Metastasis-directed radiotherapy without systemic therapy for oligometastatic clear-cell renal-cell carcinoma
Primary efficacy analysis of a single-arm, single-centre, phase 2 trial
Tang C, Sherry AD, Seo A, et al.
Lancet Oncology 2025; 26: 1289–99
Study design
- Single-arm, single-centre, phase 2 trial (MD Anderson Cancer Center)
- Enrolled July 2018 – May 2023; 121 patients
- Co-primary endpoints:
- Progression-free survival (RECIST 1.1) — per-protocol population
- Systemic therapy-free survival (STFS) — intention-to-treat population
- Prespecified threshold for success: median STFS ≥ 24 months
- Benchmark: Rini et al. surveillance study — median time to systemic therapy 14.9 months
- Sample size: 100 patients (84% power); increased to 120 for censoring
Eligibility criteria
- Inclusion:
- Age ≥ 18 years
- Histologically confirmed renal-cell carcinoma
- 1–5 sites of metastatic disease
- ECOG performance status 0–2
- Candidate for definitive radiotherapy to all disease sites
- Adequate organ function (ANC ≥ 1000, Plt ≥ 50k, Hgb ≥ 9, Tbili ≤ 1.5, ALT/AST ≤ 3× ULN)
- Off systemic therapy ≥ 1 month or systemic therapy-naïve
- Exclusion:
- Pregnancy
- Comorbidities contraindicating RT (e.g. active scleroderma)
- Diffuse metastatic processes (leptomeningeal, diffuse bone marrow, peritoneal carcinomatosis)
- RECIST 1.1 measurable disease was not required
Trial schema
Enrollment: 1–5 metastatic lesions, ccRCC, ECOG 0–2
Staging: CT chest + MRI abd/pelvis, CT CAP, or PET-CT
↓
MDT to all sites of disease (SBRT preferred: ≤ 5 fx, ≥ 7 Gy/fx)
Surgery or IR ablation permitted if ≥ 1 site treated with RT
↓
Surveillance off systemic therapy
Imaging q12 weeks × 1 year, then q18 weeks
↓
Disease progression?
↓
Oligoprogression (≤ 3 new/progressing sites)
↓
Additional round of MDT
Reset surveillance schedule
Polymetastatic progression (> 3 sites)
↓
Initiate systemic therapy
Also if: RT toxicity precludes MDT, local progression, or physician/patient choice
Patient characteristics
- 121 patients enrolled (ITT); 120 received RT (per-protocol)
- Median age: 66 years (IQR 61–72); 74% male
- 98% had prior nephrectomy; median time nephrectomy → enrollment: 71 months
- Median time metastasis diagnosis → enrollment: 10 months (IQR 2–29)
- ECOG PS: 0 (68%), 1 (29%), 2 (3%)
- IMDC risk: Favorable 52%, Intermediate 47%, Poor 1%
- Previous systemic therapy: 30% (most common: pazopanib n=11, nivo/ipi n=10)
- Prior local therapy: Metastasectomy 32%, RT 16%, IR ablation 8%
- Lesions treated (round 1): 1 lesion (59%), 2 lesions (31%), 3–5 lesions (10%)
- Most common met sites: Lung (n=76), lymph nodes (n=16)
- Median GTV of all irradiated sites: 5.7 cm³ (IQR 3.1–19.2)
Radiation and surveillance details
SBRT protocol
- ≤ 5 fractions at ≥ 7 Gy/fx to all disease sites
- Most common: 40–50 Gy / 4 fx (n=59)
- Alternative RT if location precluded SBRT
- Surgery/IR ablation if ≥ 1 site received RT
Treatment details
- Median GTV: 5.7 cm³ (IQR 3.1–19.2)
- Round 1: 1 lesion (59%), 2 (31%), 3–5 (10%)
- Most common sites: lung (n=76), LN (n=16)
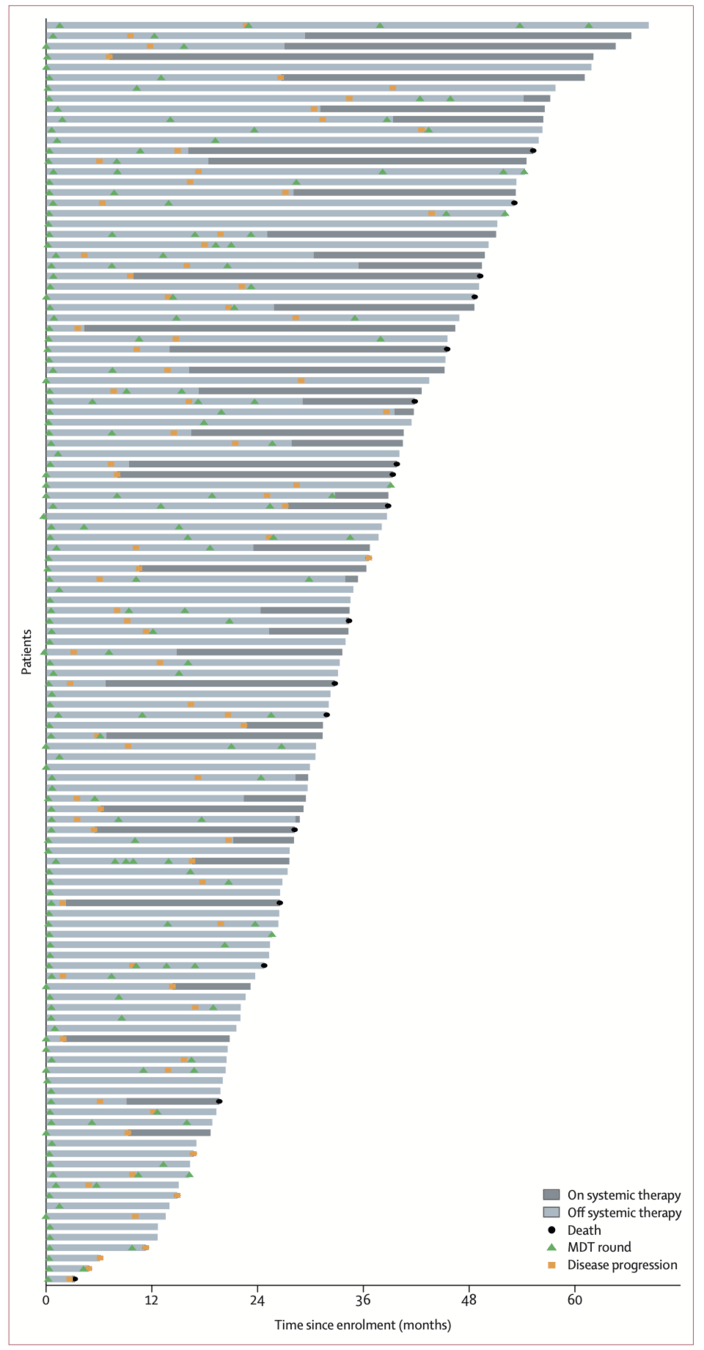
- 59% received ≥ 2 MDT rounds; up to 6 total
- Only 7% local progression in irradiated sites
Surveillance protocol
- Imaging q12 weeks × 1 year, then q18 weeks
- AE review + CBC with differential at each visit
- RECIST 1.1 with central radiology review
- Schedule resets after each MDT round
Oligoprogression → more MDT
- ≤ 3 new or progressing metastases
- Additional round of MDT, reset surveillance
Triggers for systemic therapy
- Polymetastatic progression (>3 sites)
- RT toxicity precluding further MDT
- Local progression after RT
- Physician or patient choice
51/121 (42%) ultimately started systemic Tx
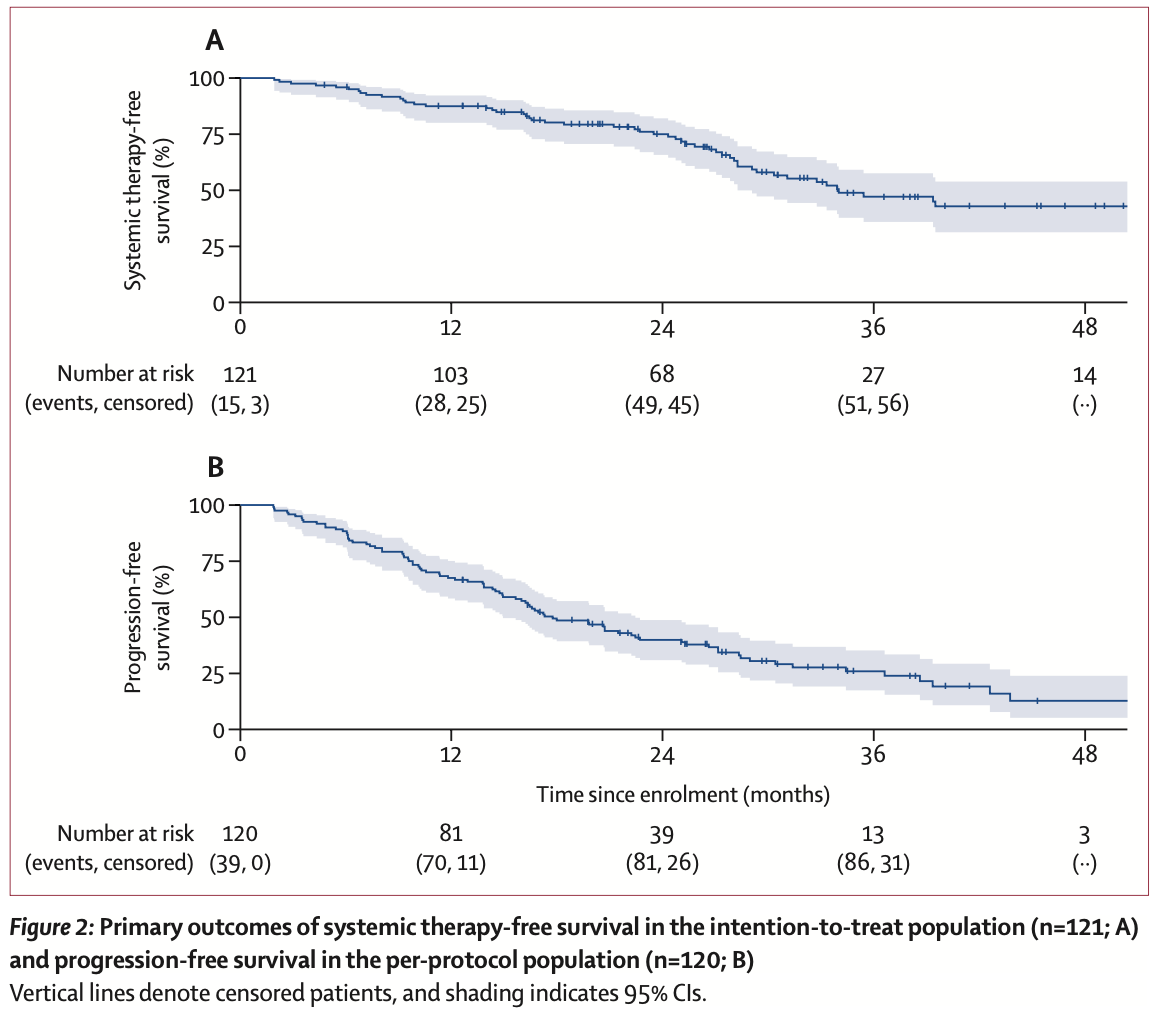
Primary outcomes
Median follow-up: 36.3 months (IQR 26.5–51.1)
Systemic therapy-free survival (co-primary endpoint, intention-to-treat, n=121)
Time from enrollment to initiation of systemic therapy or death from ccRCC
| Median | 34.0 months (95% CI 28.3–54.1) |
| 1-year rate | 87.5% (95% CI 80.0–92.2) |
| Prespecified target | Median ≥ 24 months — exceeded ✓ (lower 95% CI also exceeded) |
| Events | 51 (42%) started systemic therapy; 2 (2%) died from disease |
| Benchmark | Rini et al. surveillance: median 14.9 months (95% CI 10.6–25.0) |
Progression-free survival (co-primary endpoint, per-protocol, n=120)
Time from enrollment to RECIST 1.1 progression, clinical progression, or death from any cause
| Median | 17.7 months (95% CI 14.9–22.4) |
| 1-year rate | 67.5% (95% CI 58.3–75.1) |
| Events | 86/120 (72%) — 73 RECIST progression, 7 clinical progression, 6 death |
| Comparators | Rini surveillance: 9.4 mo; RAPPORT (MDT + pembrolizumab): 15.6 mo |
Secondary outcomes and safety
Overall survival
| Median OS | Not reached |
| 1-year OS | 96.7% (95% CI 91.4–98.7) |
| 3-year OS (post hoc) | 86.5% (95% CI 77.5–92.1) |
| Deaths | 23 total: 14 disease, 9 other causes |
Patterns of progression
| Freedom from new lesions (12-mo) | 72.7% |
| Median time to new lesion | 22.7 months |
| First event: new lesions | 57% of patients |
| First event: local progression | Only 7% — high local control |
Toxicity (per-protocol, n=120)
| Grade ≥ 2 AEs | 25 patients (21%) |
| Grade 3–4 AEs | 8 patients (7%) |
| Treatment-related deaths | 0 (none) |
| Most common grade 3 | Pain (4 events), leukocytosis (2) |
| Grade 4 event | Hyperglycemia (RT to pancreas → insulin-dependent) |
| Grade 2 pneumonitis | 5 patients (4%) |
Key safety takeaway: 7% grade 3+ with no treatment-related deaths — compare to IO combination toxicity (grade 3+ in 46–73% across CheckMate 214, KEYNOTE-426, CLEAR) and metastasectomy complications (45.7% overall, Meyer J Urol 2017)
treatment course of all patients
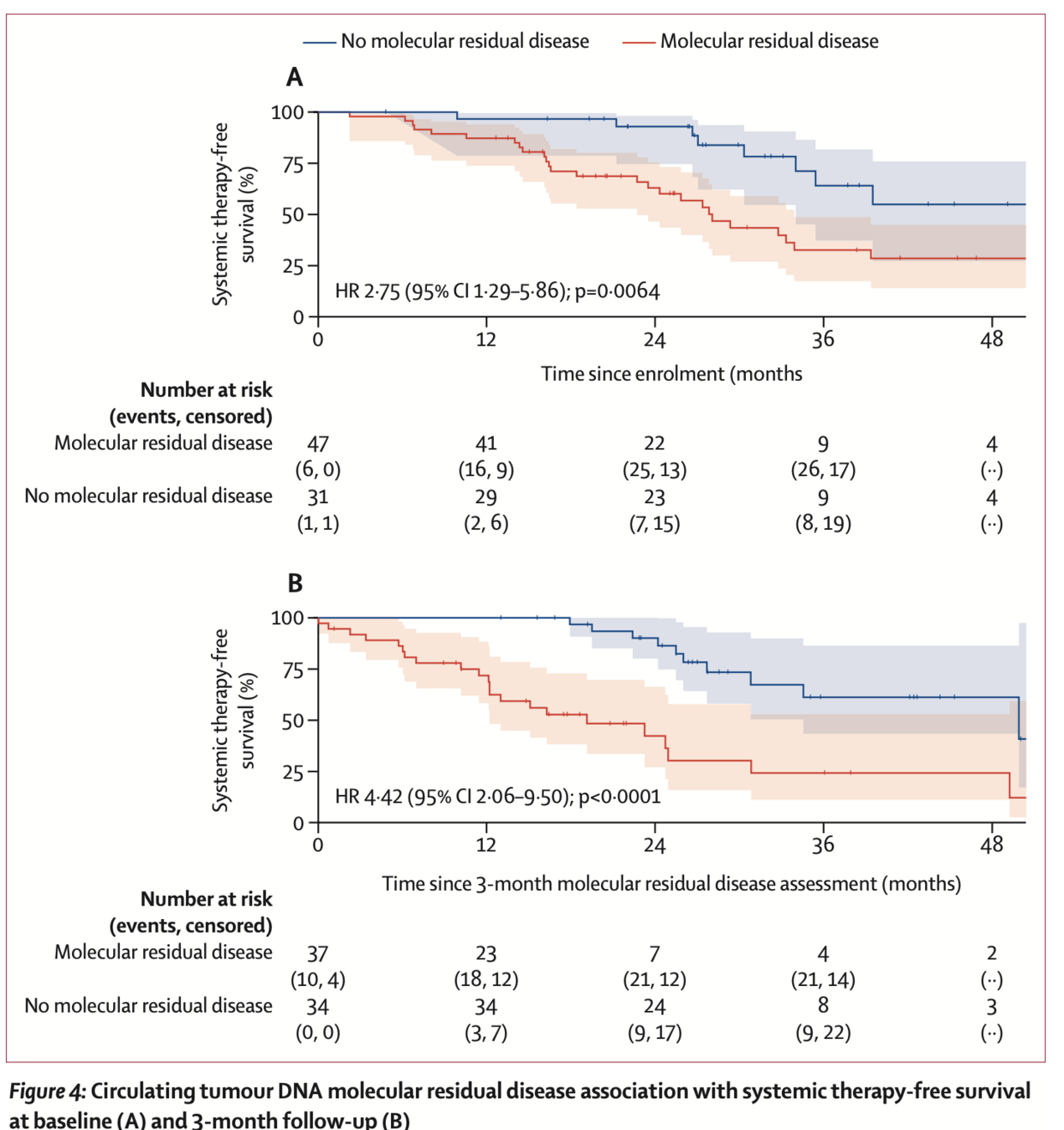
Exploratory: ctDNA molecular residual disease
- Assay: Tumor-informed, patient-specific ctDNA panels via WGS (up to 2000 somatic variants per patient; Precise MRD, Myriad Genetics)
- 87/89 patients had successful panel creation (98%); 149/168 plasma samples analyzed (89%)
- Baseline MRD: 47/78 patients (60%) were ctDNA MRD-positive
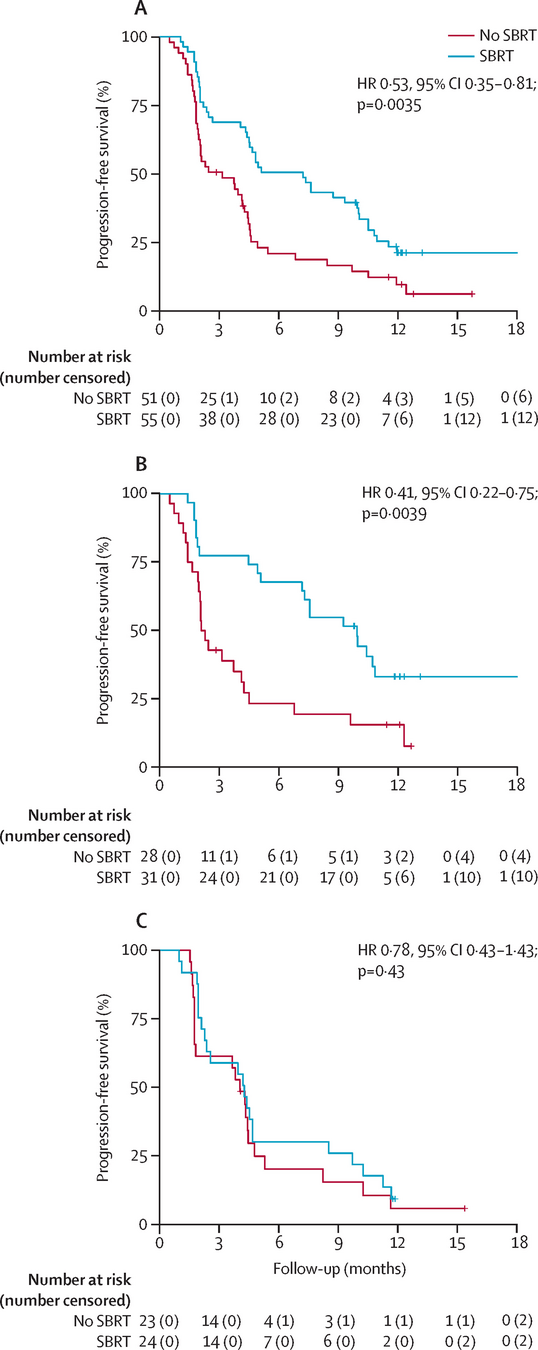
- MRD+ vs MRD−: median STFS 28.1 vs 54.1 months (HR 2.75, p=0.0064)
- 3-month MRD (landmark analysis): 37/71 patients (52%) MRD-positive
- MRD+ vs MRD−: median STFS 19.1 vs 49.9 months (HR 4.42, p<0.0001)
- 25% of baseline MRD+ patients converted to MRD− at 3 months
- Multivariable model: ctDNA MRD status remained independently prognostic after adjusting for # lesions and prior systemic therapy lines (all p<0.05)
ctDNA MRD and systemic therapy-free survival
A) Baseline
ctDNA MRD+ vs MRD− and STFS (HR 2.75, p=0.0064)
B) 3-month
ctDNA MRD+ vs MRD− and STFS (HR 4.42, p<0.0001)]
Prognostic factors for STFS
- Prespecified baseline clinical factors associated with STFS:
- Number of metastatic lesions at enrollment: HR 1.53 per lesion (95% CI 1.14–2.07, p=0.0044)
- Number of previous lines of systemic therapy: HR 1.50 per line (95% CI 1.11–2.04, p=0.0078)
- ctDNA MRD status independently prognostic on multivariable analysis (all p<0.05 with lesion count and prior systemic therapy)
- Factors associated with baseline MRD positivity (not adjusted for multiple testing):
- IMDC risk group (p=0.0034)
- Bone metastases (p=0.038)
- Total irradiated GTV (p=0.0045)
- Notably: factors associated with MRD status were not the same as those associated with STFS, suggesting independent prognostic value
Prostate cancer: the most mature MDT evidence — and model for RCC
Two key questions answered by randomized data: (1) Does MDT beat observation? (2) Should MDT be combined with systemic therapy?
Q1: MDT alone vs observation
| STOMP Ost, JCO 2018; 5-yr ASCO GU 2020 |
Median ADT-free survival: 21 vs 13 mo 5-yr ADT-free: 34% vs 8% (HR 0.57) 5-yr CRPC-free: 76% vs 53% |
| ORIOLE Phillips, JAMA Oncol 2020 |
6-mo progression: 19% vs 61% (p=0.005) PFS: not reached vs 5.8 mo (HR 0.30) |
| Pooled analysis Deek, JCO 2022 |
PFS: 11.9 vs 5.9 mo (HR 0.44, p<0.001) 15–20% durable PFS beyond 4–5 yrs High-risk mutations (ATM/BRCA/Rb1/TP53): HR 0.05 |
Q2: MDT + ADT vs ADT alone
| EXTEND Tang, JAMA Oncol 2023 |
PFS: NR vs 15.8 mo (HR 0.25, p<0.001) Eugonadal PFS: NR vs 6.1 mo (HR 0.32, p=0.03) MDT + intermittent ADT preserves testosterone |
| RADIOSA Marvaso, Lancet Oncol 2025 |
PFS: 32.2 vs 15.1 mo (HR 0.43, p=0.001) SBRT + 6-mo ADT vs SBRT alone First RCT showing ADT adds to SBRT |
| WOLVERINE X-MET, ASCO GU 2025 |
Pooled IPD from 5 RCTs (n=472) 48-mo OS: 87% vs 75% (HR 0.64, p=0.057) First signal of OS benefit with MDT |
The parallel to RCC: Both prostate and RCC have indolent biology in the oligometastatic subset. Both show SBRT can defer systemic therapy. Prostate has randomized data; RCC now has the Tang expansion cohort (n=121, STFS 34 mo). The next step for RCC is what prostate already has — randomized trials (SOAR, ASTROs).
Key difference: In prostate, the systemic therapy being deferred is ADT — with known QoL effects (sexual dysfunction, metabolic syndrome, osteoporosis). In RCC, the deferred therapy is IO combinations — with different but significant toxicity.
Conclusions
- Serial MDT without systemic therapy in oligometastatic ccRCC achieved a median STFS of 34.0 months, exceeding the 24-month prespecified threshold
- Favorable PFS (median 17.7 months)
- OS (median not reached; 3-year OS 86.5%)
- Compared favorably to surveillance alone (Rini: PFS 9.4 mo) and MDT + pembrolizumab (RAPPORT: PFS 15.6 mo, OS 20 mo)
- Modest toxicity profile: grade ≥ 3 in only 7%; no treatment-related deaths
- 59% of patients received ≥ 2 rounds of MDT, demonstrating feasibility of serial approach
- ctDNA MRD status at baseline and 3 months was an independent prognostic biomarker for STFS
- Number of metastatic lesions and prior systemic therapy lines also prognostic
- MDT can enable meaningful treatment de-escalation, potentially improving QoL and reducing costs
Strengths and limitations
- Strengths:
- Largest prospective trial of serial MDT without systemic therapy for oligometastatic ccRCC (n=121)
- Longest follow-up in this setting (median 36.3 months)
- Central radiology review (RECIST 1.1) by subspecialized radiologist
- Novel integration of second-generation ultrasensitive ctDNA assay
- 60% baseline ctDNA detection rate (vs 25% with first-generation assays)
- Prespecified endpoints, futility analyses, and exploratory biomarker analyses
- Limitations:
- Single-arm, single-institution — no randomized comparator
- No quality-of-life data collected
- Systemic therapy initiation partly physician/patient discretion
- RECIST 1.1 developed for systemic therapy assessment — may not optimally capture oligomet MDT response
- ctDNA analysis exploratory and in a subset only; tissue procurement biases
- Predominantly favorable/intermediate IMDC risk and predominantly lung metastases — limits generalizability
Discussion points
- How should radiation oncologists select patients for MDT-only strategies? Should ctDNA MRD testing be integrated into routine practice, and at what time points?
- What are the implications for non-clear-cell histologies, which have fewer effective systemic therapy options? Could MDT have an even greater role?
- In what other histologies can metastasis directed therapy be used to delay systemic therapy vs used to maintain efficacy of systemic therapy?
MDT without systemic therapy for oligometastatic ccRCC (Tang et al., Lancet Oncol 2025)
By RadMedSkiier
MDT without systemic therapy for oligometastatic ccRCC (Tang et al., Lancet Oncol 2025)
Journal club presentation on the MD Anderson Renal Cell Oligometastasis phase 2 trial
- 10



