gWear FU
23 November 2015
Table of
Contents
Follow-up
Devices
Timelines
Discussion
Follow-up
FU study III:
Commercial driven

FU study II:
Real-life experience
INTEGR8:
2-way communication
FU study II
Patient population
- Internal & external patients
- Broaden age range
- Global
- Multi-language
Study design
- Data entry / answer questions
- Extended functionality (metrics)
- Data correlation
- QOL questions with license available
- Include HPX participation

FU study II
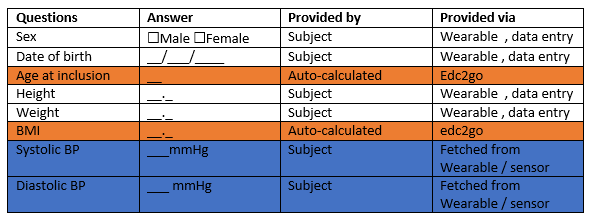
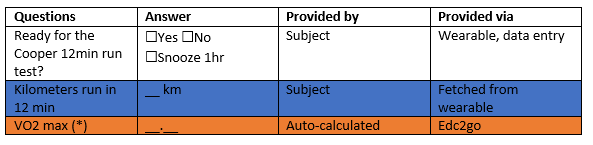
CRF
Baseline
Cooper 12min run-test
Approach
Define study
- Device
- Protocol
- CRF
- Consent Form
- Participants
- Sign-off
Development
- Mockups
- Engineering
- Testing
- Validation
Communication
- Internal
- External
Study conduct
Publication
Potential devices


Potential devices

Potential devices

Final deadline for device selection is 1-Jan-2016
- Wait for christmas deals (consumer electronics)
- More information about other devices by year's end
FU study III
Patient population
- Include hospital (s)
- Include sponsors

Boston Scientific Contest
Submission
- Completed on 17 November
- Innovation webpage online as of 17 November
Promotion
- # votes
- PCT & ICI
- Internal & external mailing
- Check for jury connections

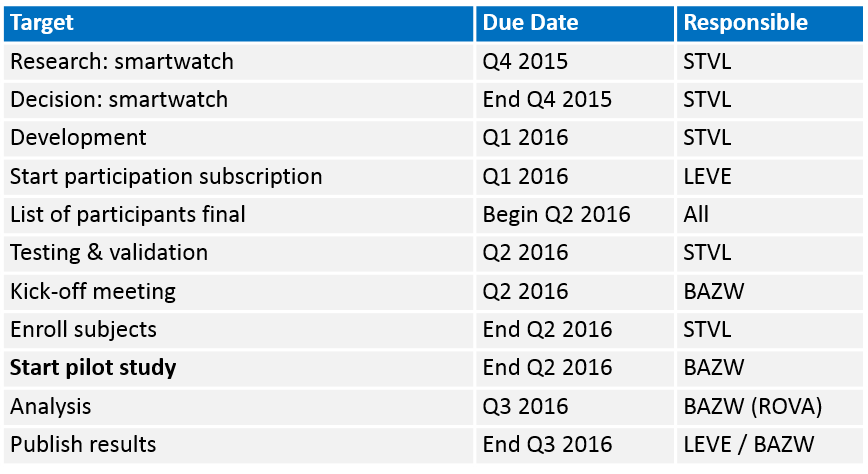
Estimated timelines
Let's discuss!
gWear FU 23 November 2015
By lennart_verstappen
gWear FU 23 November 2015
- 35



