Atoms,Compounds
by rekha abdul
Atoms
Everything is made from atoms, including you. Atoms are tiny particles that are far too small to see, even with a microscope. If people were the same size as atoms, the entire population of the world would fit into a box about a thousandth of a millimetre across!
We usually imagine atoms as being like tiny balls:
To make diagrams simpler we often draw atoms as circles:.
Elements
There are over a hundred different types of atom, and these are called elements. Each element has a special name. For example carbon, oxygen and hydrogen are all elements.
Lead and gold are elements too. A piece of pure gold contains only gold atoms. A piece of pure lead contains only lead atoms.
Chemical reactions join or split atoms to rearrange them. But they cannot change one element into another element, or anything simpler. A chemical reaction cannot turn lead into gold, becuse it can't change the atoms into different elements.
Chemical symbols
Each element is given its own chemical symbol, like O for oxygen and Cl for chlorine. Chemical symbols are usually one or two letters long, but sometimes three letters are used.
Every chemical symbol starts with a capital letter, with the second or third letters written in lower case.
For example, Mg is the correct symbol for magnesium, but mg, mG and MG are wrong.
Take care to write chemical symbols correctly
Mg mg mg MG
Symbols and names
Sometimes it is easy to tell which element a symbol stands for. For example, O.s for oxygen and Li stands for lithium.
But sometimes the symbol comes from a name for the element that is not an English word.For example, W stands for tungsten (from the word wolfram) andNa stands for sodium (from the word natrium).
The reason is that the same chemical symbols are used all over the world, no matter which language is spoken, which makes them most useful.
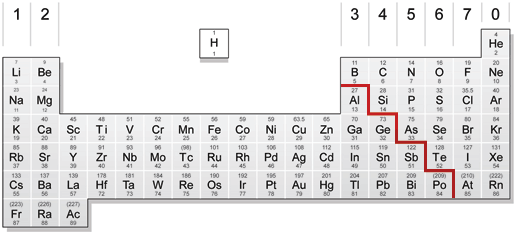
The periodic table
All the different elements are arranged in a chart called the periodic table.
-
The horizontal rows are called periods.
-
The vertical columns are called groups.
-
Elements in the same group are similar to each other.
-
The metals are on the left and the non-metals are on the right.
-
One non-metal, hydrogen, is often put in the middle.
-
The main groups are numbered from 1 to 7 going from left to right, and the last group on the right is group 0.
The zig-zag line in this diagram separates the metals, on the left, from non-metals, on the right. Hydrogen is a non-metal but it is often put in the middle.
Notice that most elements are metals, rather than non-metals.
Each element has its own chemical symbol, made from letters. Remember that you will only find elements in the periodic table and never compounds. So don't try to look for substances like water and copper sulphate in the periodic table, because they are not there.
Metals
Iron, magnesium and gold are examples of metal elements.
All metals have these properties in common:
-
They are shiny, especially when they are freshly cut.
-
They are good conductors of both heat and electricity.
-
They can be bent without breaking (they are malleable).
Most metals also have these properties:
-
they are solid at room temperature, except mercury, which is liquid at room temperature
-
they are hard and strong
-
they have a high density (they feel heavy for their size)
-
they make a ringing sound when they are hit (they are sonorous)
Mercury is the only metal that is liquid at standard room temperature and pressure
Mercury is the only liquid metal at room temperature.
Three metals are magnetic.
These are iron, cobalt and nickel. Steel is a mixture of elements but mostly iron, so it is also magnetic. The other metals are not magnetic.

Uses of metals
You may have to match the use of a metal with a property that makes it suitable for that use. For example, copper is used for electrical wiring because it is a good conductor of electricity, not because it is a good conductor of heat.
Non-metals
Oxygen, carbon, sulphur and chlorine are examples of non-metal elements.
All non-metals have these properties in common:
-
they are dull (not shiny)
-
they are poor conductors of heat and electricity (they are insulators)
-
they are weak and brittle (they easily break or shatter)
Most non-metals have these properties:
-
they have a low density (they feel light for their size)
-
they do not make a ringing sound when they are hit (they are notsonourous)
Eleven non-metals are gases at room temperature, including oxygen and chlorine.
One non-metal, bromine, is a liquid at room temperature.
The other non-metals are solids at room temperature, including carbon and sulphur.
Curious carbon
Carbon is a solid non-metal element. Pure carbon can exist in two very different forms - diamond and graphite. The table shows some differences between them.
Diamond Graphite
transparent and colour less opaque and black
hard soft
Diamonds are used in jewellery
Diamond is the hardest natural substance on Earth, but it is also very brittle and will shatter if hit with a hammer.
Graphite is unusual because it is a non-metal that conducts electricity.
Diamonds are used in jewellery

An atom is the smallest unit of matter that defines the chemical elements. Every solid, liquid, gas, and plasma is made up of neutral or ionized atoms. Atoms are very small: the size of atoms is measured in picometers – trillionths (10−12) of a meter.
Every atom is composed of a nucleus made of one or more protons and usually an equal or similar number of neutrons (except hydrogen-1, which has no neutrons). Protons and neutrons together are called nucleons. The nucleus is surrounded by one or more electrons. Over 99.94% of the atom's mass is in the nucleus.The protons have a positive electric charge, the electrons have a negative electric charge, and the neutrons have no electric charge. If the number of protons and electrons are equal, that atom is electrically neutral. If an atom has a surplus or deficit of electrons relative to protons, then it has an overall positive or negative charge, and is called an ion.
what does means atoms?