Fibroblastic Reticular Cells in Secondary Lymphoid Organs
Mechthild Lütge
Interview faculty committee, 27.11.2024
University of Zurich
Lymph Nodes
Spleen
Peyer's patches
Secondary Lymphoid organs (SLO)
Dedicated sites where adaptive immunity is mounted to pathogens in the lymph, blood or intestine
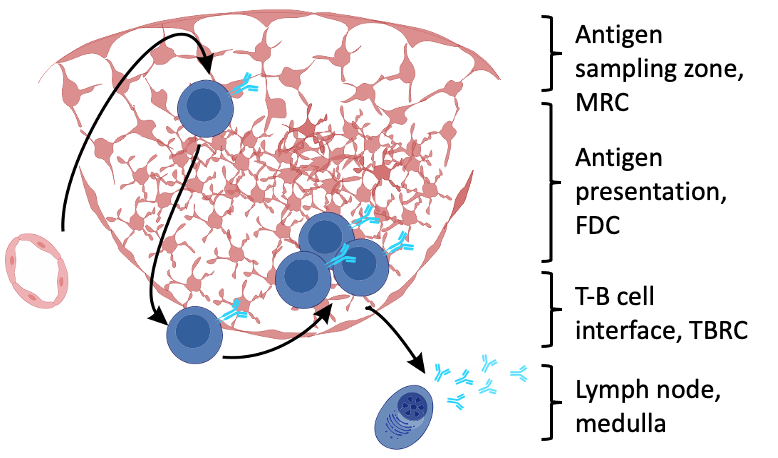
Fibroblastic reticular cells orchestrate SLO organization
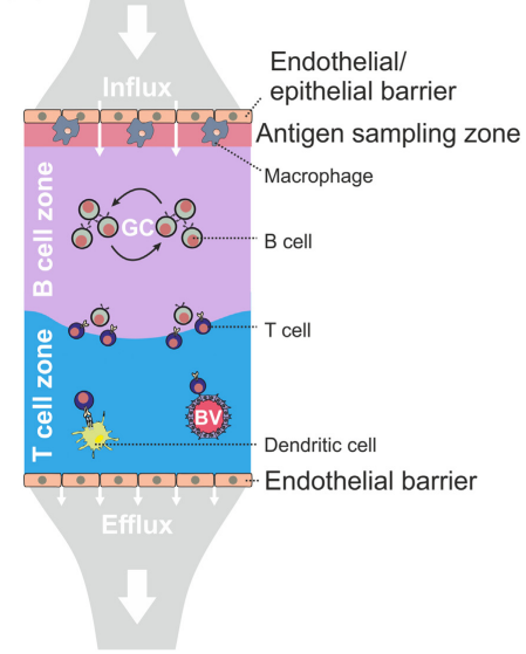
Acton et al. Trends in Immunology, 2021

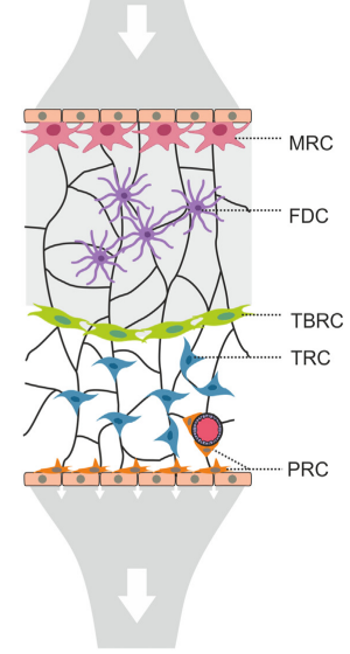
Acton et al. Trends in Immunology, 2021
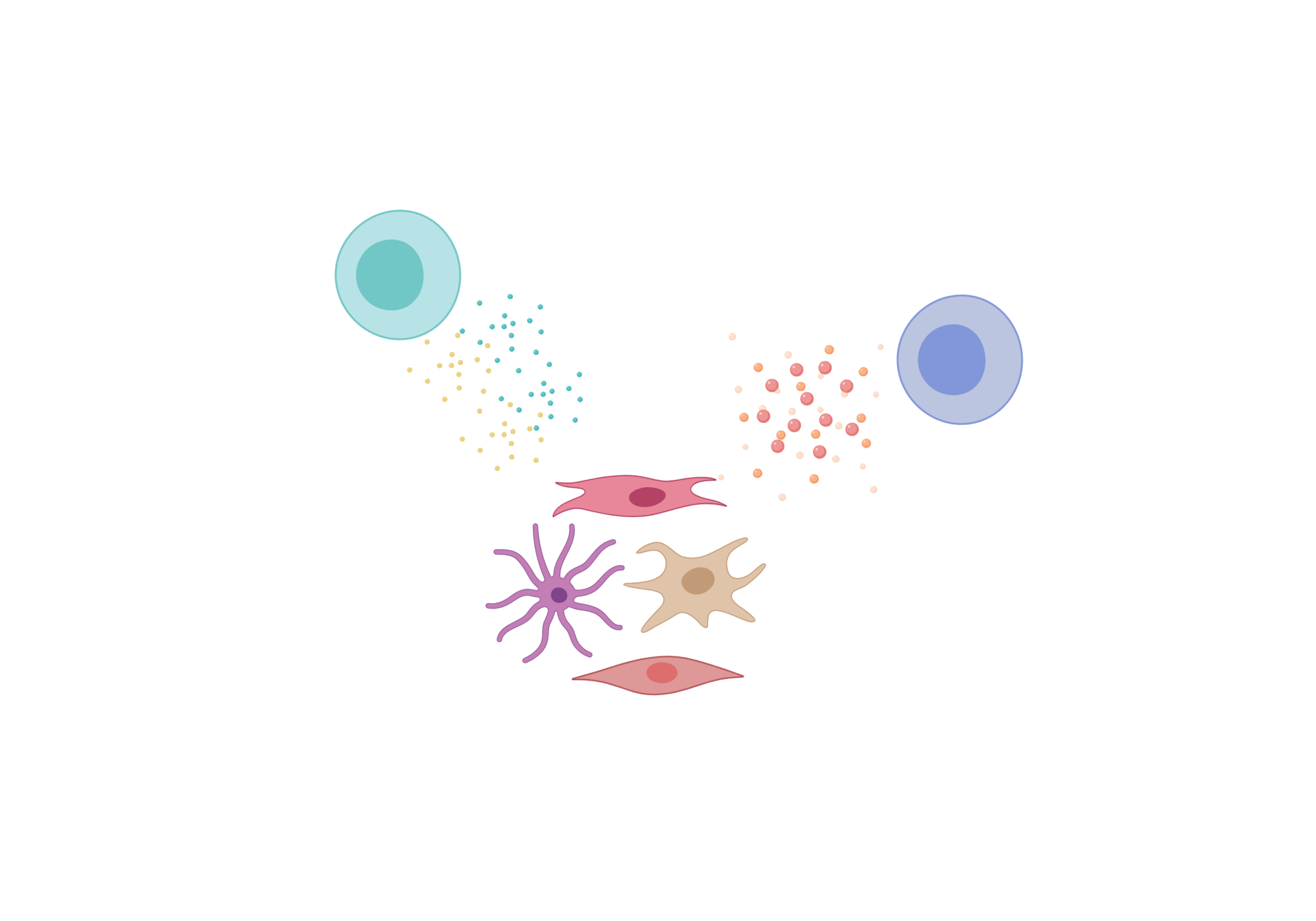
- Fibroblastic reticular cells (FRC) form specialized immune cell niches to support adaptive immune responses
- FRCs come in different flavours
Fibroblastic reticular cells orchestrate SLO organization
Fibroblastic reticular cells orchestrate SLO organization

created with biorender.com
Fibroblastic reticular cells orchestrate SLO organization

Migration
created with biorender.com
Fibroblastic reticular cells orchestrate SLO organization
Migration
Activation
and survival
created with biorender.com
Fibroblastic reticular cells orchestrate SLO organization

Migration
Activation
and survival
Extracellular matrix
created with biorender.com
Fibroblastic reticular cells orchestrate SLO organization
Migration
Activation
and survival
Extracellular matrix
Antigen presentation
and immune cell interaction
created with biorender.com
Fibroblastic reticular cells orchestrate SLO organization

Migration
Activation
and survival
Extracellular matrix
Antigen presentation
and immune cell interaction
created with biorender.com
→ Modulators of immune responses:
Strength and specificity of immune response
Fibroblastic reticular cells orchestrate SLO organization

Migration
Activation
and survival
Extracellular matrix
Antigen presentation
and immune cell interaction
created with biorender.com
→ Modulators of immune responses:
Strength and specificity of immune response
→ Prototypic immune-interacting fibroblast

Migration
Activation
and survival
Extracellular matrix
Antigen presentation
and immune cell interaction
created with biorender.com
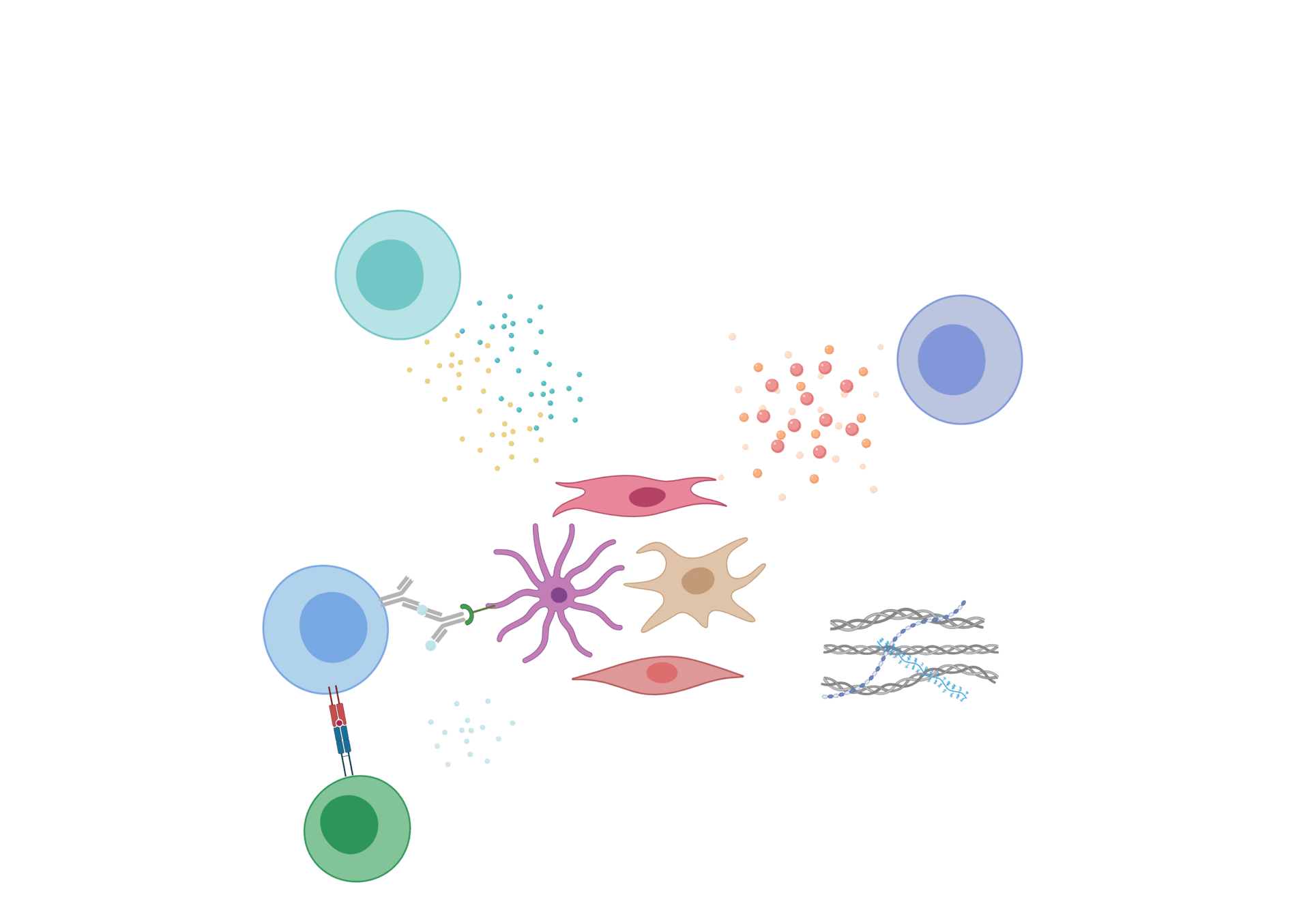
Fibroblastic reticular cells as prototypic immune-interacting fibroblasts
→ Modulators of immune responses:
- Vaccine efficacy

Migration
Activation
and survival
Extracellular matrix
Antigen presentation
and immune cell interaction
created with biorender.com
Fibroblastic reticular cells as prototypic immune-interacting fibroblasts
→ Modulators of immune responses:
- Autoimmunity and inflammatory disorders
- Vaccine efficacy

Migration
Activation
and survival
Extracellular matrix
Antigen presentation
and immune cell interaction
created with biorender.com
Fibroblastic reticular cells as prototypic immune-interacting fibroblasts
→ Modulators of immune responses:
- Autoimmunity and inflammatory disorders
- Antitumor immuntity
- Vaccine efficacy
To what extend are FRC underpinned niches functionally conserved across:

Fibroblastic reticular cells orchestrate SLO organization

To what extend are FRC underpinned niches functionally conserved across:
(1.) SLOs?
Fibroblastic reticular cells orchestrate SLO organization


To what extend are FRC underpinned niches functionally conserved across:
(1.) SLOs?
(2.) Species?
Fibroblastic reticular cells orchestrate SLO organization




To what extend are FRC underpinned niches functionally conserved across:
(1.) SLOs?
(2.) Species?
(3.) Activation?
Fibroblastic reticular cells orchestrate SLO organization




To what extend are FRC underpinned niches functionally conserved across:
(1.) SLOs?
(2.) Species?
(3.) Activation?
→ What factors shape FRC subset identity and function?
Fibroblastic reticular cells orchestrate SLO organization
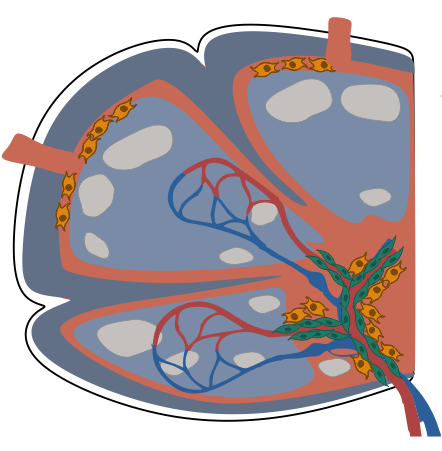
- Conserved stromal–immune cell circuits secure B cell homeostasis and function
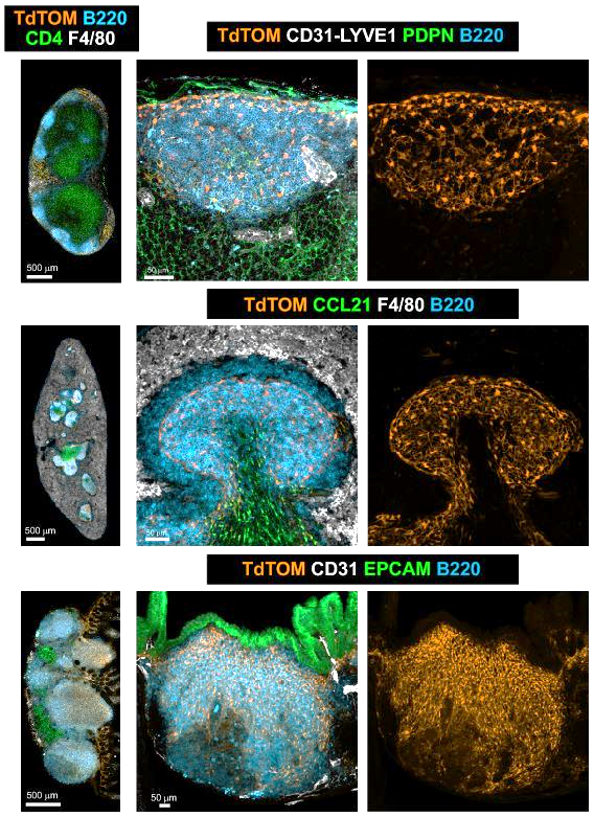
- PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Fibroblastic reticular cells orchestrate SLO organization
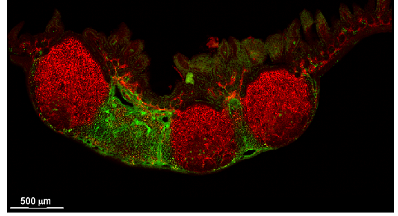
CXCL13 CCL19/CCL21
Peyer's patch
Spleen
Lymph node
CXCL13+ FRC = B cell zone reticular cells (BRCs)
Objectives



CXCL13 CCL19/CCL21
Peyer's patch
Spleen
Lymph node
CXCL13+ FRC = B cell zone reticular cells (BRCs)
Objectives
-
to what extend are BRC underpinned niches functionally conserved across SLO?



CXCL13 CCL19/CCL21
Peyer's patch
Spleen
Lymph node

CXCL13+ FRC = B cell zone reticular cells (BRCs)
Objectives
-
to what extend are BRC underpinned niches functionally conserved across SLO?
-
Systemic humoral immunity?



CXCL13 CCL19/CCL21
Peyer's patch
Spleen
Lymph node

CXCL13+ FRC = B cell zone reticular cells (BRCs)
Objectives
-
to what extend are BRC underpinned niches functionally conserved across SLO?
-
Systemic humoral immunity?
-
What are major pathways controlling BRC-immune cell interactions?



CXCL13 CCL19/CCL21
Peyer's patch
Spleen
Lymph node

CXCL13+ FRC = B cell zone reticular cells (BRCs)
Objectives
-
to what extend are BRC underpinned niches functionally conserved across SLO?
-
Systemic humoral immunity?
-
What are major pathways controlling BRC-immune cell interactions?
-
Are these interactions functionally redundant across SLOs?



CXCL13 CCL19/CCL21
Peyer's patch
Spleen
Lymph node

CXCL13+ FRC = B cell zone reticular cells (BRCs)
Objectives
Results
Lütge et al. Nat. Immunol., 2023
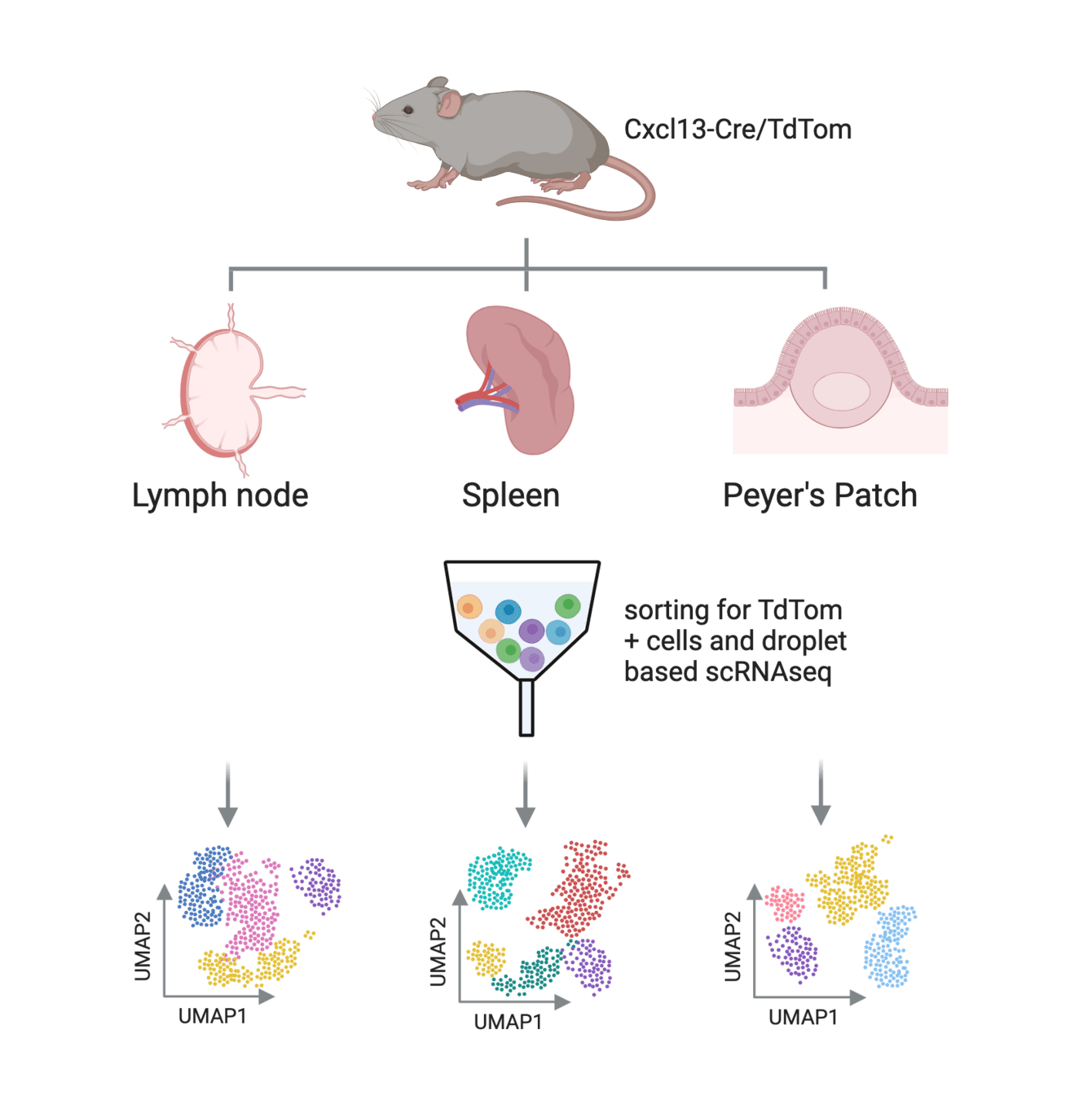
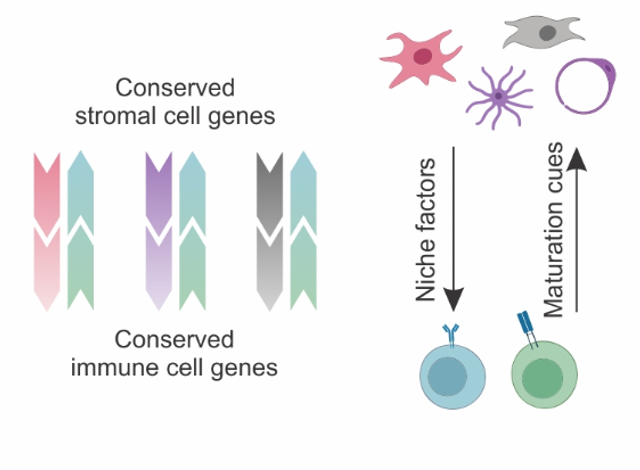
- Shared BRC subsets
- Gene expression is shaped by organ-specific imprints and subset-specific imprints (conserved across organs)
- Organ-specific imprints reflect developmental and anatomical processes
- Subset-specific imprints reflect Niche factors and signaling pathways → conserved BRC-immune cell crosstalk

Conserved across species
Summary - 1: Advanced understanding of systemic humoral immunity
Lütge et al. Nat. Immunol., 2023
Lymph node
Spleen
Peyer's patch
Organ-specific imprints
Summary - 1: Advanced understanding of systemic humoral immunity
Lütge et al. Nat. Immunol., 2023
Lymph node
Spleen
Peyer's patch
Organ-specific imprints

Functional convergence
Summary - 1: Advanced understanding of systemic humoral immunity
Lütge et al. Nat. Immunol., 2023
Lymph node
Spleen
Peyer's patch
Organ-specific imprints


Functional convergence
Feedforward paradigm: circulating immune cell imprint B cell follicle niches in an organ indiscriminate manner thereby securing efficient systemic humoral immunity
Fibroblastic reticular cells orchestrate SLO organization
- Conserved stromal–immune cell circuits secure B cell homeostasis and function


- PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Objectives
→ Characterization of the FRC landscape in a stereotypic "resting" human lymph node
Objectives
→ Characterization of the FRC landscape in a stereotypic "resting" human lymph node
→ How does inflammatory activation affect the human lymph node FRC landscape?
Objectives
→ Characterization of the FRC landscape in a stereotypic "resting" human lymph node
→ How does inflammatory activation affect the human lymph node FRC landscape?
→ Are PI16+ RCs involved in inflammatory reactions of human lymph nodes?
Objectives
→ Characterization of the FRC landscape in a stereotypic "resting" human lymph node
→ How does inflammatory activation affect the human lymph node FRC landscape?
→ Are PI16+ RCs involved in inflammatory reactions of human lymph nodes?
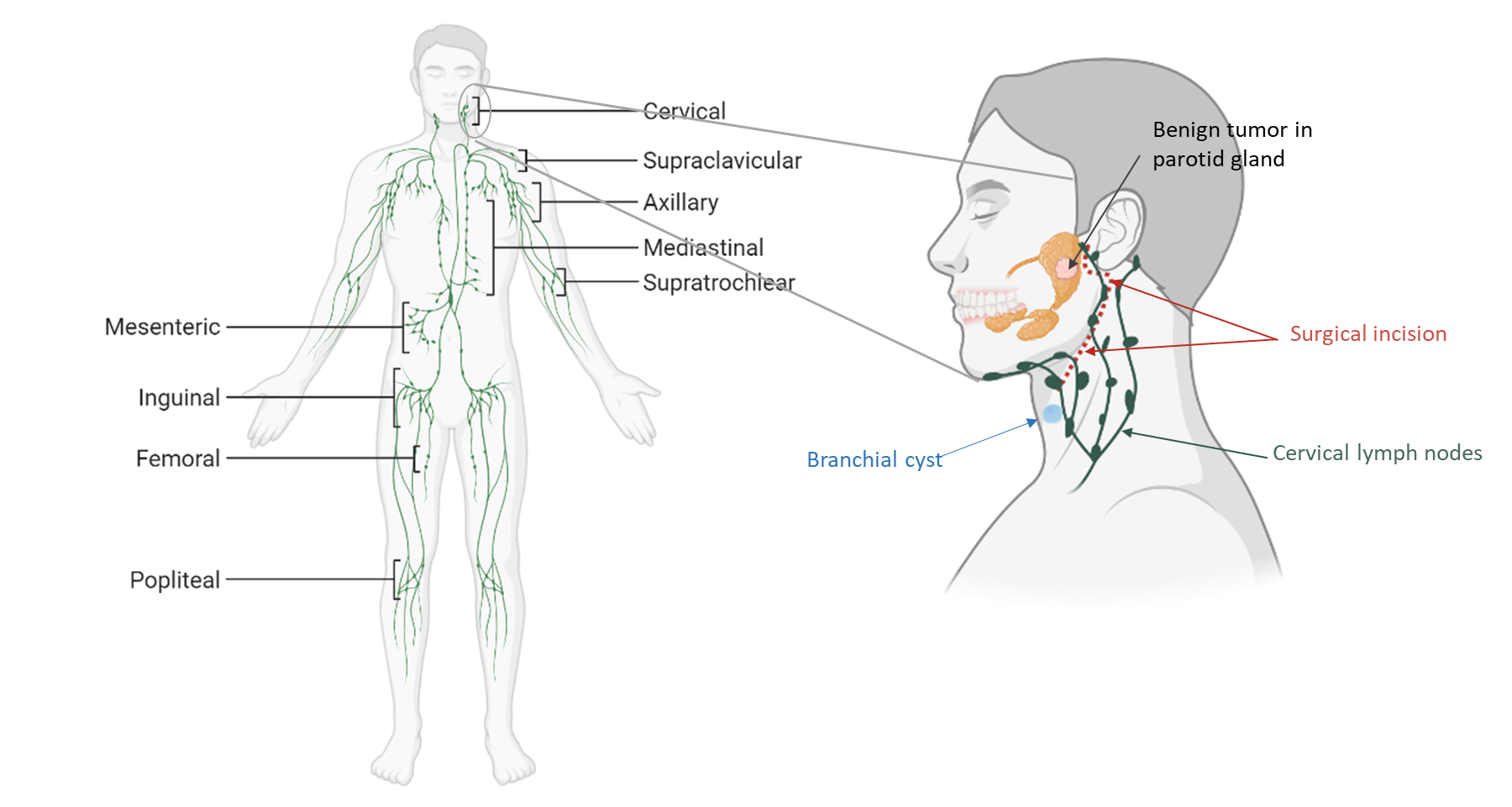
Objectives
→ Characterization of the FRC landscape in a stereotypic "resting" human lymph node
→ How does inflammatory activation affect the human lymph node FRC landscape?
→ Are PI16+ RCs involved in inflammatory reactions of human lymph nodes?

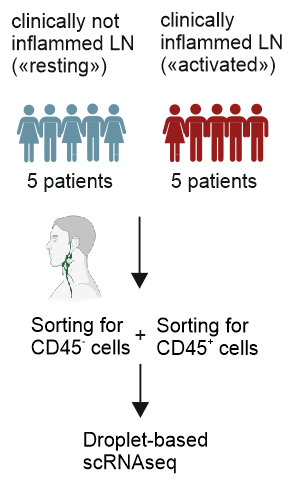
- Resting:
clinically not inflamed lymph nodes from patients with benign tumors
Objectives
→ Characterization of the FRC landscape in a stereotypic "resting" human lymph node
→ How does inflammatory activation affect the human lymph node FRC landscape?
→ Are PI16+ RCs involved in inflammatory reactions of human lymph nodes?

- Resting:
- Activated:
clinically not inflamed lymph nodes from patients with benign tumors
acute or chronically inflamed lymph nodes
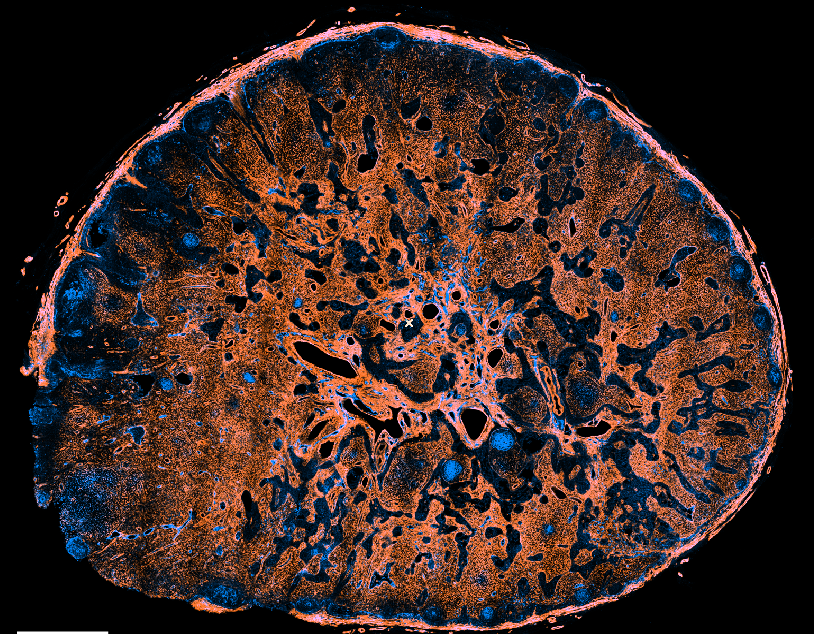
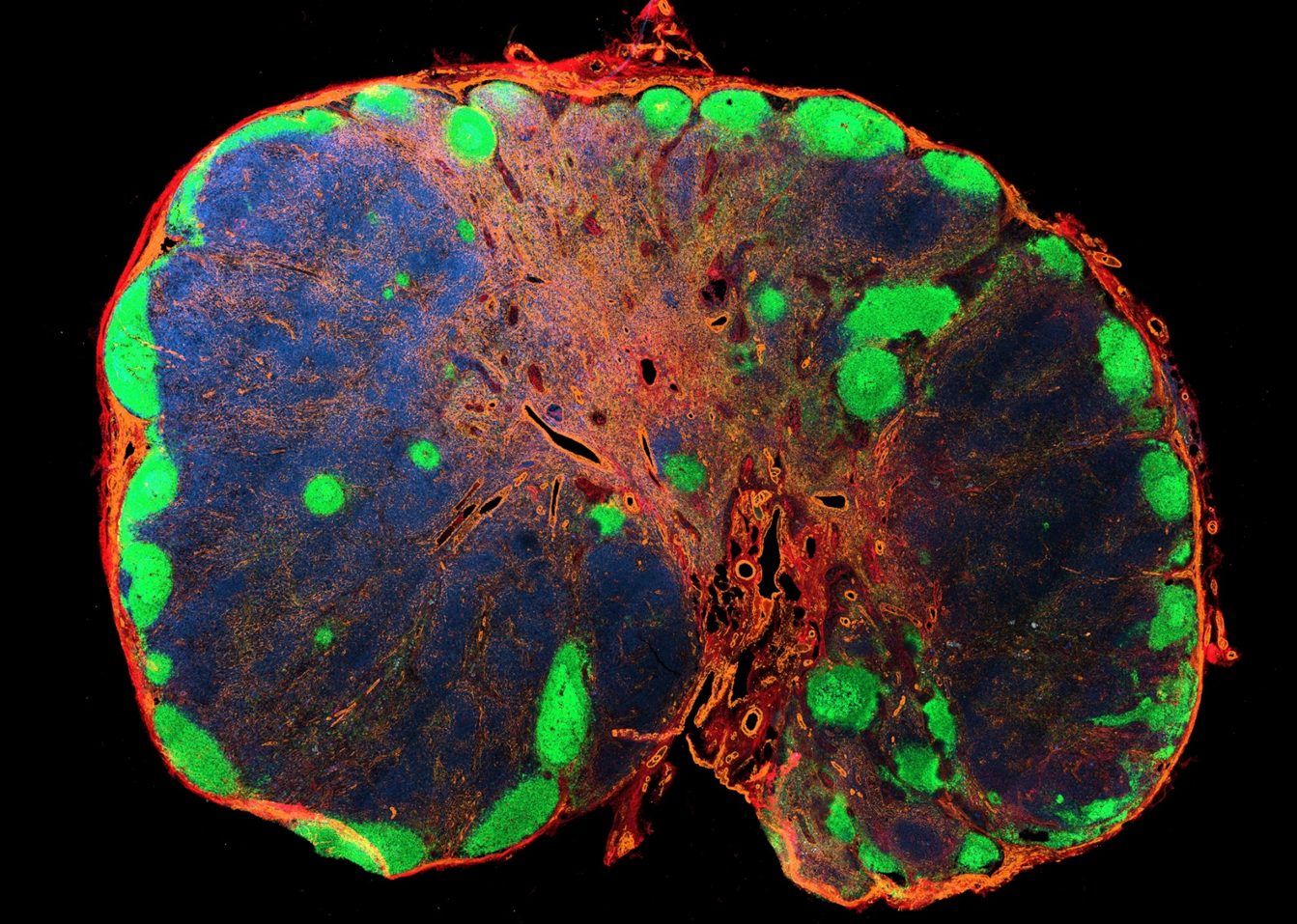
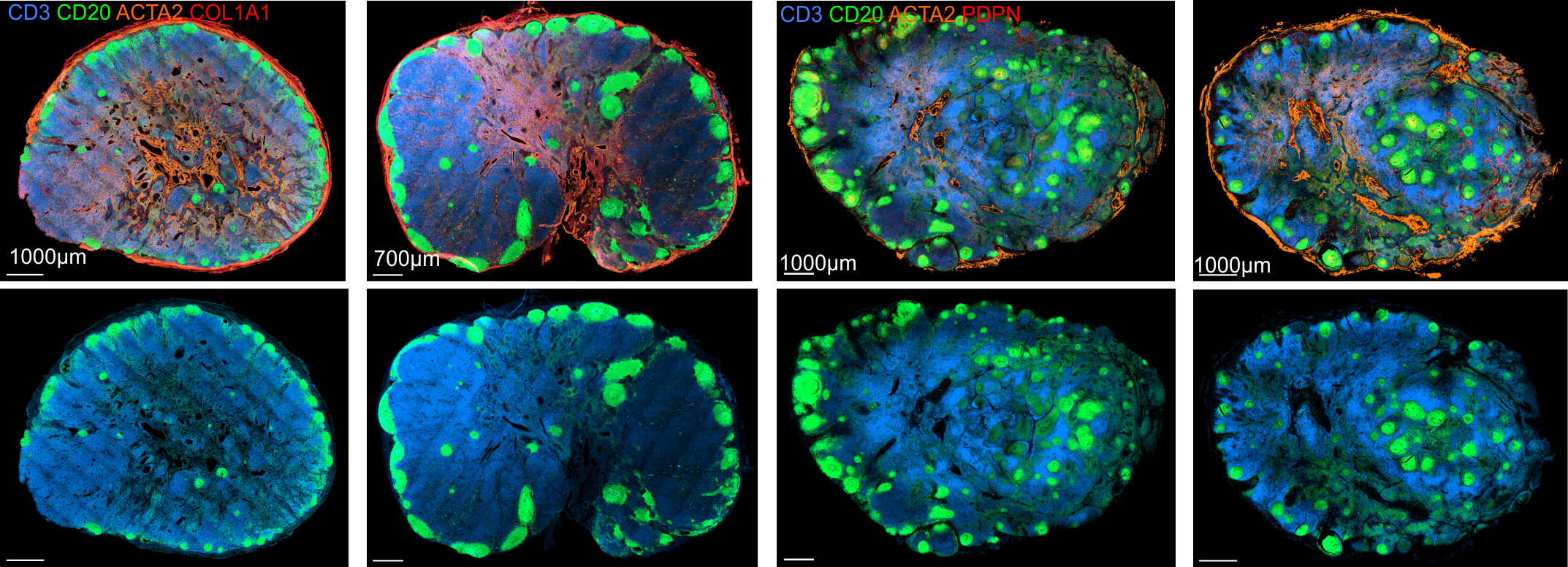
Immunoanatomy of resting human lymph nodes
CD20: B cells
CD3: T cells
ACTA2: Vascular smooth muscle cells/FRCs
-
large T cell zone
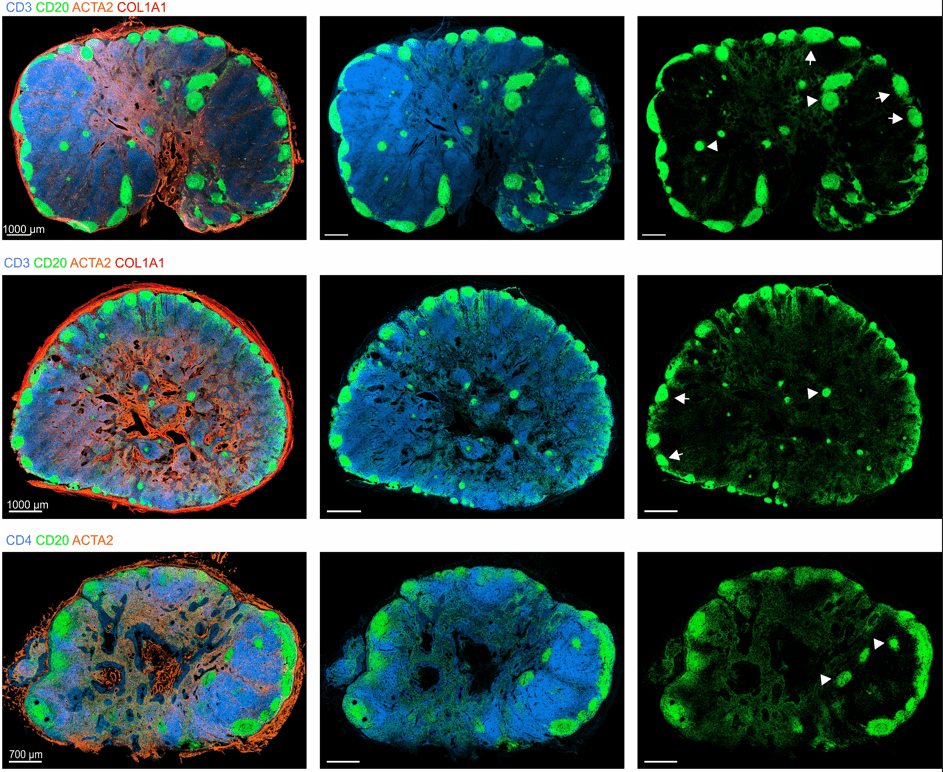
Immunoanatomy of resting human lymph nodes
CD20: B cells
CD3: T cells
ACTA2: Vascular smooth muscle cells/FRCs
-
large T cell zone
-
multiple lymph node lobules

Immunoanatomy of resting human lymph nodes
CD20: B cells
CD3: T cells
ACTA2: Vascular smooth muscle cells/FRCs
-
large T cell zone
-
multiple lymph node lobules
-
extensive vasculature with a large perivascular space

Immunoanatomy of resting human lymph nodes
CD20: B cells
CD3: T cells
ACTA2: Vascular smooth muscle cells/FRCs
-
large T cell zone
-
multiple lymph node lobules
-
extensive vasculature with a large perivascular space
-
atypical positioning of some B cell follicles

Immunoanatomy of resting human lymph nodes
CD20: B cells
CD3: T cells
ACTA2: Vascular smooth muscle cells/FRCs
-
large T cell zone
-
multiple lymph node lobules
-
extensive vasculature with a large perivascular space
-
atypical positioning of some B cell follicles
→ topological remnants of recurrent/baseline activation

Stromal cell landscape of resting human lymph nodes
LYVE1: Lymphatic endothelium
CD31: Endothelium
ACTA2: Vascular smooth muscle cells/FRCs
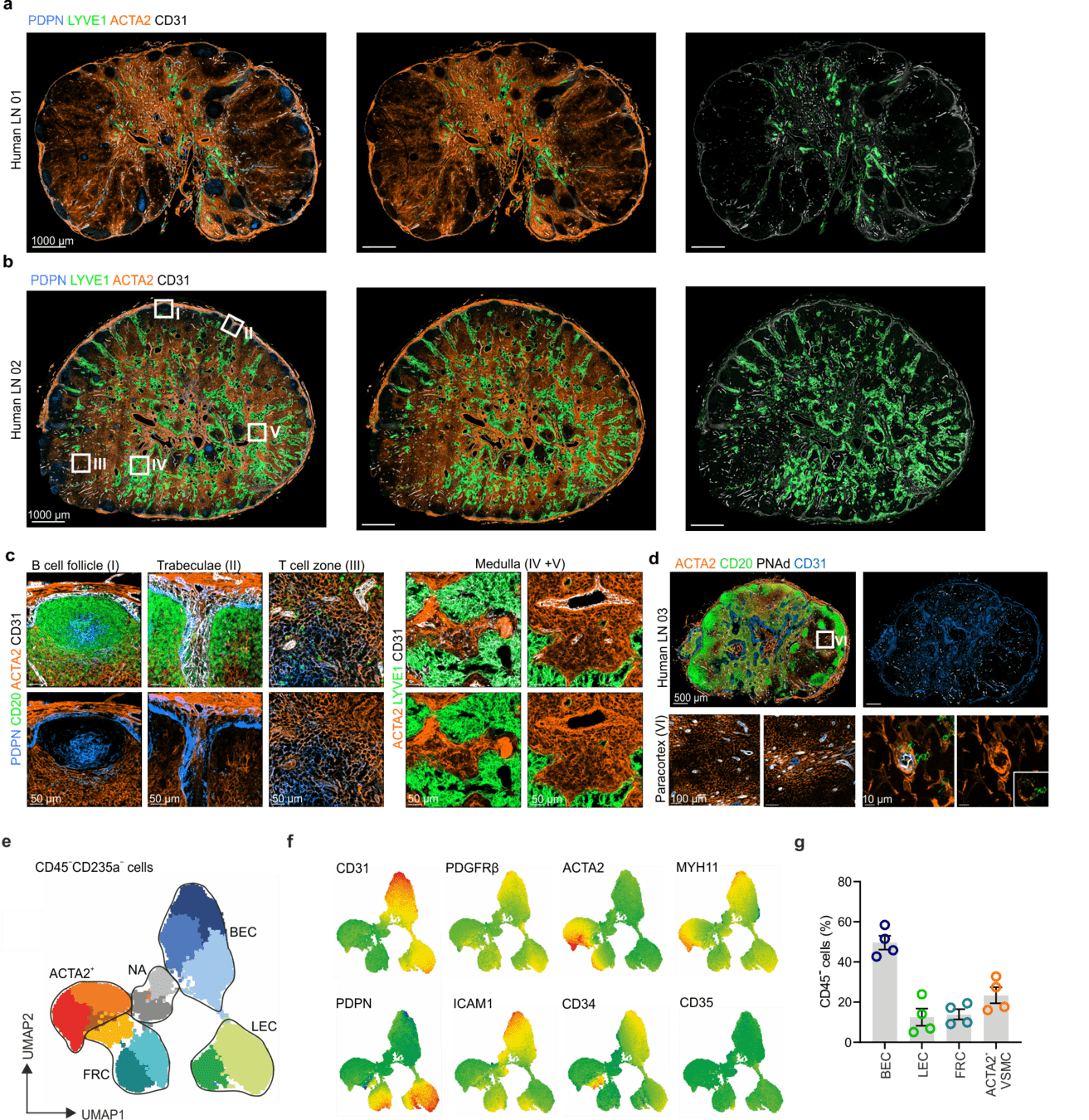
-
Extensive vasculature with a large perivascular space
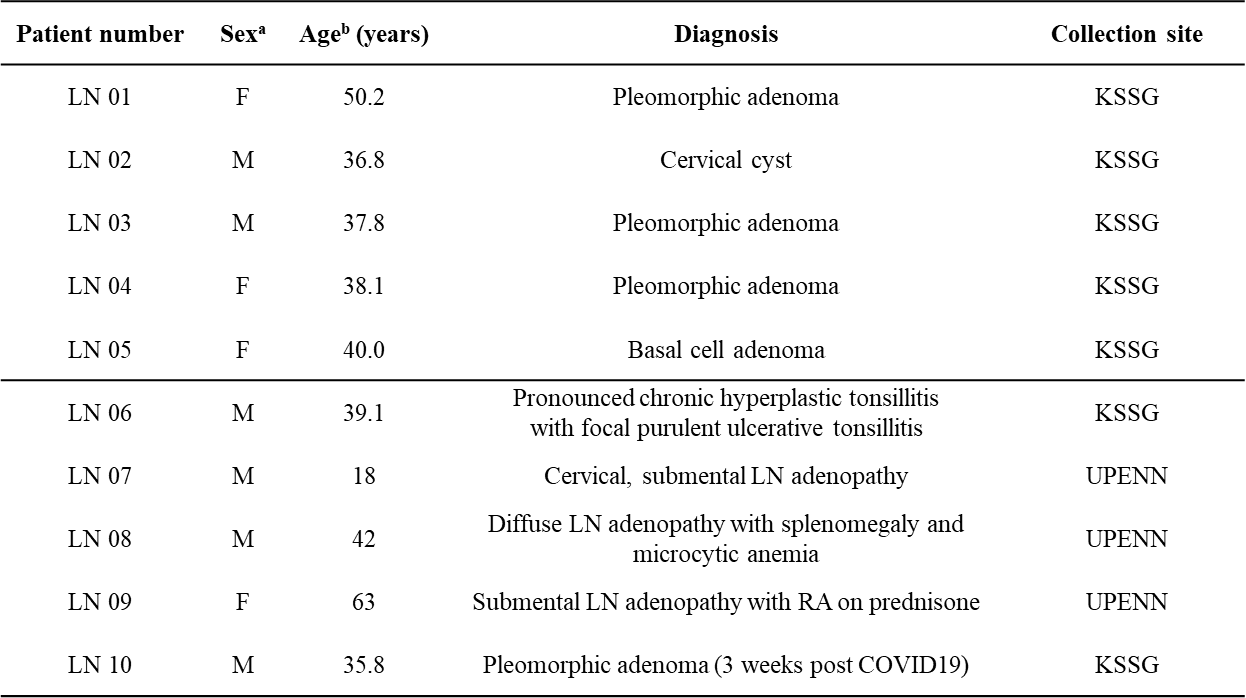
Transcriptome analyses - Patient characteristics

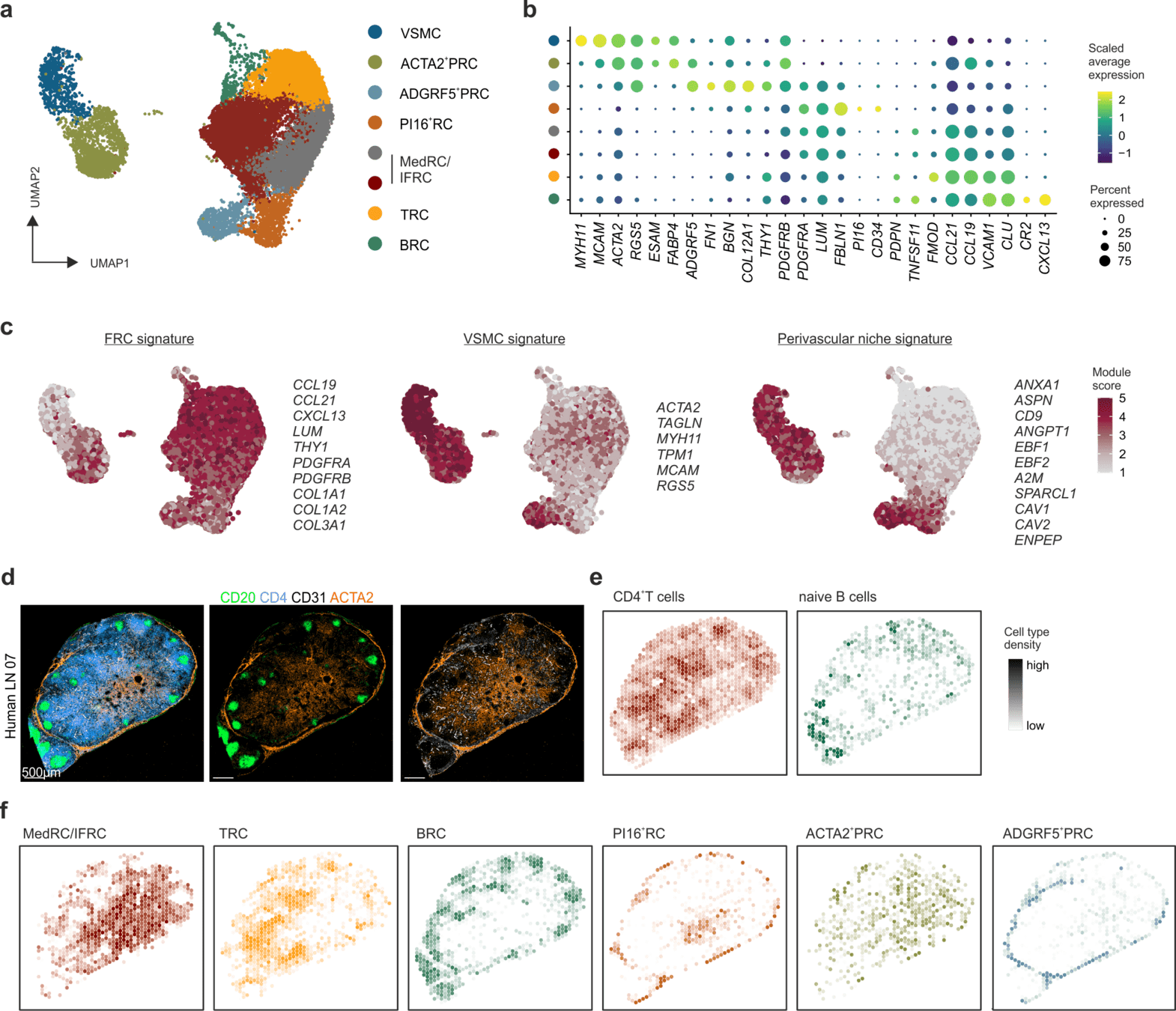
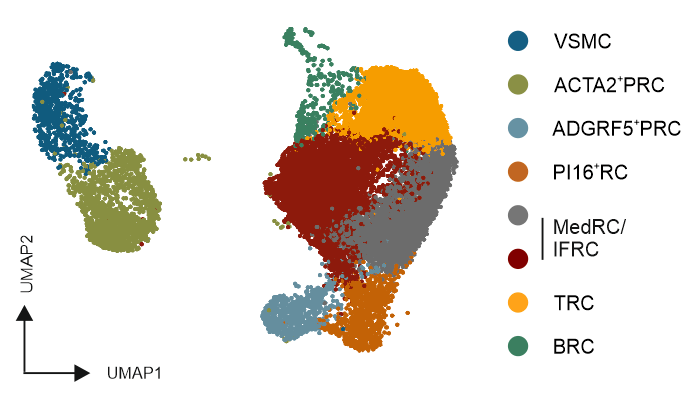
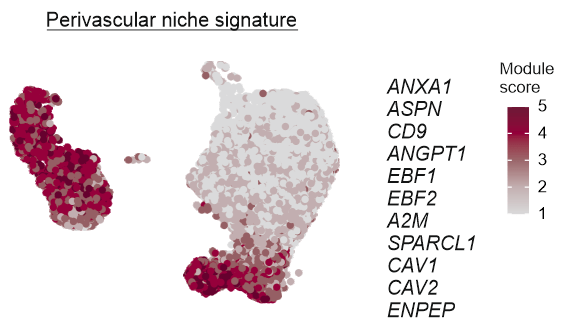
Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils
-
Distinct perivascular FRC subsets


Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils
-
Distinct perivascular FRC subsets and VSMCs share the expression of genes involved in vascular support

Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils
-
Distinct perivascular FRC subsets and VSMCs share the expression of genes involved in vascular support
Distinct FRC subsets form the perivascular niche in human lymph nodes
Distinct FRC subsets form the perivascular niche in human lymph nodes

Distinct FRC subsets form the perivascular niche in human lymph nodes

Distinct FRC subsets form the perivascular niche in human lymph nodes

Distinct FRC subsets form the perivascular niche in human lymph nodes

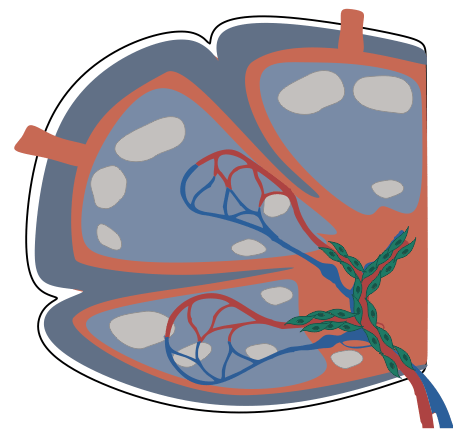
- PI16+RCs: subcapsular sinus and medulla
-
ACTA2+PRC: medulla and T cell zone
-
ADGRF5+PRC: subcapsular region
Distinct FRC subsets form the perivascular niche in human lymph nodes

- PI16+RCs: subcapsular sinus and medulla
-
ACTA2+PRC: medulla and T cell zone
-
ADGRF5+PRC: subcapsular region
→ Structured organization along the lymphatic and blood vasculature
A vascular zonation imprints FRC subset identities
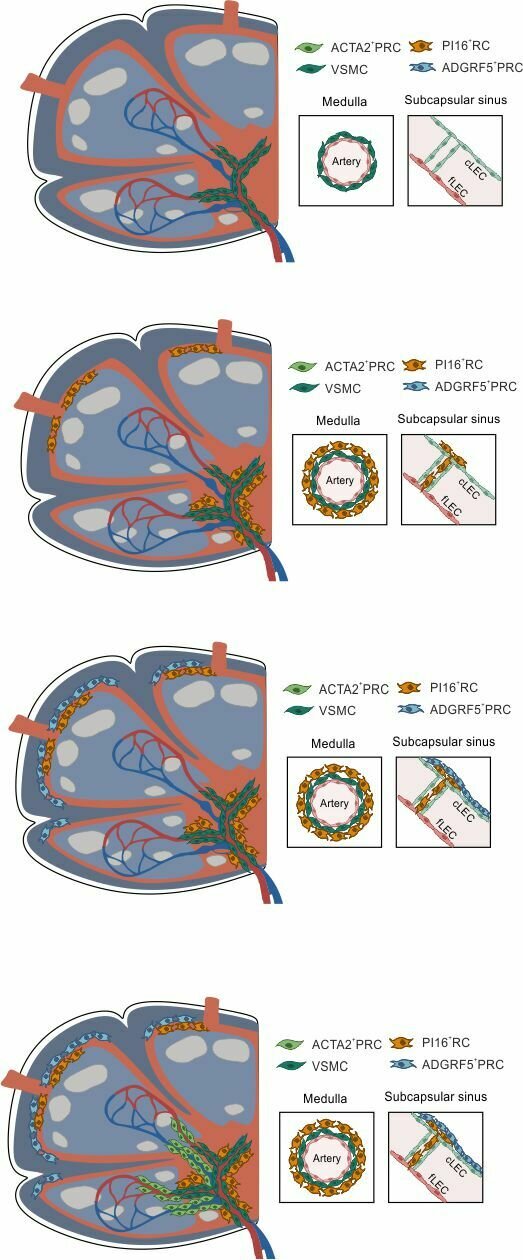




A vascular zonation imprints FRC subset identities



A vascular zonation imprints FRC subset identities



A vascular zonation imprints FRC subset identities



A vascular zonation imprints FRC subset identities

A vascular zonation imprints FRC subset identities
Distinct FRC subsets form the perivascular niche in human lymph nodes

Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils


Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils
-
PI16+RCs, PRCs and VSMCs share the expression of genes involved in vascular support


Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils
-
PI16+RCs, PRCs and VSMCs share the expression of genes involved in vascular support → Localization?



Distinct FRC subsets form the perivascular niche in human lymph nodes
-
Strong overlap with FRC landscape in human tonsils
-
PI16+RCs, PRCs and VSMCs share the expression of genes involved in vascular support → Localization?
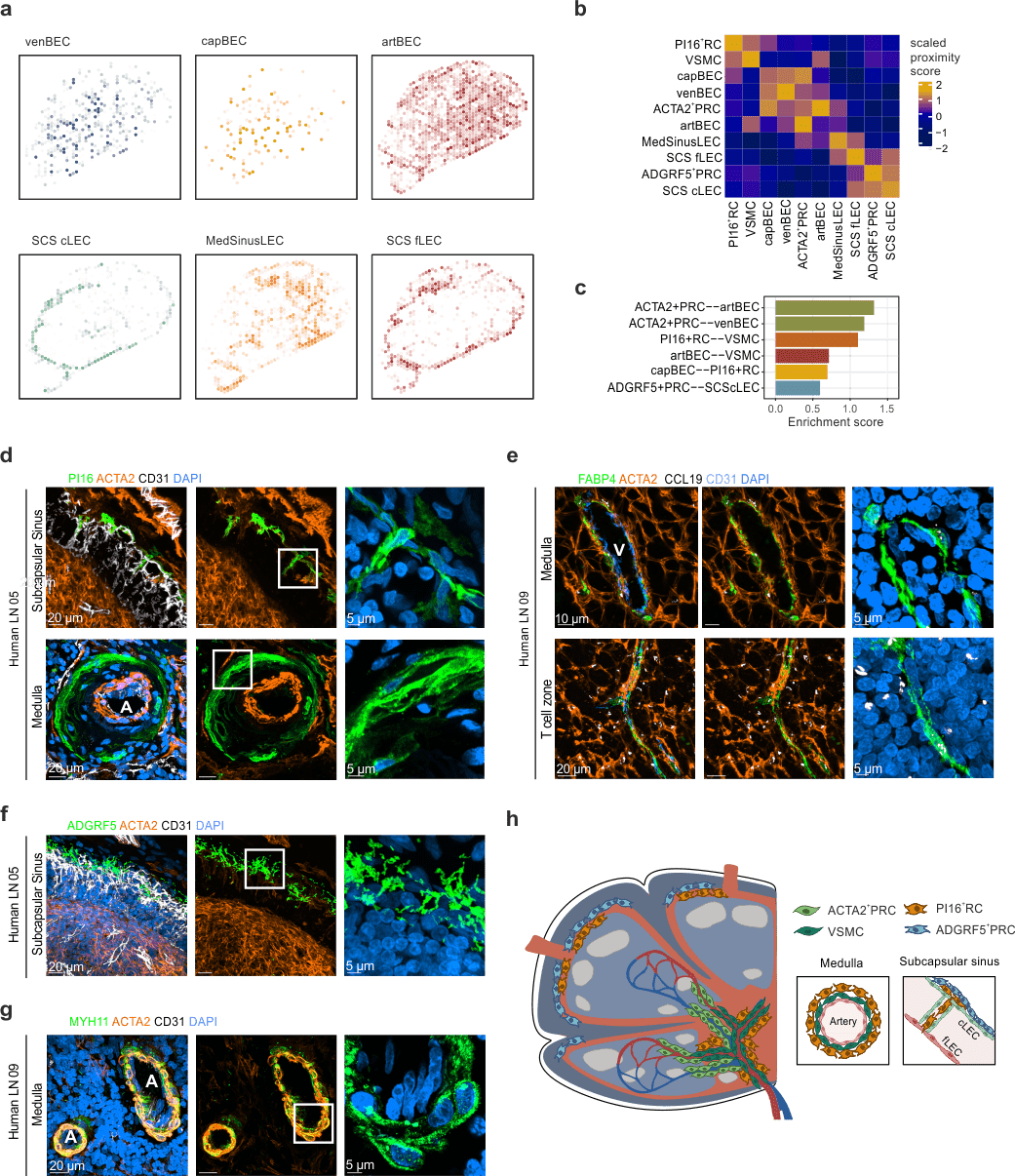
- Spatial transcriptomics predicts PI16+RCs in the subcapsular sinus and medulla



Spatial transcriptomics to predict the localization of PI16+RCs
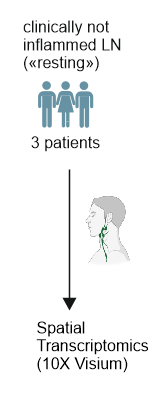
Spatial transcriptomics to predict the localization of PI16+RCs

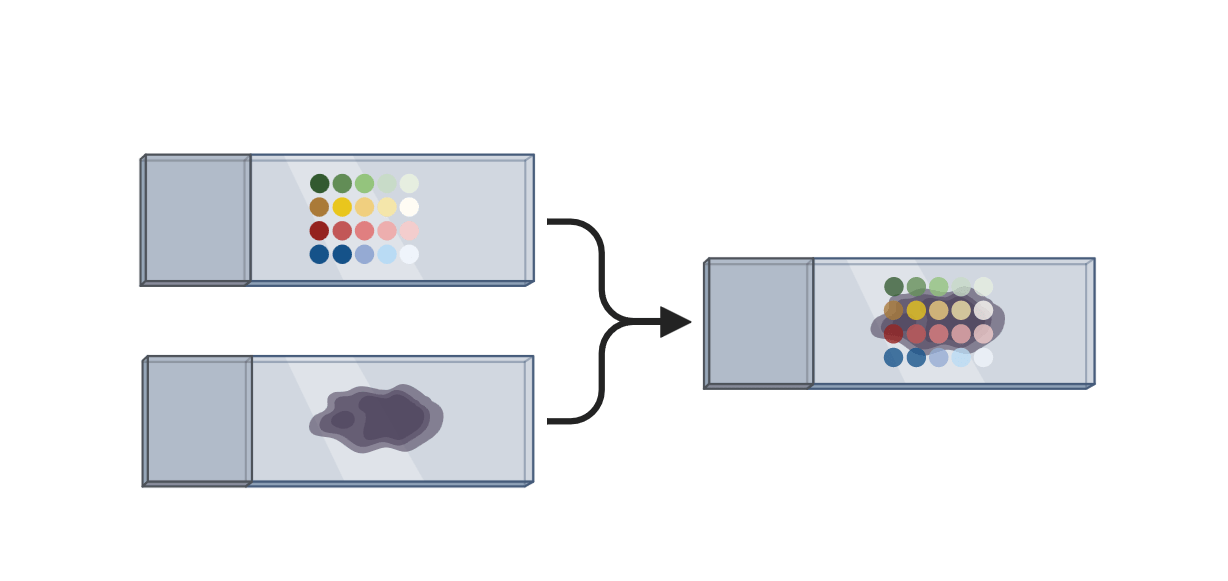
- measure gene expression for each spot on a tissue slide
Spatial transcriptomics to predict the localization of PI16+RCs


- measure gene expression for each spot on a tissue slide
- up to 30 cells per spot → predict cell type abundance based on scRNAseq data
Spatial transcriptomics to predict the localization of PI16+RCs


Spatial transcriptomics to predict the localization of PI16+RCs


Spatial transcriptomics to predict the localization of PI16+RCs


Spatial transcriptomics to predict the localization of PI16+RCs

- PI16+RCs: subcapsular sinus and medulla

PI16+RCs support the subcapsular sinus and large arteries in the medulla
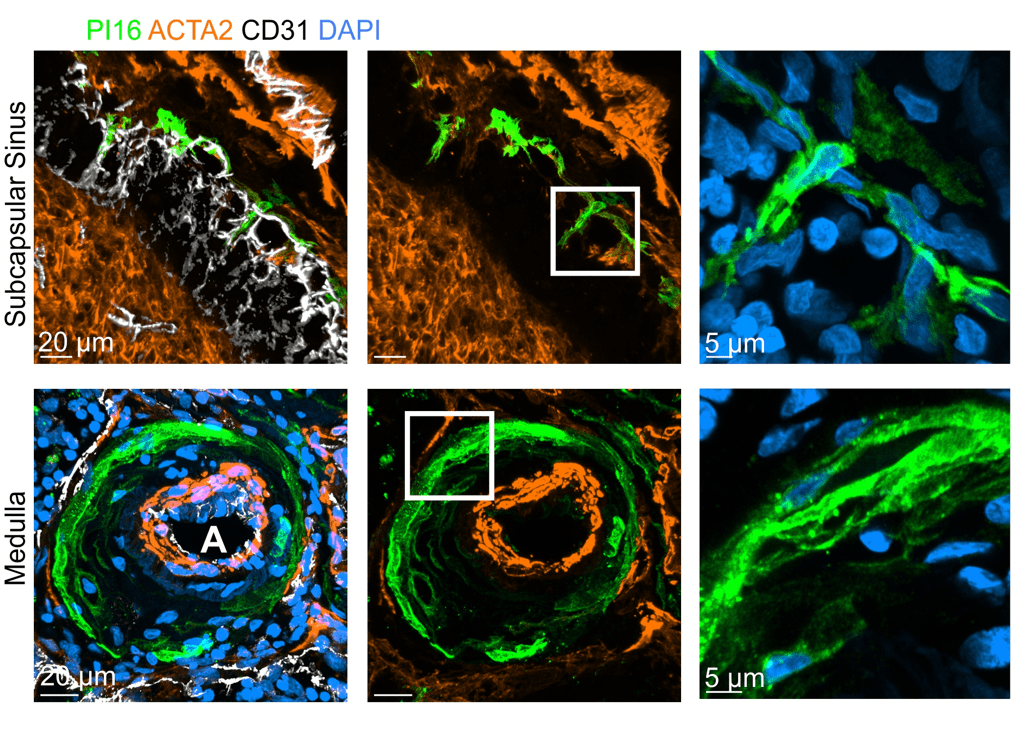
PI16+RCs support the subcapsular sinus and large arteries in the medulla

PI16+RCs support the subcapsular sinus and large arteries in the medulla


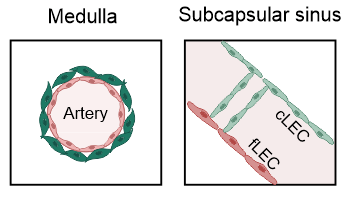

PI16+RCs support the subcapsular sinus and large arteries in the medulla

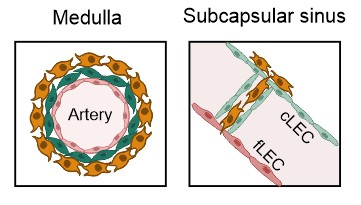
PI16+ RCs support inflammation-induced remodeling in human lymph nodes
PI16+ RCs support inflammation-induced remodeling in human lymph nodes
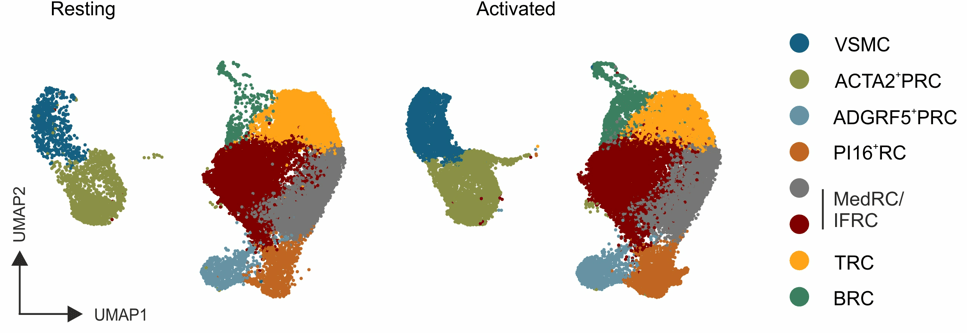

PI16+ RCs support inflammation-induced remodeling in human lymph nodes
- All FRC subset are conserved upon chronic activation


PI16+ RCs support inflammation-induced remodeling in human lymph nodes
- All FRC subset are conserved upon chronic activation
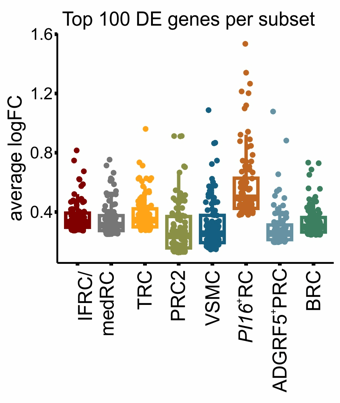


PI16+ RCs support inflammation-induced remodeling in human lymph nodes
- All FRC subset are conserved upon chronic activation
- PI16+RCs support tissue remodelling in inflamed human lymph nodes → immune cell interactions?



PI16+ RCs support inflammation-induced remodeling in human lymph nodes
- All FRC subset are conserved upon chronic activation
- PI16+RCs support tissue remodelling in inflamed human lymph nodes → immune cell interactions?
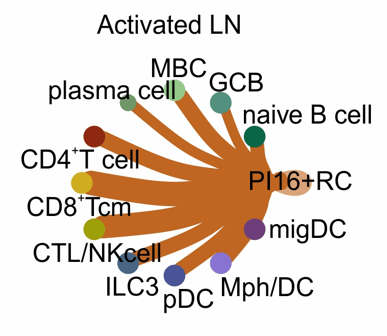



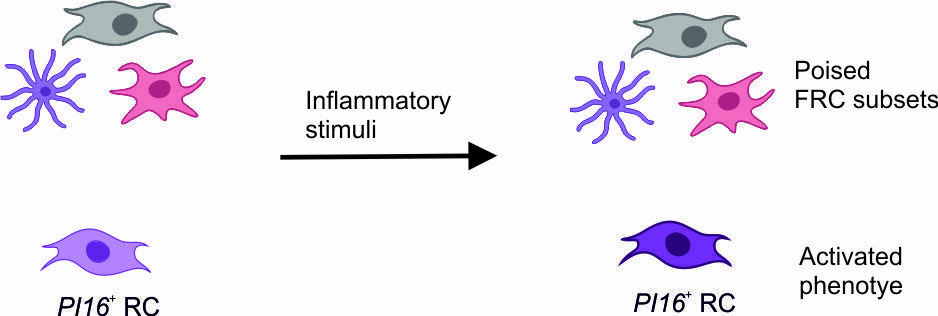
PI16+ RCs form reactive immune cell niches
- Bidirectional crosstalk between PI16+RCs and immune cells

- Bidirectional crosstalk between PI16+RCs and immune cells

Immune cell-provided activation cues
PI16+ RCs form reactive immune cell niches

- Bidirectional crosstalk between PI16+RCs and immune cells

Immune cell-provided activation cues
PI16+ RCs form reactive immune cell niches


Immune cell-provided activation cues
PI16+ RCs form reactive immune cell niches
Inferred interactions upon lymph node activation:
- PI16+RCs integrate activation cues from macrophages and dendritic cells


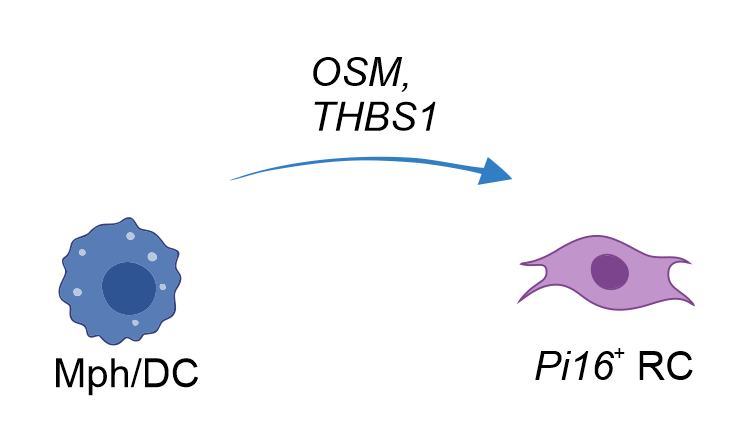
PI16+RC-provided immune-modulation cues
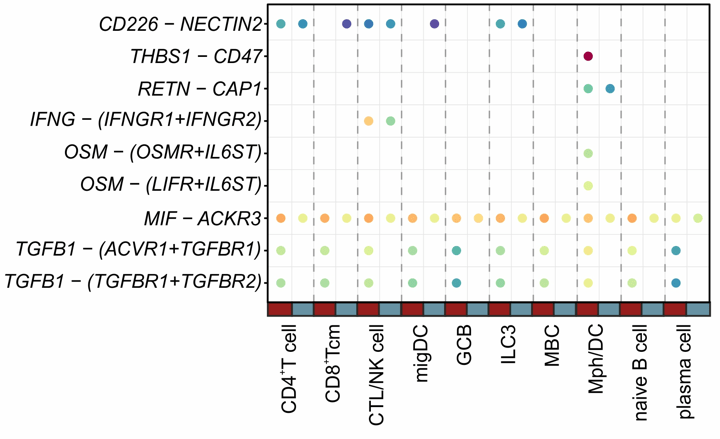
PI16+ RCs form reactive immune cell niches
Inferred interactions upon lymph node activation:
- PI16+RCs integrate activation cues from macrophages and dendritic cells

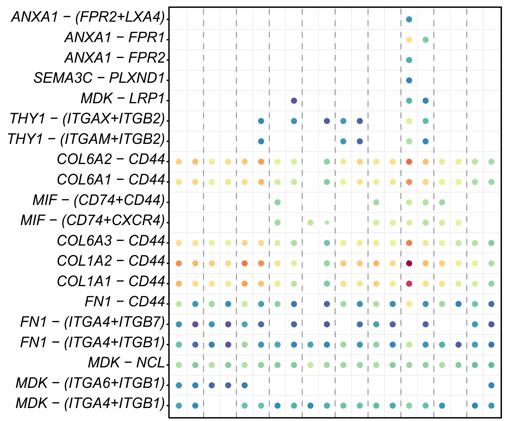


PI16+RC-provided immune-modulation cues
PI16+ RCs form reactive immune cell niches
Inferred interactions upon lymph node activation:
- PI16+RCs integrate activation cues from macrophages and dendritic cells




PI16+RC-provided immune-modulation cues
PI16+ RCs form reactive immune cell niches
Inferred interactions upon lymph node activation:
- PI16+RCs integrate activation cues from macrophages and dendritic cells
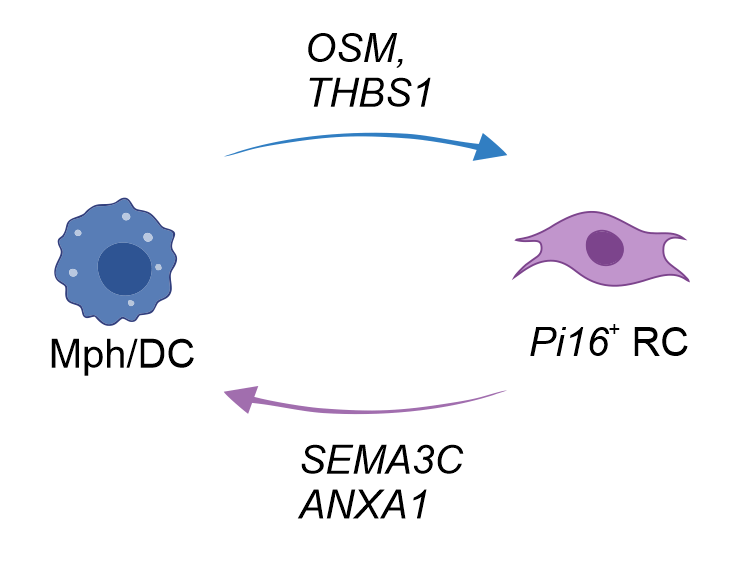
- PI16+RCs provide immune modulatory cues to macrophages and dendritic cells



PI16+RC-provided immune-modulation cues
→ Validation?
PI16+ RCs form reactive immune cell niches
Inferred interactions upon lymph node activation:
- PI16+RCs integrate activation cues from macrophages and dendritic cells
- PI16+RCs provide immune modulatory cues to macrophages and dendritic cells




PI16+ RCs form reactive immune cell niches
- Perivenous PI16+RCs in activated human lymph nodes
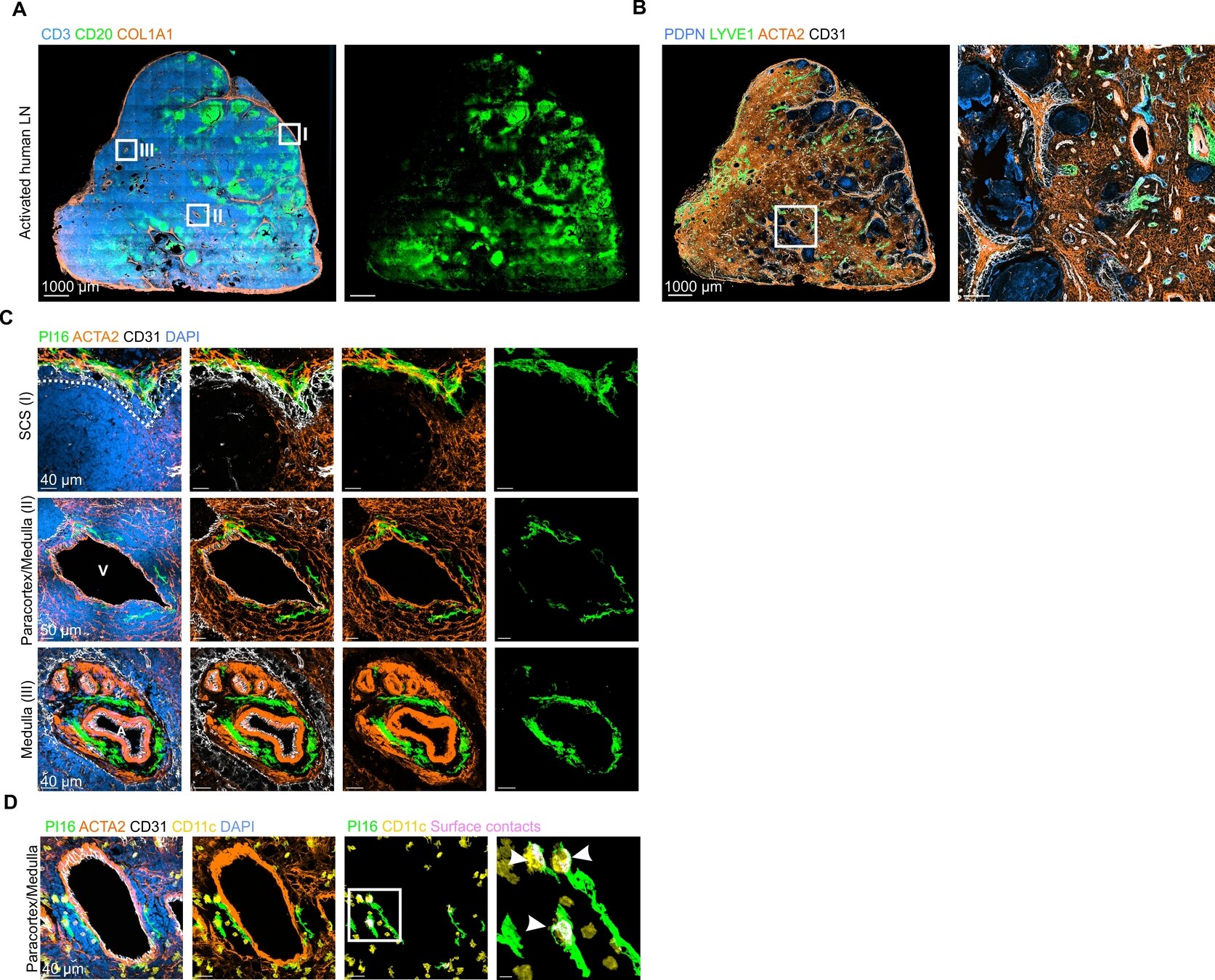
PI16+ RCs form reactive immune cell niches
- Perivenous PI16+RCs in activated human lymph nodes
- Surface contact between PI16+RCs and CD11c+myeloid cells


Summary II: PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Stereotypic «resting» human lymph node:
Summary II: PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Stereotypic «resting» human lymph node:
- Baseline activation and atypical positioning of B cell follicles likely reflecting topological remnants of recurrent activation

Summary II: PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Stereotypic «resting» human lymph node:
- Baseline activation and atypical positioning of B cell follicles likely reflecting topological remnants of recurrent activation
- Extensive vasculature with a large perivascular space formed by distinct FRC subsets including PI16+RCs

Summary II: PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Stereotypic «resting» human lymph node:
- Baseline activation and atypical positioning of B cell follicles likely reflecting topological remnants of recurrent activation
- Extensive vasculature with a large perivascular space formed by distinct FRC subsets including PI16+RCs

Inflammatory activation of human lymph nodes:
Summary II: PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Stereotypic «resting» human lymph node:
- Baseline activation and atypical positioning of B cell follicles likely reflecting topological remnants of recurrent activation
- Extensive vasculature with a large perivascular space formed by distinct FRC subsets including PI16+RCs

Inflammatory activation of human lymph nodes:
- PI16+ RCs support inflammation-induced tissue remodeling and form reactive immune cell niches (validation ongoing)

Summary II: PI16+ reticular cells form reactive immune cell niches in human lymph nodes
Stereotypic «resting» human lymph node:
- Baseline activation and atypical positioning of B cell follicles likely reflecting topological remnants of recurrent activation
- Extensive vasculature with a large perivascular space formed by distinct FRC subsets including PI16+RCs

Inflammatory activation of human lymph nodes:
- PI16+ RCs support inflammation-induced tissue remodeling and form reactive immune cell niches (validation ongoing)
- Perivenous PI16+RCs engage with immune cells in activated human lymph nodes

Burkhard Ludewig Group
Lisa Kurz
Angelina De Martin
Nadine Cadosch
Christian Perez-Shibayama
Cristina Gil-Cruz
Hung-Wei Cheng
Lucas Onder
Natalia Pikor Group
Sarah Grabherr
Acknowledgements

Department of
Otorhinolarnygology (KSSG)
Sandro Stöckli
Yves Stanossek
Samuel Meili
PhD Committee
Burkhard Ludewig
Mark D. Robinson
Maries van den Broek
University of Zurich
Charlotte Soneson
Almut Lütge
University of Pennsylvania
Joshua Brandstadter
Ivan Maillard



10MinutesSummaryPhD_ML
By mechthildlue
10MinutesSummaryPhD_ML
- 136



