such use should be approached with added caution."@en .
<http://bio2rdf.org/drugbank_resource:DB00398_DB06414>
void:inDataset <http://www.openphacts.org/bio2rdf/drugbank> ;
ns0:interactingDrug <http://bio2rdf.org/drugbank:DB06414> ;
<http://bio2rdf.org/drugbank_vocabulary:text> "DDI between Sorafenib and Etravirine - Sorafebib, when used concomitantly with etravirine, may experience a decrease in serum concentration. It is recommended to avoid concurrent therapy."@en .
<http://aers.data2semantics.org/resource/drug/NEXAVAR>
skos:prefLabel "NEXAVAR" ;
void:inDataset <http://aers.data2semantics.org/> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/HEAD_INJURY> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/SUPRAVENTRICULAR_TACHYCARDIA> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/SWOLLEN_TONGUE> ;
# ...
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/APHAGIA> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/LIVER_FUNCTION_TEST_ABNORMAL> .
<http://aers.data2semantics.org/resource/drug/SORAFENIB>
skos:prefLabel "SORAFENIB" ;
void:inDataset <http://aers.data2semantics.org/> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/CARDIAC_FAILURE_ACUTE> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/RENAL_IMPAIRMENT> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/HYPERURICAEMIA> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/TUMOUR_LYSIS_SYNDROME> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/LEFT_VENTRICULAR_DYSFUNCTION> ;
ns0:reportedAdverseEvent <http://aers.data2semantics.org/resource/diagnosis/METABOLIC_ACIDOSIS> .
<http://bio2rdf.org/drugbank:DB00398>
void:inDataset <http://www.openphacts.org/bio2rdf/drugbank> ;
<http://bio2rdf.org/drugbank_vocabulary:metabolism> "Sorafenib is metabolized primarily in the liver, undergoing oxidative metabolism, mediated by CYP3A4, as well as glucuronidation mediated by UGT1A9. Sorafenib accounts for approximately 70-85% of the circulating analytes in plasma at steady- state. Eight metabolites of sorafenib have been identified, of which five have been detected in plasma. The main circulating metabolite of sorafenib in plasma, the pyridine N-oxide, shows <i>in vitro</i> potency similar to that of sorafenib. This metabolite comprises approximately 9-16% of circulating analytes at steady-state."@en ;
<http://bio2rdf.org/drugbank_vocabulary:toxicity> "The highest dose of sorafenib studied clinically is 800 mg twice daily. The adverse reactions observed at this dose were primarily diarrhea and dermatologic events. No information is available on symptoms of acute overdose in animals because of the saturation of absorption in oral acute toxicity studies conducted in animals."@en ;
<http://bio2rdf.org/drugbank_vocabulary:description> "Sorafenib (rINN), marketed as Nexavar by Bayer, is a drug approved for the treatment of advanced renal cell carcinoma (primary kidney cancer). It has also received \"Fast Track\" designation by the FDA for the treatment of advanced hepatocellular carcinoma (primary liver cancer), and has since performed well in Phase III trials.\nSorafenib is a small molecular inhibitor of Raf kinase, PDGF (platelet-derived growth factor), VEGF receptor 2 & 3 kinases and c Kit the receptor for Stem cell factor. A growing number of drugs target most of these pathways. The originality of Sorafenib lays in its simultaneous targeting of the Raf/Mek/Erk pathway."@en ;
<http://bio2rdf.org/drugbank_vocabulary:proteinBinding> "99.5% bound to plasma proteins."@en ;
<http://bio2rdf.org/drugbank_vocabulary:genericName> "Sorafenib"@en ;
<http://bio2rdf.org/drugbank_vocabulary:drugType> "investigational"@en ;
<http://bio2rdf.org/drugbank_vocabulary:drugType> "approved"@en ;
ns0:drugInteraction <http://bio2rdf.org/drugbank_resource:DB00398_DB00755> ;
ns0:drugInteraction <http://bio2rdf.org/drugbank_resource:DB00398_DB00958> ;
ns0:drugInteraction <http://bio2rdf.org/drugbank_resource:DB00398_DB06414> ;
ns0:drugInteraction <http://bio2rdf.org/drugbank_resource:DB00072_DB00398> ;
ns0:drugInteraction <http://bio2rdf.org/drugbank_resource:DB00112_DB00398> .
<http://bio2rdf.org/drugbank_resource:DB00072_DB00398>
void:inDataset <http://www.openphacts.org/bio2rdf/drugbank> ;
ns0:interactingDrug <http://bio2rdf.org/drugbank:DB00072> ;
<http://bio2rdf.org/drugbank_vocabulary:text> "DDI between Trastuzumab and Sorafenib - Trastuzumab may increase the risk of neutropenia and anemia. Monitor closely for signs and symptoms of adverse events."@en .
<http://bio2rdf.org/drugbank_resource:DB00112_DB00398>
void:inDataset <http://www.openphacts.org/bio2rdf/drugbank> ;
ns0:interactingDrug <http://bio2rdf.org/drugbank:DB00112> ;
<http://bio2rdf.org/drugbank_vocabulary:text> "DDI between Bevacizumab and Sorafenib - Monitor therapy due to increased adverse effects of sorafenib, especially hand-foot skin reaction."@en .
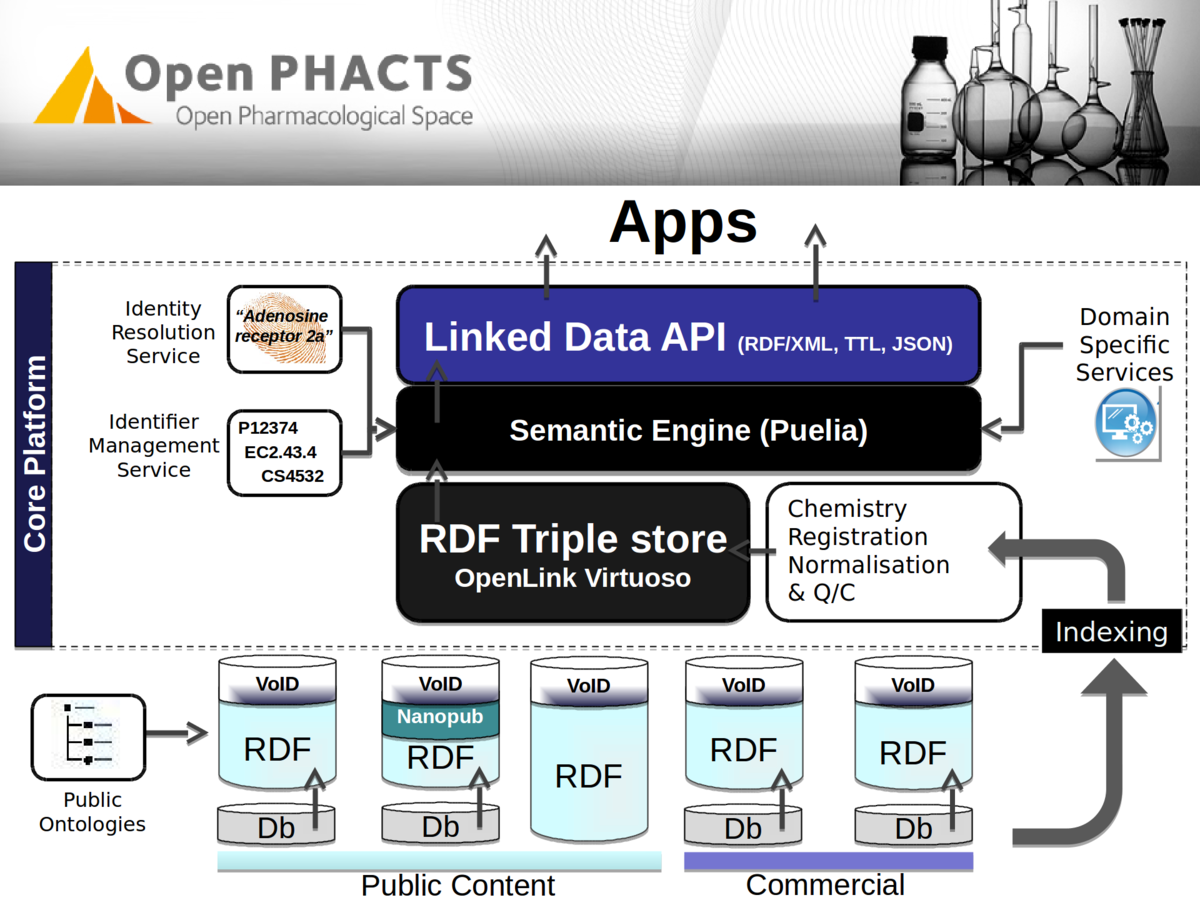
Data loading
The identity problem
Are These Two Molecules The Same(*)?
*Really: Is it sensible to combine data associated with these two molecules?
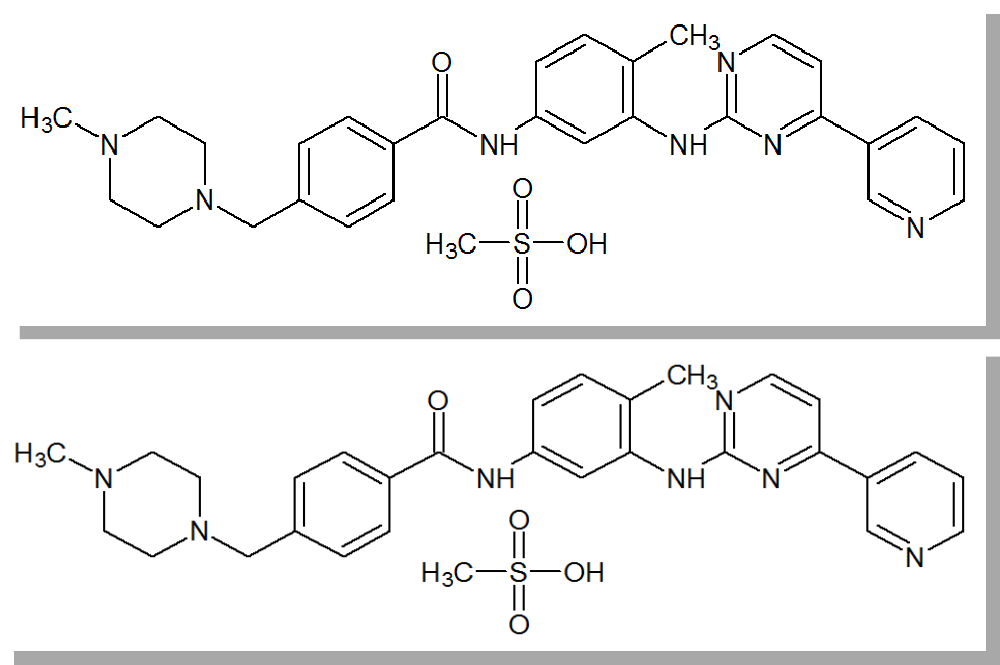
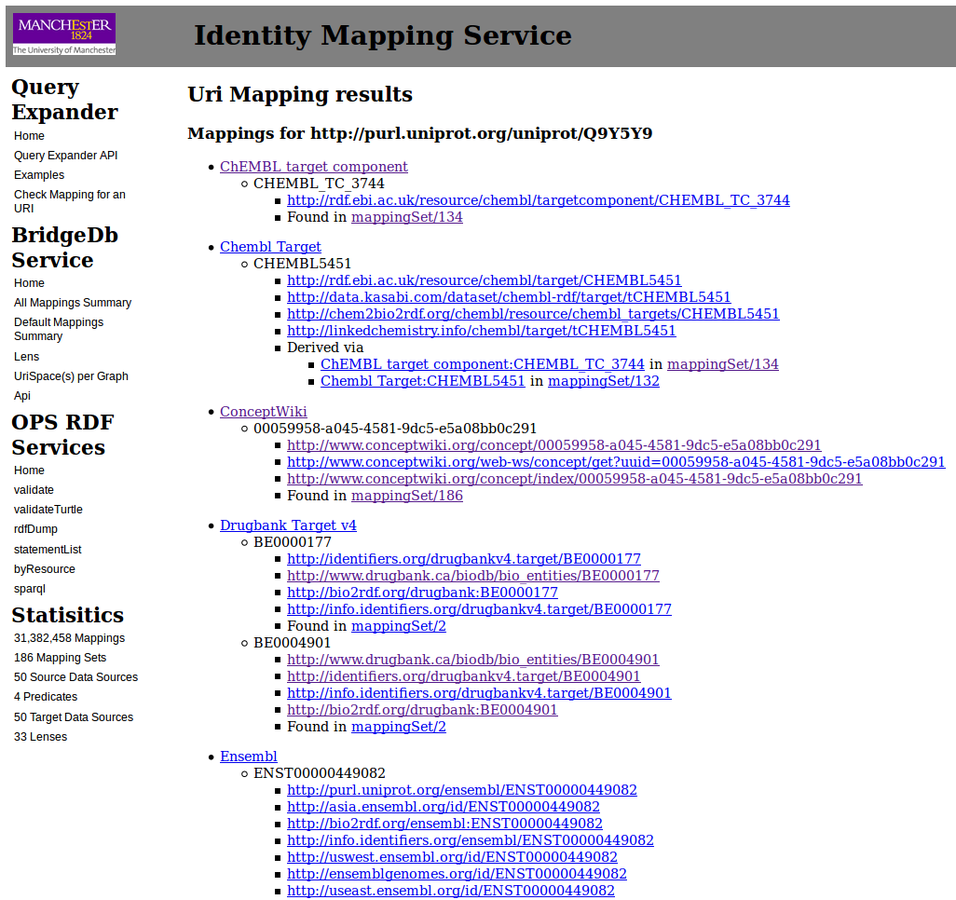
Following transitives
VoID linkset descriptions
:chembl_rdf_dataset a void:Dataset ;
dcterms:title "The ChEMBL Database" ;
dcterms:description "ChEMBL is a database of bioactive drug-like small molecules, it contains 2-D structures, calculated properties (e.g. logP, Molecular Weight, Lipinski Parameters, etc.) and abstracted bioactivities (e.g. binding constants, pharmacology and ADMET data). The data is abstracted and curated from the primary scientific literature, and cover a significant fraction of the SAR and discovery of modern drugs." ;
pav:createdBy <http://orcid.org/0000-0003-2634-7400> ;
pav:createdOn "2009-10-28T00:00:00.000Z"^^xsd:dateTime ;
pav:lastUpdateOn "2015-01-14T00:00:00.000Z"^^xsd:dateTime ;
dcat:landingPage <https://www.ebi.ac.uk/chembl> ;
foaf:page <ftp://ftp.ebi.ac.uk/pub/databases/chembl/> ;
dcterms:license <http://creativecommons.org/licenses/by-sa/3.0/> ;
void:uriSpace "http://rdf.ebi.ac.uk/resource/chembl/" ;
pav:version "20.1" ;
pav:previousVersion <http://rdf.ebi.ac.uk/dataset/chembl/20.0/void.ttl#chembl_rdf_dataset> ;
dcterms:publisher <https://www.ebi.ac.uk/> ;
dcterms:created "2009-10-28T00:00:00.000Z"^^xsd:dateTime ;
dcterms:modified "2015-01-14T00:00:00.000Z"^^xsd:dateTime ;
void:subset :chembl_rdf_target_relation_dataset , :chembl_rdf_cell_line_dataset , :chembl_rdf_molhierarchy_dataset , :chembl_rdf_bindingsite_dataset , :chembl_rdf_moa_dataset , :chembl_rdf_molecule_dataset , :chembl_rdf_biocmpt_dataset , :chembl_rdf_target_dataset , :chembl_rdf_targetcmpt_dataset , :chembl_rdf_assay_dataset , :chembl_rdf_activity_dataset , :chembl_rdf_document_dataset , :chembl_rdf_journal_dataset , :chembl_rdf_protclass_dataset , :chembl_rdf_source_dataset , :chembl_rdf_unichem_dataset ;
void:vocabulary <http://purl.org/ontology/bibo> , <http://www.bioassayontology.org/bao> , <http://semanticscience.org/ontology/cheminf.owl> , <http://purl.org/spar/cito> , <http://purl.org/dc/terms> , <http://www.w3.org/2002/07/owl> , <http://www.w3.org/1999/02/22-rdf-syntax-ns> , <http://www.w3.org/2000/01/rdf-schema> , <http://semanticscience.org/ontology/sio.owl> , <http://www.w3.org/2004/02/skos/core> , <http://www.w3.org/2001/XMLSchema> ;
void:exampleResource chembl_molecule:CHEMBL941 ;
void:sparqlEndpoint <http://www.ebi.ac.uk/rdf/services/chembl/sparql> ;
voag:frequencyOfChange freq:quarterly .
:molecule_chebi_linkset a void:Linkset ;
dcterms:title "The ChEMBL Molecule to ChEBI Molecule Linkset" ;
dcterms:description "The ChEMBL Molecule to ChEBI Molecule Linkset" ;
dcterms:publisher <http://www.ebi.ac.uk/> ;
dcterms:license <http://creativecommons.org/licenses/by-sa/3.0/> ;
dcterms:issued "2015-01-27T15:29:59.000Z"^^xsd:dateTime ;
void:dataDump <ftp://ftp.ebi.ac.uk/pub/databases/chembl/ChEMBL-RDF/20.1/chembl_20.1_molecule_chebi_ls.ttl.gz> ;
void:subjectsTarget :chembl_rdf_molecule_dataset ;
void:objectsTarget :chebi_dataset ;
void:linkPredicate skos:exactMatch ;
dul:expresses sio:CHEMINF_000059 ;
pav:authoredBy <http://orcid.org/0000-0003-2634-7400> ;
pav:authoredOn "2015-09-09T09:53:07.000+01:00"^^xsd:dateTime ;
pav:createdBy <http://orcid.org/0000-0003-2634-7400> ;
pav:createdOn "2015-09-09T09:53:07.000+01:00"^^xsd:dateTime .
:chembl_singletarget_targetcmpt_linkset a void:Linkset ;
dcterms:title "The ChEMBL Single Protein Target to Target Component Linkset" ;
dcterms:description "The ChEMBL Single Protein Target to Target Component Linkset maps ChEMBL targets that are single proteins to their equivalent target component (a simple 1:1 mapping)." ;
dcterms:publisher <http://www.ebi.ac.uk/> ;
dcterms:license <http://creativecommons.org/licenses/by-sa/3.0/> ;
dcterms:issued "2015-09-09T09:53:07.000+01:00"^^xsd:dateTime ;
void:dataDump <ftp://ftp.ebi.ac.uk/pub/databases/chembl/ChEMBL-RDF/20.1/chembl_20.1_singletarget_targetcmpt_ls.ttl.gz> ;
void:subjectsTarget :chembl_rdf_target_dataset ;
void:objectsTarget :chembl_rdf_targetcmpt_dataset ;
void:linkPredicate skos:exactMatch ;
dul:expresses sio:SIO_010043 ;
pav:authoredBy <http://orcid.org/0000-0003-2634-7400> ;
pav:authoredOn "2015-09-09T09:53:07.000+01:00"^^xsd:dateTime ;
pav:createdBy <http://orcid.org/0000-0003-2634-7400> ;
pav:createdOn "2015-09-09T09:53:07.000+01:00"^^xsd:dateTime .
How do we find those identifiers?
Next: ops-search
API: Text Query to URI
ElasticSearch-based
Indexes triple stores by SPARQL
Selects label-like properties
TODO: Integrate with Identity mapping service
How do we load data?
Manual ad-hoc approach
Dataset update - Mirror and load RDF @dev
Ontology changes - Edit queries
URI changes - Update linksets and IMS
Manual testing - does it still integrate?
Next: Maven data builds
Maven project per dataset/linkset, which:
- Downloads dataset from upstream
- Clean/filter data
- Versioned, licensed
Built by Jenkins CI
- Deploys to Maven repository
TODO: Make a Research Object,
add provenance/VoID
<plugin>
<groupId>org.codehaus.mojo</groupId>
<artifactId>wagon-maven-plugin</artifactId>
<version>1.0</version>
<executions>
<execution>
<id>download-rdf</id>
<phase>generate-resources</phase>
<goals>
<goal>download</goal>
</goals>
<configuration>
<includes>*.ttl.gz</includes>
<url>http://ftp.ebi.ac.uk/pub/databases/chembl/ChEMBL-RDF/${chembl.version}</url>
<toDir>${project.build.outputDirectory}/data/${project.artifactId}</toDir>
</configuration>
</execution>Continuous data integration
Running Open PHACTS on Docker
Docker overview
Linux container technology
Download images from Docker Hub
Philosophy: One application per container
ops-docker

Docker Compose
1) Data staging
- Downloads from http://data.openphacts.org/1.5/
2) Starts linked Docker containers
3) Customizable per container
Provides: Explorer, API, Virtuoso, IMS


