AHOD0431: PET-adapted therapy for early-stage, low-risk pediatric Hodgkin lymphoma
Parekh et al.
Blood 2022
Background
- Pediatric early-stage classic Hodgkin lymphoma (cHL) is highly curable
- Late toxicity from chemotherapy and radiation therapy is a concern
- Treatment strategies aim to maintain high cure rates while minimizing late effects
- Response-adapted protocols using PET scans can identify patients for whom radiotherapy may be omitted
- COG developed AHOD0431 to de-escalate treatment in early-stage, low-risk cHL
Study Objectives
- Primary: Evaluate if reducing up-front treatment using a response-based approach with minimal initial chemotherapy and omission of IFRT in complete responders could maintain overall survival and event-free survival while reducing late toxicity
- Secondary: Investigate the impact of PET response after 1 cycle (PET1) on outcomes and patterns of relapse
Eligibility Criteria
- Age: 0 to 21 years
- Diagnosis: Low-risk classic Hodgkin lymphoma
- Stage: Ann Arbor stage IA and IIA nonbulky disease
- Bulk definition:
- Mediastinal mass > one-third of longest thoracic dimension on upright PA chest x-ray
- Any contiguous nodal aggregate > 6 cm across longest transverse diameter on axial imaging
AHOD0431 Trial Schema
PET Response Assessment
PET negative defined as:
Activity below or at the level of the mediastinal blood pool (Below Deauville 3)
PR Definition:
defined as >50% but <80% decrease in perpendicular dimension on CT or positive PET or gallium scan
CR Definition:
≥80% reduction in the product of perpendicular dimensions and no residual extramediastinal lymph node mass >2.0 cm
Study Accrual
AHOD0431 was temporarily closed to accrual on 4 December2008 because of an increased risk of relapse among PET1 negative patients who did not receive IFRT because they had achieved a CR
It was recommended that all patients with equivocal or positive PET1 receive 21-Gy IFRT, unless they were 12 months from completion of chemotherapy.
Radiation Therapy Details
- Dose: 21 Gy
- Fractionation: 1.5 Gy per fraction
- Total treatment time: 14 days
- Timing: 3-4 weeks after final cycle of chemotherapy
- Technique: Involved-field radiotherapy (IFRT)
- Volume: Sites initially involved by disease
- Delivery: Balanced anterior-posterior fields
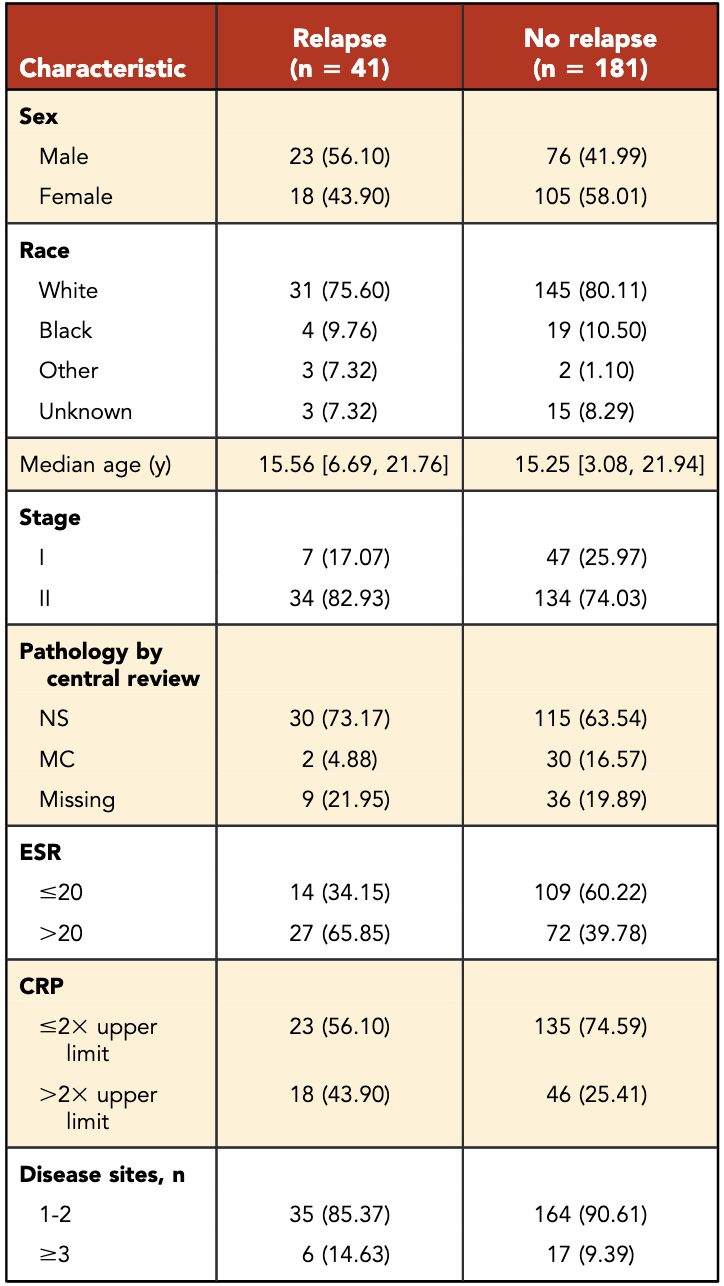
Patient Characteristics
- Total patients: 222
- Median age: 15.3 years
- Male: 44.6%
- Stage II: 75.7%
- Rapid early responders (RERs): 54% (n=119)
- Slow early responders (SERs): 46% (n=103)
- SERs more likely to have:
- Stage II disease (p=0.0017)
- ESR >20 (p=0.0001)
- ≥3 sites of disease (p=0.0052)
Primary Outcome
- 10-year progression-free survival (PFS):
- RERs: 87.1%
- SERs: 73.5%
- p = 0.01
- RERs:
- With IFRT: 96.6%
- Without IFRT: 84.1%
- p = 0.10
- SERs:
- With IFRT: 80.9%
- Without IFRT: 64.0%
- p = 0.03
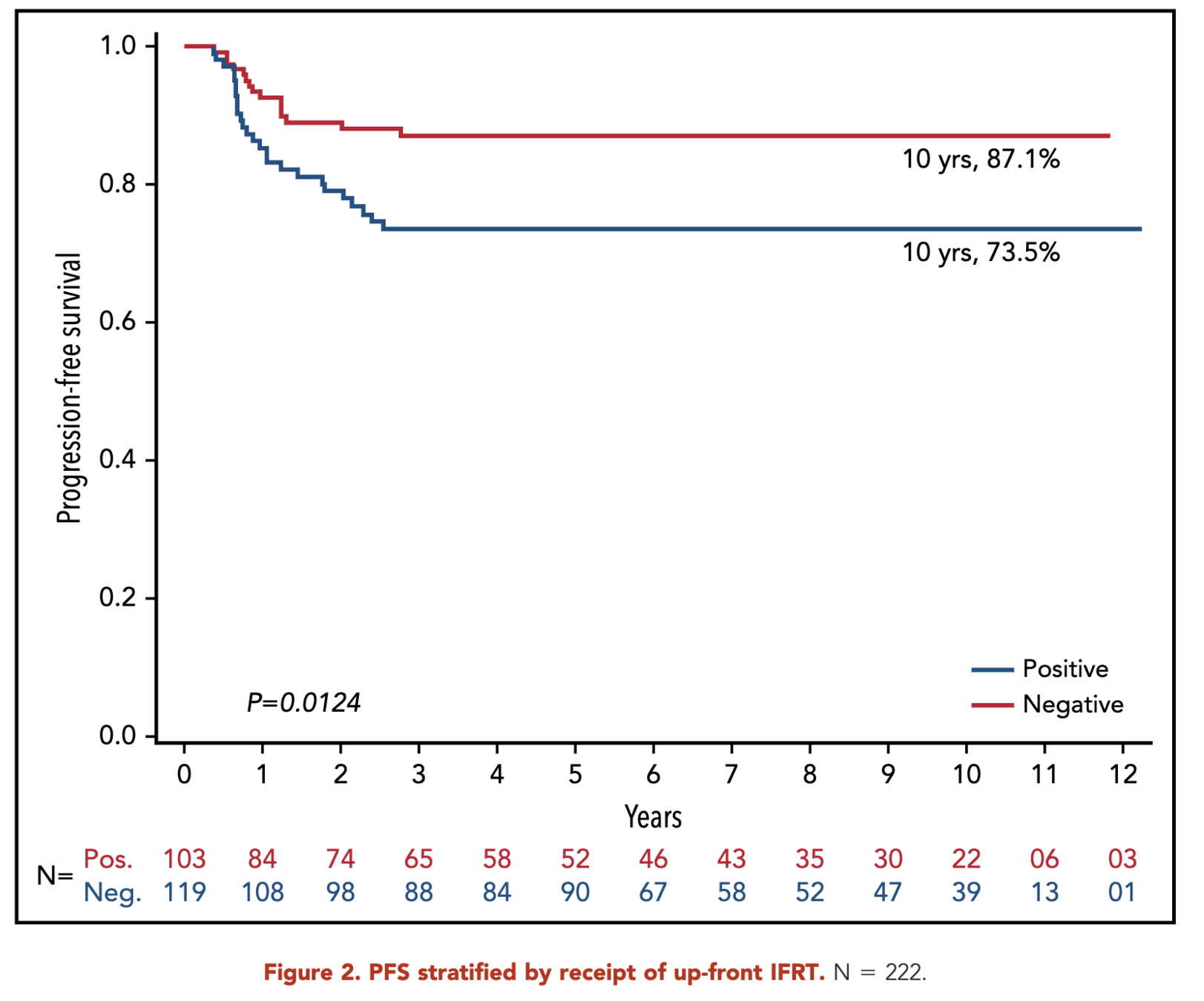
- 10-year progression-free survival (PFS):
- RERs: 87.1%
- SERs: 73.5%
- p = 0.01
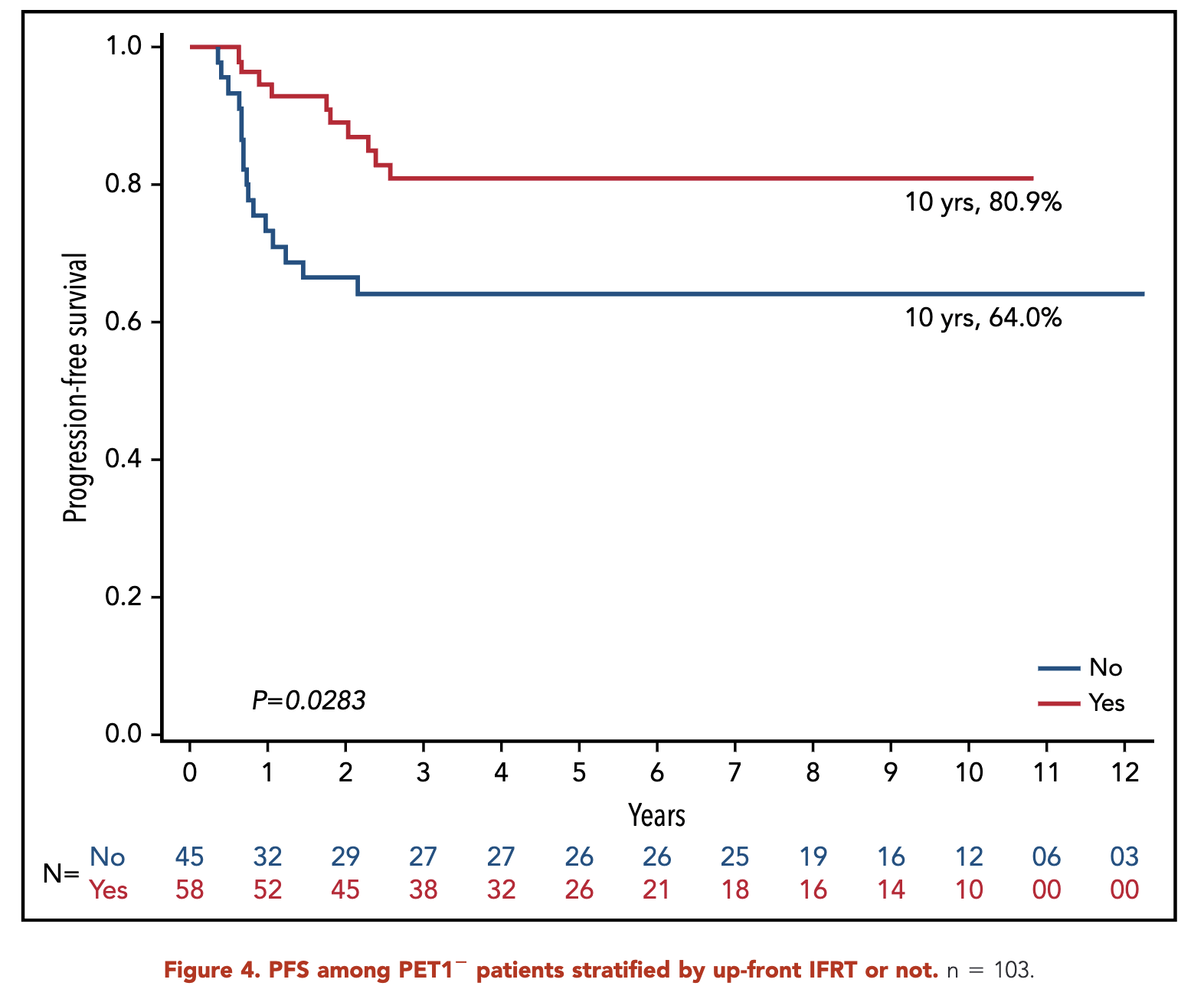
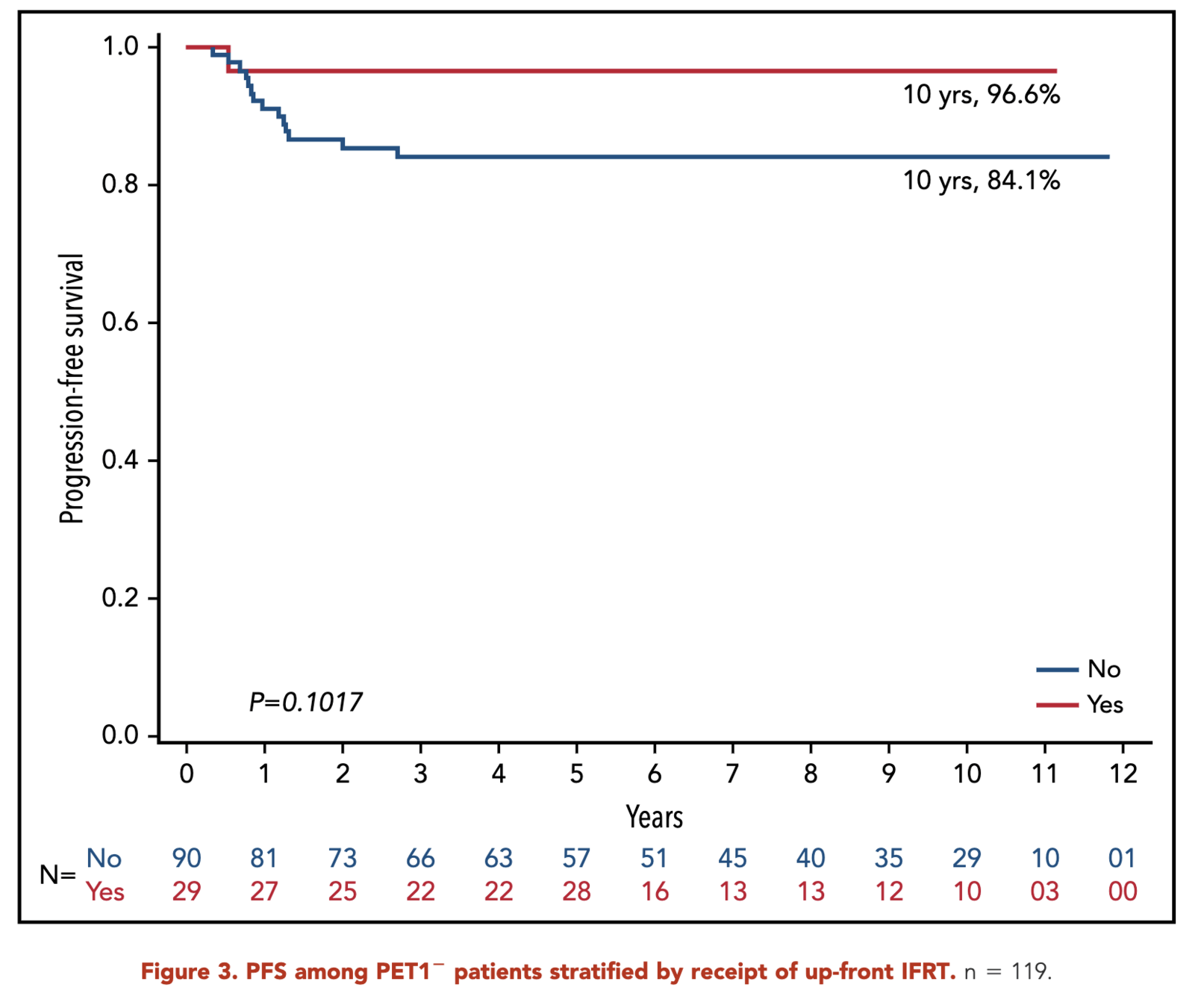

- RERs:
- With IFRT: 96.6%
- Without IFRT: 84.1%
- p = 0.10
- SERs:
- With IFRT: 80.9%
- Without IFRT: 64.0%
- p = 0.03
Primary Outcome (RERS)
- 10-year progression-free survival (PFS):
- RERs: 87.1%
- SERs: 73.5%
- p = 0.01
- RERs:
- With IFRT: 96.6%
- Without IFRT: 84.1%
- p = 0.10
- SERs:
- With IFRT: 80.9%
- Without IFRT: 64.0%
- p = 0.03

- 10-year progression-free survival (PFS):
- RERs: 87.1%
- SERs: 73.5%
- p = 0.01



- RERs:
- With IFRT: 96.6%
- Without IFRT: 84.1%
- p = 0.10
- SERs:
- With IFRT: 80.9%
- Without IFRT: 64.0%
- p = 0.03
Primary Outcome (sERs)
- 10-year progression-free survival (PFS):
- RERs: 87.1%
- SERs: 73.5%
- p = 0.01
- RERs:
- With IFRT: 96.6%
- Without IFRT: 84.1%
- p = 0.10
- SERs:
- With IFRT: 80.9%
- Without IFRT: 64.0%
- p = 0.03

Patterns of Relapse
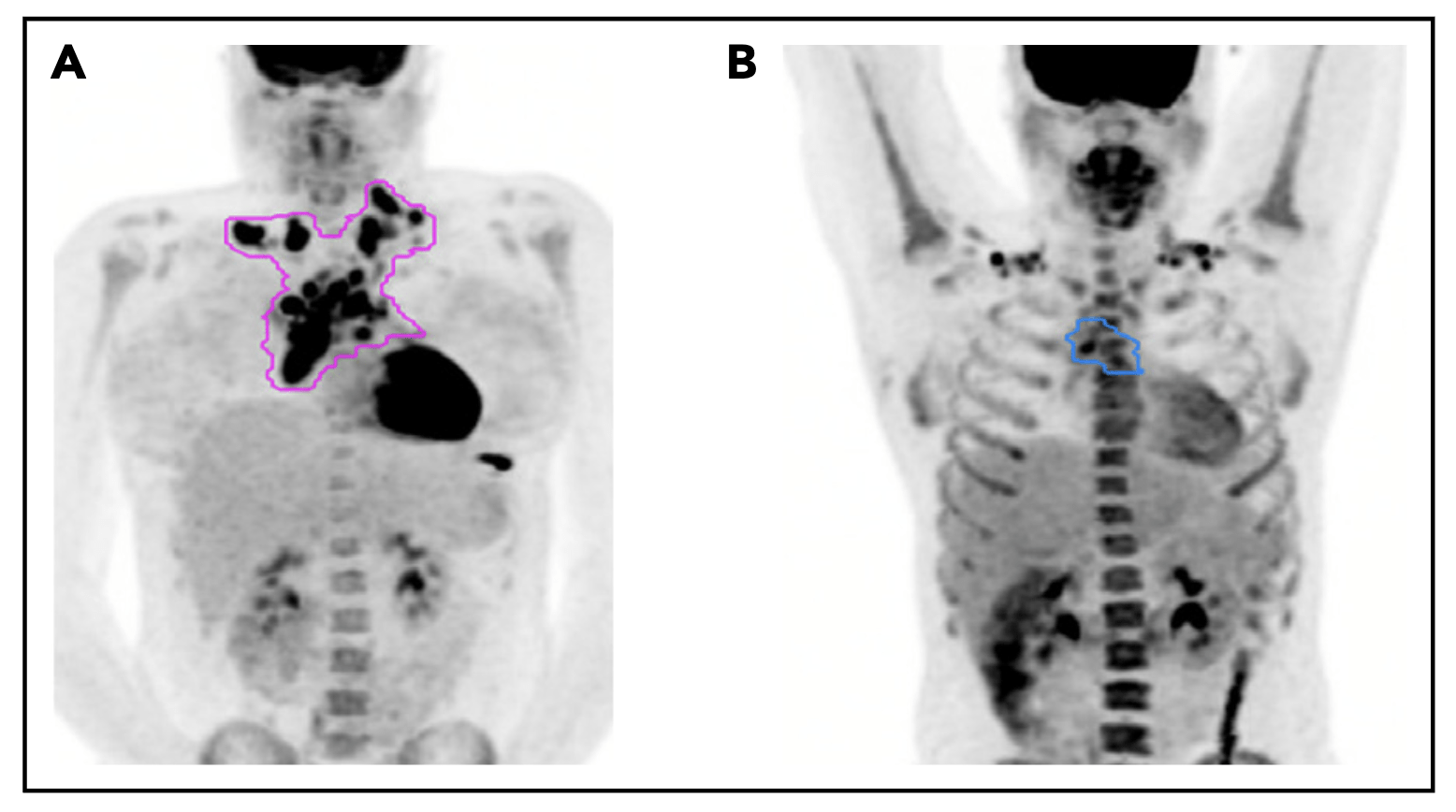
Pattern of relapse definitions. (A) Theoretical outline (pink) of initial site of disease based on the prechemotherapy PET/CT scan. A relapse within the out-lined region would have been categorized as a relapse in the “initial site” of disease. A relapse outside of the outlined region would be considered a “new site” ofrelapse. (B) Theoretical outline (blue) of fluorodeoxyglucose-avid residual disease greater than the mediastinal blood pool on PET/CT after 1 cycle of AVPC. For PET11patients, a relapse within the blue region would be considered a relapse at the “PET11 site,” whereas a relapse outside of the blue region but within the pink regionfrom panel A would be considered “initial site beyond the PET11 site.”
Patterns of Relapse - RERs
- Total relapses: 15
- All relapses involved an initial site of disease
- Without IFRT (14 relapses):
- 11 (78%): Initial site only
- 3 (21%): Initial plus new site
- With IFRT (1 relapse):
- 1 (100%): Initial irradiated site only
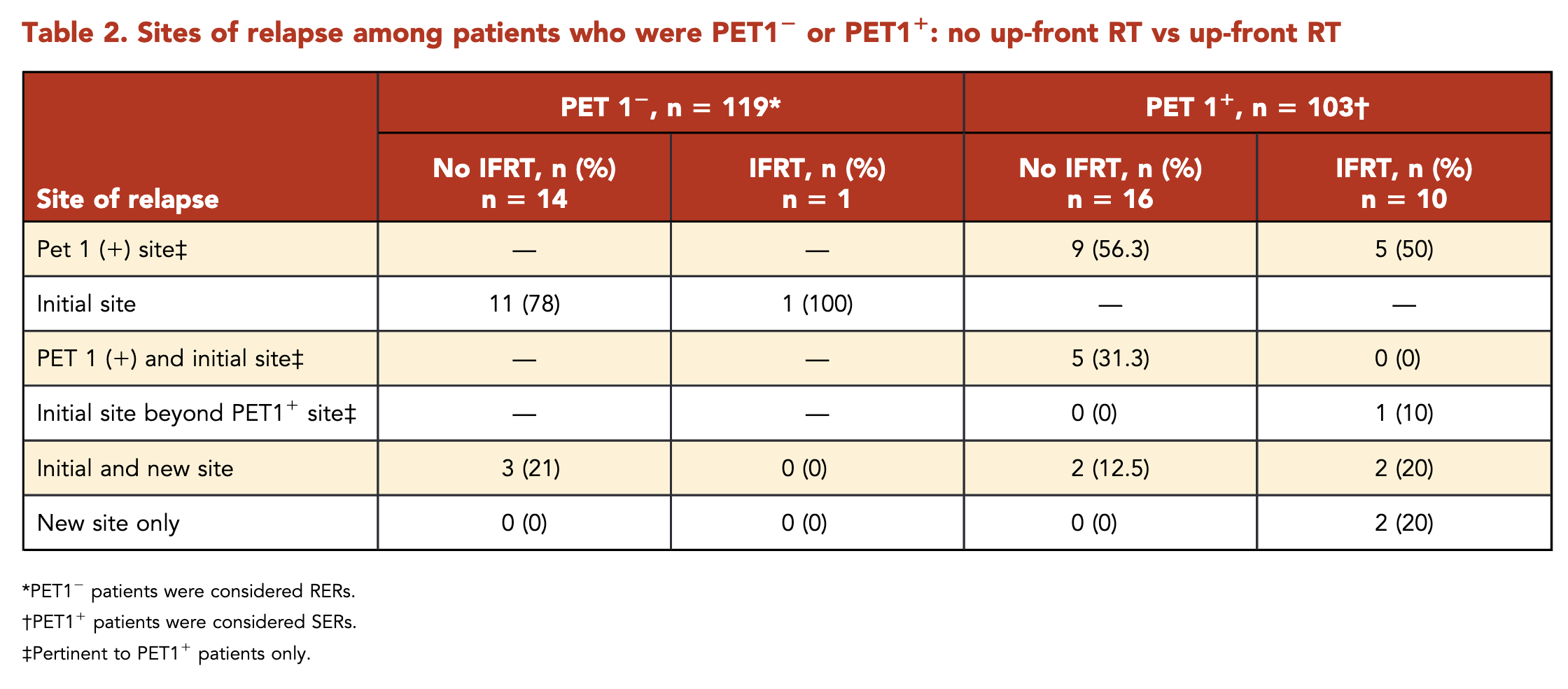
Patterns of Relapse - SERs

- Total relapses: 26
- 24 (92%) had relapse at an initial site
- 14 (54%) solely at the PET1+ site
- Without IFRT (16 relapses):
- 9 (56%): PET1+ site only
- 5 (31%): PET1+ site and outside PET1+ but within initial site
- 2 (12%): Initial and new site
- With IFRT (10 relapses):
- 5 (50%): PET1+ site only
- 1 (10%): Initial site beyond PET1+ site
- 2 (20%): Initial and new site
- 2 (20%): New site only
Comparison with Other Studies
| Study | PET Timing | Treatment | RT Criteria | RT Dose | Outcome |
|---|---|---|---|---|---|
| AHOD0431 (Current) | PET1 & PET3 | AVPC x3 | PR after 3 cycles (PET3+) | 21 Gy IFRT | 10-year PFS: RER: 96.6% (CMT) vs 84.1% (chemo alone) SER: 80.9% (IFRT) vs 64.0% (no IFRT) |
| EORTC H10 | PET2 | Standard: ABVD x3 + IFRT Experimental: ABVD x4 |
All in standard arm PET2+ in experimental arm |
30 Gy IFRT | 5-year PFS: 99.0% vs 87.1% |
| UK RAPID | PET3 | ABVD x3 ± IFRT | PET3+ or randomized to RT arm if PET3- | 30 Gy IFRT | 3-year PFS: 97.1% vs 90.8% |
| GHSG HD16 | PET2 | Standard: ABVD x2 + IFRT Experimental: ABVD x2 |
All in standard arm PET2+ in experimental arm |
20 Gy IFRT | 5-year PFS: 93.4% vs 86.1% |
- AHOD0431 unique in using both PET1 and PET3 for response assessment and allowing RT omission for SERs (PET1+) with CR at PET3
- Other studies used either PET2 or PET3 for response-adapted approach
- EORTC H10 and GHSG HD16 gave RT to all patients in standard arm, regardless of PET response
- UK RAPID randomized PET3- patients to RT or no RT
- AHOD0431 used lower RT dose (21 Gy) compared to EORTC H10 and UK RAPID (30 Gy)
- Results across studies consistently show improved outcomes with inclusion of RT, even in PET-negative patients
Comparison with Other Studies
| Study | PET Timing | Treatment | Outcome |
|---|---|---|---|
| AHOD0431 (Current) | PET1 | CMT Chemo alone |
10-year PFS: 96.6% 10-year PFS: 84.1% |
| EORTC H10 | PET2 | ABVD x3 + 30Gy IFRT ABVD x4 alone |
5-year PFS: 99.0% 5-year PFS: 87.1% |
| UK RAPID | PET3 | ABVD x3 + 30Gy IFRT ABVD x3 alone |
3-year PFS: 97.1% 3-year PFS: 90.8% |
| GHSG HD16 | PET2 | ABVD x2 + 20Gy IFRT ABVD x2 alone |
5-year PFS: 93.4% 5-year PFS: 86.1% |
- AHOD0431 used PET1, resulting in lower proportion of patients (54%) eligible for RT omission compared to PET2/PET3 studies (66-91%)
- In-field recurrences drove PFS differences in patients not receiving IFRT across studies
- AHOD0431 unique in allowing RT omission for SERs (PET1+)
- SERs showed improved outcomes with IFRT (10-year PFS: 80.9% vs 64%, p < .05)
- Results suggest potential benefit from dose intensification above 21 Gy to PET1+ regions in SERs
Conclusions
- PET1 response is a significant predictor of treatment outcome
- RERs showed favorable results with AVPC alone
- SERs had unfavorable outcomes with AVPC alone, but improved with 21 Gy IFRT
- RT remains an important component of treatment for SERs
- Most relapses occurred within the initial site of disease, often within the PET1+ site
- SERs may benefit from alternative systemic therapy and/or targeted RT intensification to PET1+ sites
Strengths and Limitations
- Strengths:
- Long-term follow-up (median 118 months)
- Centralized PET review
- Detailed analysis of relapse patterns
- Limitations:
- Exploratory analysis, not initially planned
- Small sample size in some subgroups
- Comparison with other response-adapted trials challenging due to differences in inclusion criteria and treatment regimens
Discussion Points
- How does the use of PET1 in this study compare to other trials using PET2 or PET3 for response assessment?
- What are the implications of the relapse patterns observed in this study for future RT planning in pediatric Hodgkin lymphoma?
- Should the radiation dose be increased for SERs, particularly to PET1+ sites? What are the potential benefits and risks?
- How might the results of this study influence the design of future pediatric Hodgkin lymphoma trials?
- What alternative systemic therapies could be considered for SERs to improve outcomes?
AHOD0431: PET-adapted therapy for early-stage, low-risk pediatric Hodgkin lymphoma
By RadMedSkiier
AHOD0431: PET-adapted therapy for early-stage, low-risk pediatric Hodgkin lymphoma
AHOD0431 trial, exploring PET-adapted therapy for low-risk pediatric Hodgkin lymphoma, with insights into patient outcomes, relapse patterns, and comparisons to existing studies.
- 27



