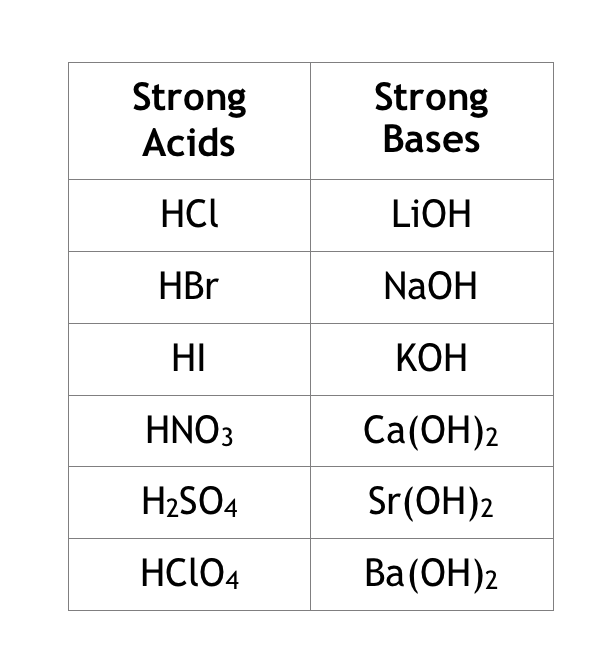
Acids and Bases
Hydrogen fluoride
Hydrochloric acid
Ammonium ion
Acetic acid
Sulphuric acid
Sodium hydroxide
Potassium hydroxide
Acetate ion
Ammonia
Ammonium Hydroxide
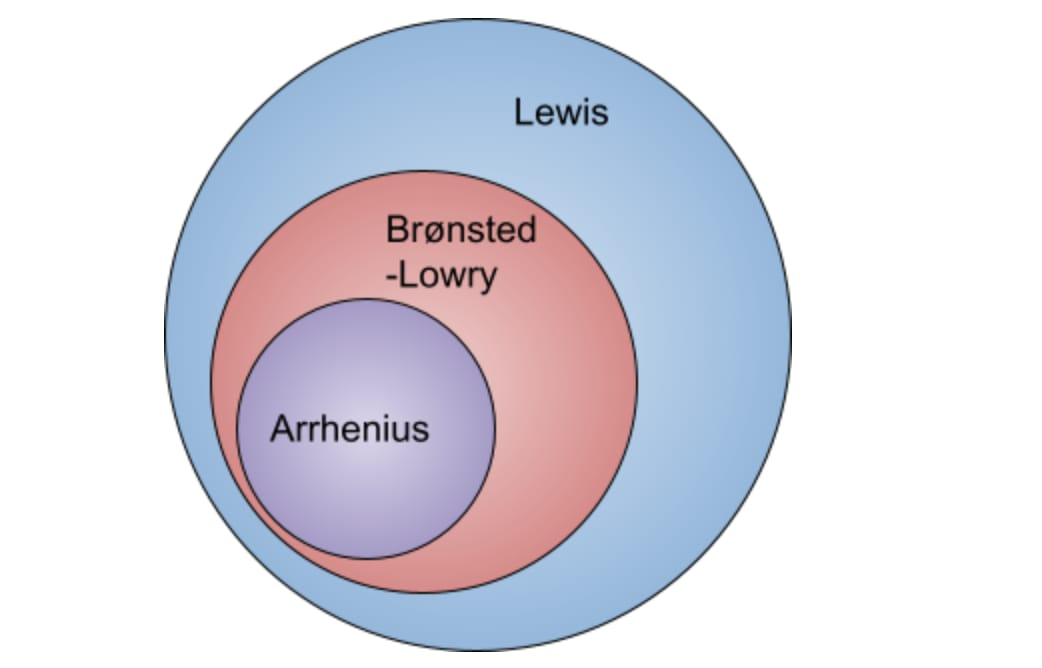
Types of acids & bases
Arrhenius definition
acids are hydrogen-containing compounds that dissociate in water to give H+ ions or protons, while bases are hydroxide compounds that dissociate to give OH– ions
H+(aq) + OH-(aq) ⟶ H2O(l)
Li(aq) + F-(aq) ⟶ LiF(aq)
HF(aq) + LiOH(aq) ⟶ H2O(l) + LiF(aq)
Arrhenius acid increases H+ conc
HF(aq) ⇌ H+(aq) + F-(aq)
LiOH(aq) ⟶ Li+(aq) + OH-(aq)
Arrhenius base increases OH- conc
Neutralisation reaction
Examples:
Arrhenius acids
| Formula | Name |
|---|---|
| CH3COOH | acetic acid |
| HClO3 | chloric acid |
| HCl | hydrochloric acid |
| HBr | hydrobromic acid |
| HI | hydriodic acid |
| HF | hydrofluoric acid |
| HNO3 | nitric acid |
| H2C2O4 | oxalic acid |
| HClO4 | perchloric acid |
| H3PO4 | phosphoric acid |
| H2SO4 | sulfuric acid |
| H2SO3 | sulfurous acid |
| HClO | Hypochlorous acid |
Arrhenius bases
| Formula | Name |
|---|---|
| LiOH | lithium hydroxide |
| NaOH | sodium hydroxide |
| KOH | potassium hydroxide |
| RbOH | rubidium hydroxide |
| CsOH | caesium hydroxide |
| Ca(OH)2 | calcium hydroxide |
| Sr(OH)2 | strontium hydroxide |
| Ba(OH)2 | barium hydroxide |
| Co(OH)2 | cobalt hydroxide |
| Fe(OH)2 | iron (II) hydroxide |
| Tl(OH)3 | thallium(III) hydroxide |
Brønsted–Lowry definition
An acid is a species that donates a proton, while a base is one that accepts it
In this reaction, HCl donates a proton to NH3 and hence they act as Brønsted–Lowry acid and base respectively. According to Arrhenius theory, the above reaction would not be an acid-base reaction because neither species is forming H+ or OH- in water, but the chemistry involved is very similar to what would occur in aqueous phase
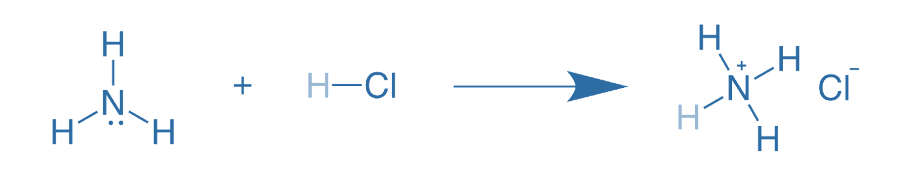
Brønsted–Lowry acids & bases
| Acid | Formula | Conjugate Base | Formula |
|---|---|---|---|
| Perchloric acid | HClO4 | Perchlorate ion | ClO4- |
| Sulphuric acid | H2SO4 | Hydrogen sulfate ion | HSO4- |
| Nitric acid | HNO3 | Nitrate ion | NO3- |
| Hydronium ion | H3O+ | Water | H2O |
| Phosphoric acid | H3PO4 | Dihydrogen phosphate | H2PO4- |
| Acetic acid | CH3COOH | Acetate ion | CH3COO- |
| Carbonic acid | H2CO3 | Hydrogen carbonate | HCO3- |
| Hydrochloric acid | HCl | Chloride ion | Cl- |
| Sulfuric acid | H2SO4 | Sulphate ion | [SO4](2-) |
| Hydrogen sulfite | HSO3- | Sulfite ion | [SO3](2-) |
| Bicarbonate ion | HCO3- | Carbonate ion | [CO3](2-) |
| Bisulfide ion | HS- | Hydroxide ion | OH- |
Brønsted–Lowry acids & bases
| Acid | Formula | Conjugate Base | Formula |
|---|---|---|---|
| Hydrogen sulfide | H2S | Hydro sulfide ion | HS- |
| Ammonium ion | NH4+ | Ammonia | NH3 |
| Hydrogen cyanide | HCN | Cyanide ion | CN- |
| Phenol | C6H5OH | Phenoxide ion | C6H5O- |
| Water | H2O | Hydroxide ion | OH- |
| Ammonia | NH3 | Amide ion | NH2- |
| Hydrogen | H2 | Hydride ion | H- |
| Methane | CH4 | Methide ion | CH3- |
Lewis definition
A Lewis acid accepts an electron pair and Lewis base donates an electron pair.
The Lewis definition focuses on the movement of electrons rather than protons. AlCl3 and BCl3 are two examples of species that come under Lewis acids and none of the others, as they do not have protons to donate in a reaction.
BF3 + NH3 ⟶ BF3NH3
Lewis acid
Lewis base
Acid-base adduct
Lewis acids & bases
Acids:
- Boron trihalides (ex BF3, BCl3)
- Organoboranes (ex B(OCH3)3 [trimethyl borate])
- RMgX (C6H5-Mg-Br, [phenylmagnesium bromide])
- AlX3 (ex AlCl3, AlH3, AlF3)
Bases:
- X- (ex F-, Br-, Cl-)
- PR3 (ex P(C6H5)3)
- Ethers (CH3–CH2–O–CH3 [methoxyethane])
- Thioethers ((CH3)2S [dimethylsulfide])
- Amines (C6H5NH2 [Aniline])
- Phosphine
- Compounds of O, S, Se and Te in oxidation state 2- (H2O)
- Simple & complex anions (H-, F-, SO2 4-)
- Lone-pair containing species (NH3, HO−, and CH3−)
- electron-rich π-system Lewis bases (ethyne, ethene, and benzene)
HSAB concept
| Property | Hard acids and bases | Soft acids and bases |
|---|---|---|
| atomic/ionic radius | small | large |
| oxidation state | high | low or zero |
| polarizability | low | high |
| electronegativity (bases) | high | low |
| HOMO energy of bases | low | higher |
| LUMO energy of acids | high | lower (but more than soft-base HOMO) |
| affinity | ionic bonding | covalent bonding |
HSAB concept - acids
| Hard Acid | Soft Acid |
|---|---|
| H+, Li+,Na+,K+ | Cu+, Ag+, Au+, Tl+, Hg+ |
| Be2+, Mg2+, Ca2+, Sr2+ | Pd2+, Cd2+, Pt2+, Hg2+, Pt4+ |
| Al3+, Sc3+, Ga3+, In3+, Ln3+, La3+ | Tl3+, BH3, GaI3, InCl3 |
| H3O+, BF3, R3C+ | I+, Br+, HO+, RO+ |
| Th4+, U4+, Si4+, Zr4+, Pu4+ | I2, Br2, ICN |
| Cr3+, Co3+, Fe3+, As3+, Ce3+ | O, Cl, Br, I, N |
| UO2 2+, VO2 2+, MoO3+ | CH2, carbenes, BH3 |
| AlCl3, Al(CH3)3, AlH3 | CH3Hg+, Hg2 2+, Pt+, Pd+ |
| I7+, I5+, Cl7+, Cr6+, | C6Cl4O2 |
| RCO+, CO2, NC+, HX | Au+ |
HSAB concept - bases
| Hard Base | Soft Base |
|---|---|
| NH3, RNH2, N2H4 | H-, R-, C3H4, C6H6 |
| H2O, OH-, O2-, ROH, RO- | CN-, RNC, CO |
| CH3COO-, CO3 2-, NO3-, CH3CF- | S2O3 2-, SCN-, R3P, RS- |
| PO4 3-, SO4 2-, ClO4- | R3As, I- |
| F-, Cl- |
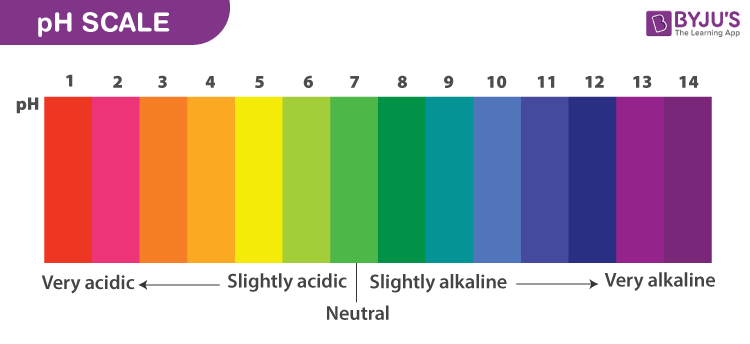
pH scale
Measure of Hydrogen ion concentrations at logarithmic scale
Hydrogen ions concentrations in solutions are often very small, but can quickly change at an exponential rate hence a logarithmic scale is useful in quantifying these changes.
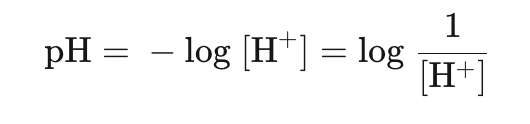
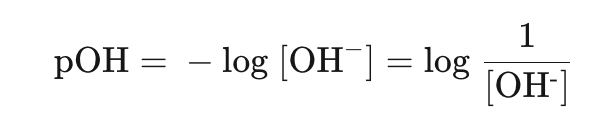
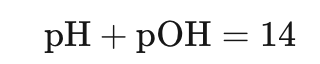
Similarly, hydroxide can be measured on a pOH scale:
Testing pH values
pH indicators are dyes that change colour at different concentrations of hydronium
Red litmus paper turns blue when exposed to a basic solution
Blue litmus paper turns red when exposed to an acidic solution
Ka, pKa, Kb, and pKb
Ka, pKa, Kb, and pKb are most helpful when predicting whether a species will donate or accept protons at a specific pH value.
Unlike pH, which depends on hydronium ion concentration, these are constant for a given acid or base
Ka = [H+][A-]/[HA]
pKa = - log Ka
Kb = [HA][OH-]/[A-]
pKb = -log Kb
Ka * Kb = Kw
Acid properties
Base properties
Acid vs Bases
1. Tastes sour
2. Turns blue litmus red
3. ph < 7
4. Can conduct electricity in solutions
5. React with active metals to produce H2
6. Arrhenius acids release H+ ions in solution
7. Brønsted–Lowry acids are H+ donors
8. Lewis acids are e- pair acceptors
1. Tastes bitter and feels slippery
2. Turns red litmus blue
3. ph > 7
4. Can conduct electricity in solutions
5.
6. Arrhenius bases release OH- ions in solution
7. Brønsted–Lowry bases are H+ acceptors
8. Lewis bases are e- pair donors
| Formula | Acid Name |
|---|---|
| H2SO3 | Sulfurous Acid |
| H2SO4 | Sulfuric Acid |
| H2SO2 | Hyposulfurous Acid |
| H2SO5 | Persulfuric Acid |
| H2S2O7 | Pyrosulfuric Acid |
| H2S2O5 | Disulfurous Acid |
| H2S2O4 | Dithionous Acid |
| H2S4O6 | Tetrathionic Acid |
| H2S2O2 | Thiosulfurous Acid |
| H2S | Hydrosulfuric Acid |
| H2S2O8 | Peroxydisulfuric Acid |
| HClO4 | Perchloric Acid |
| HCl | Hydrochloric Acid |
| HClO | Hypochlorous Acid |
| HClO2 | Chlorous Acid |
| HClO3 | Chloric Acid |
| HNO | Hyponitrous Acid |
| HNO2 | Nitrous Acid |
| HNO3 | Nitric Acid |
List of Inorganic Acids
| Formula | Acid Name |
|---|---|
| HNO4 | Pernitric Acid |
| H2CO2 | Carbonous Acid |
| H2CO3 | Carbonic Acid |
| H2CO3 | Carbonic Acid |
| H2CO | Hypocarbonous Acid |
| H2CO4 | Percarbonic Acid |
| H2C2O4 | Oxalic Acid |
| CH3COOH | Acetic Acid |
| H3PO4 | Phosphoric Acid |
| H3PO3 | Phosphorous Acid |
| H3PO2 | Hypophosphous Acid |
| H3PO5 | Perphosphoric Acid |
| H4P2O6 | Hypophosphoric Acid |
| H4P2O7 | Pyrophosphoric Acid |
| H3P | Hydrophosphoric Acid |
| HBr | Hydrobromic Acid |
| HBrO2 | Bromous Acid |
| HBrO3 | Bromic Acid |
| HBrO | Hypobromous Acid |
| Formula | Acid Name |
|---|---|
| HIO | Hypoiodous Acid |
| HIO2 | Iodous Acid |
| HIO3 | Iodic Acid |
| HIO4 | Periodic Acid |
| HI | Hydroiodic Acid |
| HFO2 | Fluorous Acid |
| HFO3 | Fluoric Acid |
| HFO | Hypofluorous Acid |
| HFO4 | Perfluoric Acid |
| HF | Hydrofluoric Acid |
| H2CrO4 | Chromic Acid |
| H2CrO3 | Chromous Acid |
| H2CrO2 | Hypochromous Acid |
| H2CrO5 | Perchromic Acid |
| H2Se | Hydroselenic Acid |
| H2SeO4 | Selenic Acid |
| Formula | Acid Name |
|---|---|
| H2SiF6 | Silicofluoric Acid |
| H6TeO6 | Telluric Acid |
| H2TeO3 | Tellurous Acid |
| H2WO4 | Tungstic Acid |
| H2XeO4 | Xenic Acid |
| C6H8O7 | Citric Acid |
| HCOOH | Formic Acid |
| H4Sb2O7 | Pyroantimonic Acid |
| HMnO4 | Permanganic Acid |
| H2MnO4 | Manganic Acid |
| HSbO3 | Antimonic Acid |
| H3SbO3 | Antimonous Acid |
| H2SiO3 | Silicic Acid |
| H2TiO3 | Titanic Acid |
| H3AsO4 | Arsenic Acid |
| HTcO4 | Pertechnetic Acid |
| Formula | Acid Name |
|---|---|
| H2SnO3 | Metastannic Acid |
| H2C2O2 | Hypooxalous Acid |
| H3[F3(CN)6] | Ferricyanic Acid |
| HCNO | Cyanic Acid |
| H2SiO2 | Silicous Acid |
| HCN | Hydrocyanic Acid |
| H2MoO4 | Molybdic Acid |
| H4XeO6 | Perxenic Acid |
| H2UO4 | Uranic Acid |
| H2U2O7 | Diuranic Acid |
| HSCN | Thiocyanic Acid |
| H2SeO3 | Selenous Acid |
| HN3 | Hydronitric Acid |
| H3BO3 | Boric Acid |
| H3As | Hydroarsenic Acid |
| H2Cr2O7 | Dichromic Acid |
| H2B4O7 | Tetraboric Acid |
List of Organic Acids
| Formula | Acid Name |
|---|---|
| CH2(COOH)2 | Malonic Acid |
| H2C6H6O7 | Citric Acid |
| H2C4H4O6 | Tartartic Acid |
| HC5H8NO4 | Glutamic Acid |
| H2C8H4O4 | Phthalic Acid |
| H2C9H14O4 | Azelaic Acid |
| HC4H3N2O3 | Barbituric Acid |
| HC14H11O3 | Benzilic Acid |
| C9H8O2 | Cinnamic Acid |
| C4H4O4 | Fumaric Acid |
| C5H8O4 | Glutaric Acid |
| C6H12O7 | Gluconic Acid |
| C5H11COOH | Hexanoic Acid |
| CH3COOH | Acetic Acid |
| Formula | Acid Name |
|---|---|
| HC3H5O3 | Lactic Acid |
| H2C4H4O5 | Malic Acid |
| HC18H33O2 | Oleic Acid |
| C19H19N7O6 | Folic Acid |
| HC2COOH | Propiolic Acid |
| CH3CH2COOH | Propionic Acid |
| C19H14O3 | Rosolic Acid |
| C17H35COOH | Stearic Acid |
| C76H53O46 | Tannic Acid |
| C2HF3O2 | Trifluoroacetic Acid |
| H2C5H2N4O3 | Uric Acid |
| HC6H7O6 | Ascorbic Acid |
| HC7H5O5 | Gallic Acid |
| HC9H7O4 | Acetylsalicylic Acid |

List of Bases
| Name | Formula |
|---|---|
| La(OH)3 | Lanthanum Hydroxide |
| Pb(OH)2 | Lead(II) Hydroxide |
| Pb(OH)4 | Lead(IV) Hydroxide |
| LiOH | Lithium Hydroxide |
| Mg(OH)2 | Magnesium Hydroxide |
| Mn(OH)2 | Manganese(II) Hydroxide |
| Mn(OH)3 | Manganese(III) Hydroxide |
| Mn(OH)4 | Manganese(IV) Hydroxide |
| Mn(OH)7 | Manganese(VII) Hydroxide |
| Hg2(OH)2 | Mercury(I) Hydroxide |
| Hg(OH)2 | Mercury(II) Hydroxide |
| Mo(OH)3 | Molybdenum Hydroxide |
| Nd(OH)3 | Neodymium Hydroxide |
| NiOOH | Nickel Oxo-hydroxide |
| Ni(OH)2 | Nickel(II) Hydroxide |
| Ni(OH)3 | Nickel(III) Hydroxide |
| Nb(OH)3 | Niobium Hydroxide |
| Os(OH)4 | Osmium(IV) Hydroxide |
| Pd(OH)2 | Palladium(II) Hydroxide |
| Pd(OH)4 | Palladium(IV) Hydroxide |
| Name | Formula |
|---|---|
| Pt(OH)2 | Platinum(II) Hydroxide |
| Pt(OH)4 | Platinum(IV) Hydroxide |
| Pu(OH)4 | Plutonium(IV) Hydroxide |
| KOH | Potassium Hydroxide |
| Ra(OH)2 | Radium Hydroxide |
| RbOH | Rubidium Hydroxide |
| Ru(OH)3 | Ruthenium(III) Hydroxide |
| Sc(OH)3 | Scandium Hydroxide |
| Si(OH)4 | Silicon Hydroxide |
| AgOH | Silver Hydroxide |
| NaOH | Sodium Hydroxide |
| Sr(OH)2 | Strontium Hydroxide |
| Ta(OH)5 | Tantalum(V) Hydroxide |
| Tc(OH)2 | Technetium(II) Hydroxide |
| C4H12NOH | Tetramethylammonium Hydroxide |
| TlOH | Thallium(I) Hydroxide |
| Tl(OH)3 | Thallium(III) Hydroxide |
| Th(OH)4 | Thorium Hydroxide |
| Sn(OH)2 | Tin(II) Hydroxide |
| Sn(OH)4 | Tin(IV) Hydroxide |
| Name | Formula |
|---|---|
| Al(OH)3 | Aluminum Hydroxide |
| NH4OH | Ammonium Hydroxide |
| As(OH)3 | Arsenic Hydroxide |
| Ba(OH)2 | Barium Hydroxide |
| Be(OH)2 | Beryllium Hydroxide |
| Bi(OH)3 | Bismuth(III) Hydroxide |
| B(OH)3 | Boron Hydroxide |
| Cd(OH)2 | Cadmium Hydroxide |
| Ca(OH)2 | Calcium Hydroxide |
| Ce(OH)3 | Cerium(III) Hydroxide |
| CsOH | Cesium Hydroxide |
| Cr(OH)2 | Chromium(II) Hydroxide |
| Cr(OH)3 | Chromium(III) Hydroxide |
| Cr(OH)5 | Chromium(V) Hydroxide |
| Cr(OH)6 | Chromium(VI) Hydroxide |
| Co(OH)2 | Cobalt(II) Hydroxide |
| Co(OH)3 | Cobalt(III) Hydroxide |
| CuOH | Copper(I) Hydroxide |
| Cu(OH)2 | Copper(II) Hydroxide |
| Ga(OH)2 | Gallium(II) Hydroxide |
| Name | Formula |
|---|---|
| Ga(OH)3 | Gallium(III) Hydroxide |
| AuOH | Gold(I) Hydroxide |
| Au(OH)3 | Gold(III) Hydroxide |
| InOH | Indium(I) Hydroxide |
| In(OH)2 | Indium(II) Hydroxide |
| In(OH)3 | Indium(III) Hydroxide |
| Ir(OH)3 | Iridium(III) Hydroxide |
| Fe(OH)2 | Iron(II) Hydroxide |
| Fe(OH)3 | Iron(III) Hydroxide |
| V(OH)2 | Vanadium(II) Hydroxide |
| V(OH)3 | Vanadium(III) Hydroxide |
| V(OH)5 | Vanadium(V) Hydroxide |
| Yb(OH)3 | Ytterbium Hydroxide |
| Y(OH)3 | Yttrium Hydroxide |
| Zn(OH)2 | Zinc Hydroxide |
| Zr(OH)4 | Zirconium Hydroxide |
| Ti(OH)2 | Titanium(II) Hydroxide |
| Ti(OH)3 | Titanium(III) Hydroxide |
| Ti(OH)4 | Titanium(IV) Hydroxide |
| W(OH)2 | Tungsten(II) Hydroxide |
| (UO2)2(OH)4 | Uranyl Hydroxide |