gWear FU
21 December 2015
Table of
Contents
FU Study II
Device
Action items
Timelines
Discussion
FU study II
Patient population
- Internal & external patients
- Broaden age range
- Global
- Multi-language
Study design
- Data entry / answer questions
- Extended functionality (metrics)
- Data correlation
- QOL questions

FU study II
Study design
- 20 patients will wear the smartwatch
- 3 patients will also use the HPX patch
- Objective: demonstrate 2 way communication AND interaction with a different device
- Work with another consultant to improve connection issues (most likely Cognitec)

Chosen device
Why not others:
- Samsung GS: with 3G sim will not work in the EU
- Blocks: not shipped until June 2016
- genae’s previous experience with Microsoft Band
- Improved Bluetooth connectivity
- More ergonomic design: curved AMOLED screen
- Works with Windows Phone, iOS and Android

Action items
- Set-up meeting with gWear team and BASE: discuss HPX and Vital connect
- Set-up meeting with STLO and PEBO: discuss technical aspects
- Set-up structured brainstorming session:
- Objective: scope of conduct / design of study
- Concept: 2 hour session during lunch
- Group: mixed group of CRO / Sr Management / Support roles
- Timeline: mid January
Boston Scientific Contest
Update
- Most views (1.900) & votes (45)
- 36 submissions
- 19 days until submission ends
- 40 days until voting ends

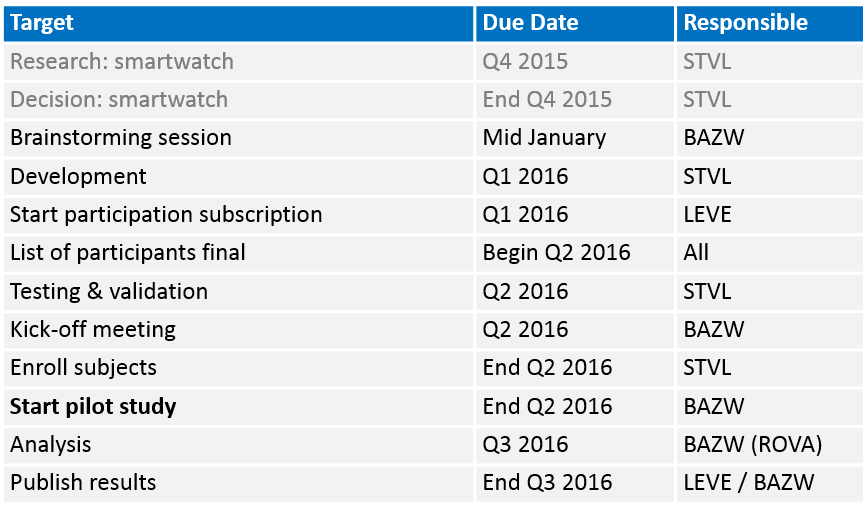
Estimated timelines
Let's discuss!
gWear FU 21 December 2015
By lennart_verstappen
gWear FU 21 December 2015
- 33



