| AME | PL / Headache | Central Nervous System | Treatment | Security | Benefits |
|---|
Musclease

Breathing
Speaking
Swallowing
Movement
| AME | PL / Headache | Central Nervous System | Treatment | Security | Benefits |
|---|
Spinal muscular atrophy (SMA) is a genetic disease of the central nervous system.
Independence

Breathing
Speak
Swallowing
March
Spinal muscular atrophy (SMA) is a genetic disease of the central nervous system.
1
Independence
| AME | PL / Headache | Central Nervous System | Treatment | Security | Benefits |
|---|

Common procedure.
2
Important diagnostic tool in Neurology.
2
Direct administration of drugs in the Central Nervous System.
3
2
Proven security.
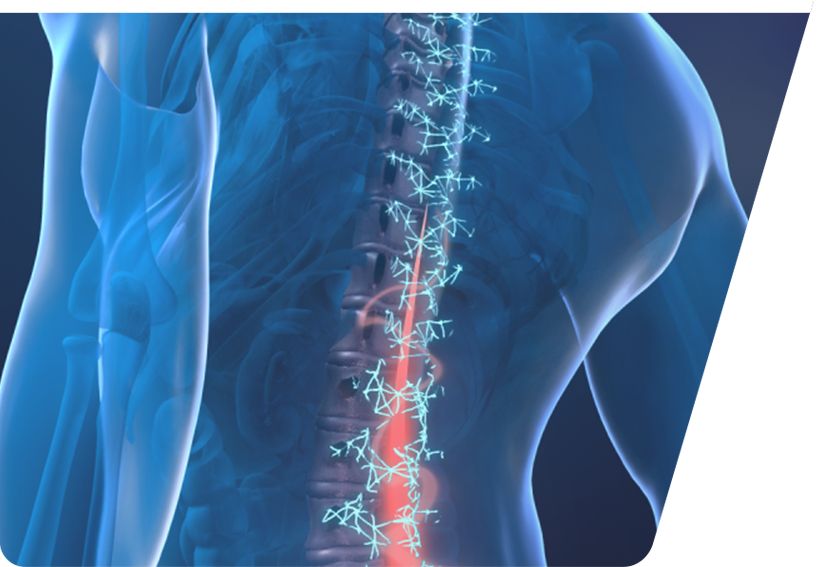
Lumbar puncture (PL)
| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|

Lumbar puncture (PL)
i

It is recommended:
-
Lie down in a comfortable position (usually on your back)
-
Rehydration
-
Analgesics
-
Anti-emetics or opioids
4
Generally, more than 85% of headaches caused by a lumbar puncture disappear without using any treatment.
4
Headache after lumbar puncture usually appears after 24-48 hours of the procedure.
4
| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
Common procedure.
2
Important diagnostic tool in Neurology.
2
Direct administration of drugs in the Central Nervous System.
3
2
Proven security.

| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
Common procedure.
2
Important diagnostic tool in Neurology.
2
Direct administration of drugs in the Central Nervous System.
3
2
Proven security.
Lumbar puncture (PL)
Management of headache associated with lumbar puncture
i
Discovering the origin of spinal muscular atrophy
i

| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|

5

| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
Discovering the origin of spinal muscular atrophy
i
Mejora de la unidad motora tras los primeros meses
Continued increase in the number of functional motor units (>18 months) without reaching a plateau phase.
6
6
+Protein SMN
SMA is a genetic disease caused by a reduction of the SMN protein in the motor neurons of the CNS5
5

Improvement of the motor unit after the first months.
Continued increase in the number of functional motor units (>18 months) without reaching a plateau phase.
6
6

Apply MusclEase

| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
+Protein SMN
5
SMA is a genetic disease caused by a reduction in SMN protein in the motor neurons of the CNS5.
5

| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
MUSCLEASE treatment demonstrated significant improvements in motor function
*1,8-10
Regardless of outpatient status.
Independently of the age.
1
1
In all patients with SMA type II and III.
7
* (change in mean aggregate=+2.27 (95% CI 1.41-3.13) vs -1 (95% CI -1.33;-0.67) p<0.0001).
1

MUSCLEASE treatment demonstrated significant improvements in motor function
*1,8-10
Regardless of outpatient status
Regardless of age.
1
1
In all patients with SMA type II and III
7
| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
* (change in mean aggregate=+2.27 (95% CI 1.41-3.13) vs -1 (95% CI -1.33;-0.67) p<0.0001).
1

No new serious adverse effects related to the study drug, with a positive and manageable safety profile that remains consistent in real-world studies
9,12
| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
*70.9% of the patients would recommend Musclease.
11

Only treatment for SMA that does not require additional follow up.
MUSCLEASE's long half-life supports low dosing frequency: 3 injections per year in maintenance
Most patients experienced relief from symptoms and motor deficits. Satisfaction with the treatment was high and drug recommendation rates were excellent.
5
*11
| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
*NPS values greater than 0 are classified as a positive rating, while a total NPS score of >50 is considered excellent. 95.7% of patients were satisfied with treatment with MusclEase overall, 88.8% of patients were satisfied with the relief of symptoms produced by MusclEase, and 70.9% of patients would recommend MusclEase.
11


References
| AME | PL / Headache | Central Nervous System | Treatment | Safety | Benefits |
|---|
- Coratti G, et al. Motor function in type 2 and 3 SMA patients treated with Nusinersen: a critical review and meta‑analysis. Orphanet J Rare Dis. 2021;16(1):430.
- Engelborghs S, et al. Consensus guidelines for lumbar puncture in patients with neurological diseases. Alzheimers Dement (Amst). 2017;8(1):111–26
- Johannsen J, et al. Intrathecal administration of nusinersen in pediatric SMA patients with and without spine deformities: Experiences and challenges over 3 years in a single center. Neuropediatrics. 2021;52(3):179–85
- Ahmed SV, et al. Post lumbar puncture headache: diagnosis and management. Postgrad Med J. 2006;82(973):713–6.
- Kariyawasam D, et al. Motor unit changes in children with symptomatic spinal muscular atrophy treated with nusinersen. Neurol Neurosurg Psychiatry 2020;92(1):78-85.
- Lusakowska A, et al. Nusinersen in a wide severity spectrum of SMA in long-term observation – real Word experiences. Presented at EAN, Vienna 2022
- Walter MC, et al. Safety and Treatment Effects of Nusinersen in Longstanding Adult 5q-SMA Type 3 - A Prospective Observational Study. J Neuromuscul Dis. 2019;6:453-65.
- Hagenacker T, et al. Nusinersen in adults with 5q spinal muscular atrophy: a non-interventional, multicentre, observational cohort study. Lancet Neurol. 2020;19(4):317-25.
- Osmanovic A, et al. Treatment expectations and patient-reported outcomes of nusinersen therapy in adult spinal muscular atrophy. J Neurol. 2020;267(8):2398-407.
- Meyer T, et al. Treatment expectations and perception of therapy in adult patients with spinal muscular atrophy receiving nusinersen. Eur J Neurol. 2021;28(8):2582-2595.
- Maggi L, et al. Nusinersen safety and effects on motor function in adult spinal muscular atrophy type 2 and 3. J Neurol Neurosurg Psychiatry. 2020;91(11):1166-74.