gWear FU
22 October 2015
Table of
Contents
Lessons learned
Follow-up
Devices
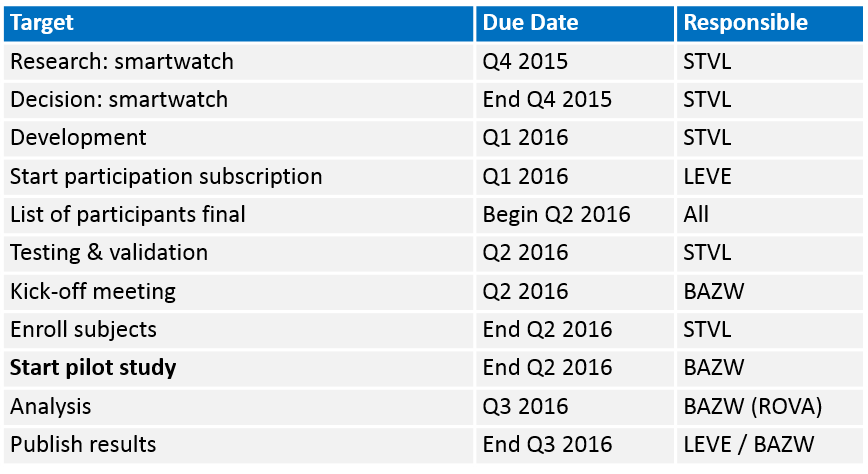
Timelines
Discussion
Lessons learned
User experience
- Improve connectivity
- Omit smart phone in the process
- Variations in questions / topics

Study experience
- Licensing 'Quality of Life' questionnaire
- Study or not (Regulatory aspect)
- More extensive testing needed
Follow-up
FU study III:
Commercial driven

FU study II:
Real-life experience
INTEGR8:
2-way communication
FU study II
Patient population
- Internal & external patients
- Broaden age range
- Global
- Multi-language
Study design
- Data entry / answer questions
- Extended functionality (metrics)
- Provide support community
- Data correlation
- edc2go (vs. e-capture)

FU study II
Study names
- Fusion
- Mesh
- Unify
- Ittaika
- Ikkan

Approach
Define study
- Device
- Protocol
- CRF
- Consent Form
- Participants
- Sign-off
Development
- Mockups
- Engineering
- Testing
- Validation
Communication
- Internal
- External
Study conduct
Publication
Potential devices

Potential devices

Potential devices

Potential devices

Estimated timelines